Abstract
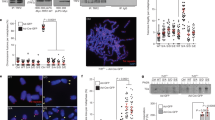
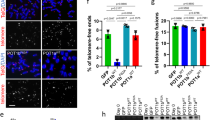
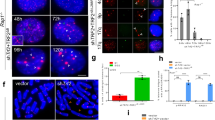
When telomeres are rendered dysfunctional through replicative attrition of the telomeric DNA or by inhibition of shelterin1, cells show the hallmarks of ataxia telangiectasia mutated (ATM) kinase signalling2,3,4. In addition, dysfunctional telomeres might induce an ATM-independent pathway, such as ataxia telangiectasia and Rad3-related (ATR) kinase signalling, as indicated by the phosphorylation of the ATR target CHK1 in senescent cells2,5 and the response of ATM-deficient cells to telomere dysfunction6,7. However, because telomere attrition is accompanied by secondary DNA damage, it has remained unclear whether there is an ATM-independent pathway for the detection of damaged telomeres. Here we show that damaged mammalian telomeres can activate both ATM and ATR and address the mechanism by which the shelterin complex represses these two important DNA damage signalling pathways. We analysed the telomere damage response on depletion of either or both of the shelterin proteins telomeric repeat binding factor 2 (TRF2) and protection of telomeres 1 (POT1) from cells lacking ATM and/or ATR kinase signalling. The data indicate that TRF2 and POT1 act independently to repress these two DNA damage response pathways. TRF2 represses ATM, whereas POT1 prevents activation of ATR. Unexpectedly, we found that either ATM or ATR signalling is required for efficient non-homologous end-joining of dysfunctional telomeres. The results reveal how mammalian telomeres use multiple mechanisms to avoid DNA damage surveillance and provide an explanation for the induction of replicative senescence and genome instability by shortened telomeres.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
de Lange, T. Shelterin: the protein complex that shapes and safeguards human telomeres. Genes Dev. 19, 2100–2110 (2005)
d'Adda di Fagagna, F. et al. A DNA damage checkpoint response in telomere-initiated senescence. Nature 426, 194–198 (2003)
Karlseder, J., Broccoli, D., Dai, Y., Hardy, S. & de Lange, T. p53- and ATM-dependent apoptosis induced by telomeres lacking TRF2. Science 283, 1321–1325 (1999)
Takai, H., Smogorzewska, A. & de Lange, T. DNA damage foci at dysfunctional telomeres. Curr. Biol. 13, 1549–1556 (2003)
Herbig, U., Jobling, W. A., Chen, B. P., Chen, D. J. & Sedivy, J. M. Telomere shortening triggers senescence of human cells through a pathway involving ATM, p53, and p21(CIP1), but not p16(INK4a). Mol. Cell 14, 501–513 (2004)
Qi, L. et al. Short telomeres and ataxia-telangiectasia mutated deficiency cooperatively increase telomere dysfunction and suppress tumorigenesis. Cancer Res. 63, 8188–8196 (2003)
Wong, K. K. et al. Telomere dysfunction and Atm deficiency compromises organ homeostasis and accelerates ageing. Nature 421, 643–648 (2003)
Celli, G. B. & de Lange, T. DNA processing not required for ATM-mediated telomere damage response after TRF2 deletion. Nature Cell Biol. 7, 712–718 (2005)
Smogorzewska, A. & de Lange, T. Different telomere damage signaling pathways in human and mouse cells. EMBO J. 21, 4338–4348 (2002)
Barlow, C. et al. Atm-deficient mice: a paradigm of ataxia telangiectasia. Cell 86, 159–171 (1996)
Lazzerini Denchi, E., Celli, G. & de Lange, T. Hepatocytes with extensive telomere deprotection and fusion remain viable and regenerate liver mass through endoreduplication. Genes Dev. 20, 2648–2653 (2006)
Jazayeri, A. et al. ATM- and cell cycle-dependent regulation of ATR in response to DNA double-strand breaks. Nature Cell Biol. 8, 37–45 (2006)
Dimitrova, N. & de Lange, T. MDC1 accelerates nonhomologous end-joining of dysfunctional telomeres. Genes Dev. 20, 3238–3243 (2006)
Zou, L. & Elledge, S. J. Sensing DNA damage through ATRIP recognition of RPA-ssDNA complexes. Science 300, 1542–1548 (2003)
Bertuch, A. A. & Lundblad, V. The maintenance and masking of chromosome termini. Curr. Opin. Cell Biol. 18, 247–253 (2006)
Enomoto, S., Glowczewski, L. & Berman, J. MEC3, MEC1, and DDC2 are essential components of a telomere checkpoint pathway required for cell cycle arrest during senescence in Saccharomyces cerevisiae. Mol. Biol. Cell 13, 2626–2638 (2002)
Garvik, B., Carson, M. & Hartwell, L. Single-stranded DNA arising at telomeres in cdc13 mutants may constitute a specific signal for the RAD9 checkpoint. Mol. Cell. Biol. 15, 6128–6138 (1995)
Hockemeyer, D., Daniels, J. P., Takai, H. & de Lange, T. Recent expansion of the telomeric complex in rodents: Two distinct POT1 proteins protect mouse telomeres. Cell 126, 63–77 (2006)
Wu, L. et al. Pot1 deficiency initiates DNA damage checkpoint activation and aberrant homologous recombination at telomeres. Cell 126, 49–62 (2006)
Brown, E. J. & Baltimore, D. Essential and dispensable roles of ATR in cell cycle arrest and genome maintenance. Genes Dev. 17, 615–628 (2003)
Hockemeyer, D. et al. Telomere protection by mammalian POT1 requires interaction with TPP1. Nature Struct. Mol. Biol. advanced online publication, doi:10.1038/nsmb1270 (15 July 2007)
Cortez, D., Guntuku, S., Qin, J. & Elledge, S. J. ATR and ATRIP: partners in checkpoint signaling. Science 294, 1713–1716 (2001)
Hockemeyer, D., Sfeir, A. J., Shay, J. W., Wright, W. E. & de Lange, T. POT1 protects telomeres from a transient DNA damage response and determines how human chromosomes end. EMBO J. 24, 2667–2678 (2005)
Karlseder, J. et al. The telomeric protein TRF2 binds the ATM kinase and can inhibit the ATM-dependent DNA damage response. PLoS Biol. 2, E240 (2004)
Loayza, D. & de Lange, T. POT1 as a terminal transducer of TRF1 telomere length control. Nature 423, 1013–1018 (2003)
Celli, G. B., Lazzerini Denchi, E. & de Lange, T. Ku70 stimulates fusion of dysfunctional telomeres yet protects chromosome ends from homologous recombination. Nature Cell Biol. 8, 885–890 (2006)
Griffith, J. D. et al. Mammalian telomeres end in a large duplex loop. Cell 97, 503–514 (1999)
Lobrich, M. & Jeggo, P. A. Harmonising the response to DSBs: a new string in the ATM bow. DNA Repair (Amst.) 4, 749–759 (2005)
Ye, J. Z. et al. POT1-interacting protein PIP1: a telomere length regulator that recruits POT1 to the TIN2/TRF1 complex. Genes Dev. 18, 1649–1654 (2004)
Acknowledgements
We thank D. White for invaluable help in maintaining our mouse colonies, D. Argibay for assistance with genotyping, E. Brown for providing the ATR conditional knockout mice, H. Takai for technical advice and suggestions, D. Hockemeyer for providing Pot1 double knockout cells and the Tpp1 and Pot1a shRNA constructs, S. Soll for his contributions to the initial stages of this research and members of the de Lange laboratory for critical comments on the manuscript. This work was supported by a grant from the NIH.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at www.nature.com/reprints. The authors declare no competing financial interests.
Supplementary information
Supplementary Figures
This file contains Supplementary Figures S1-S9 with Legends. Figures S1-S5 show γH2AX TIFs (S1), ChIP for shelterin (S2), ATR-independent DNA damage response (S3), ATM-dependent NEHJ (S4-S5) following TRF2 deletion. Figures S6-S7 show activation of ATR but not ATM upon POT1 inhibition. Figures S8-S9 show TIF formation and NHEJ in TRF2-/- ATM-/- cells upon POT1 inhibition. (PDF 1740 kb)
Rights and permissions
About this article
Cite this article
Denchi, E., de Lange, T. Protection of telomeres through independent control of ATM and ATR by TRF2 and POT1. Nature 448, 1068–1071 (2007). https://doi.org/10.1038/nature06065
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature06065
This article is cited by
-
Mitochondria and telomeres: hand in glove
Biogerontology (2024)
-
The crosstalk between telomeres and DNA repair mechanisms: an overview to mammalian somatic cells, germ cells, and preimplantation embryos
Journal of Assisted Reproduction and Genetics (2024)
-
Potential effects of assisted reproductive technology on telomere length and telomerase activity in human oocytes and early embryos
Journal of Ovarian Research (2023)
-
The shelterin component TRF2 mediates columnar stacking of human telomeric chromatin
The EMBO Journal (2023)
-
Non-canonical telomere protection role of FOXO3a of human skeletal muscle cells regulated by the TRF2-redox axis
Communications Biology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.