Abstract
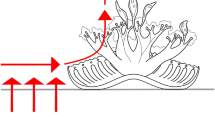
Most bony fishes rely on suction mechanisms to capture and transport prey1. Once captured, prey are carried by water movement inside the oral cavity to a second set of jaws in the throat, the pharyngeal jaws, which manipulate the prey and assist in swallowing1,2. Moray eels display much less effective suction-feeding abilities3. Given this reduction in a feeding mechanism that is widespread and highly conserved in aquatic vertebrates, it is not known how moray eels swallow large fish and cephalopods4,5,6,7. Here we show that the moray eel (Muraena retifera) overcomes reduced suction capacity by launching raptorial pharyngeal jaws out of its throat and into its oral cavity, where the jaws grasp the struggling prey animal and transport it back to the throat and into the oesophagus. This is the first described case of a vertebrate using a second set of jaws to both restrain and transport prey, and is the only alternative to the hydraulic prey transport reported in teleost fishes. The extreme mobility of the moray pharyngeal jaws is made possible by elongation of the muscles that control the jaws8, coupled with reduction of adjacent gill-arch structures9. The discovery that pharyngeal jaws can reach up from behind the skull to grasp prey in the oral jaws reveals a major innovation that may have contributed to the success of moray eels as apex predators hunting within the complex matrix of coral reefs10,11. This alternative prey transport mode is mechanically similar to the ratcheting mechanisms used in snakes12,13—a group of terrestrial vertebrates that share striking morphological, behavioural14 and ecological convergence with moray eels.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Lauder, G. V. in Fish Biomechanics (eds Webb, P. W. & Weihs, D.) 280–311 (Praeger, New York, 1983)
Wainwright, P. C. in Fish Biomechanics (eds Shadwick, R. & Lauder, G. V.) 77–101 (Academic, New York, 2006)
Mehta, R. M. & Wainwright, P. C. Biting releases constraints on moray eel feeding kinematics. J. Exp. Biol. 210, 495–505 (2007)
Randall, J. E. Food habits of reef fishes of the West Indies. Stud. Trop. Oceanogr. 5, 655–847 (1967)
Young, R. F. & Winn, H. E. Activity patterns, diet, and shelter site use for two species of moray eels, Gymnothorax moringa and Gymnothorax vicinus, in Belize. Copeia 2003, 44–55 (2003)
Yukihira, H., Shibuno, T., Hashimoto, H. & Gushima, K. Feeding habits of moray eels (Pisces: Muraenidae) at Kuchierabu-ji. Appl. Biol. Sci. 33, 159–166 (1994)
Parrish, J. D. et al. Piscivory in a coral reef fish community. Environ. Biol. Fish. 14, 285–297 (1986)
Nelson, G. J. Branchial muscles in representatives of five eel families. Pacif. Sci. 21, 348–363 (1967)
Nelson, G. J. Gill arches of Teleostean fishes of the Order Anguilliformes. Pacif. Sci. 20, 391–408 (1966)
Abrams, R. W., Abrams, M. D. & Schein, M. W. Diurnal observations on the behavioral ecology of Gymnothorax moringa (Cuvier) and Muraena miliaris (Kaup) on a Caribbean coral reef. Coral Reefs 1, 185–192 (1983)
Böhlke, E. B., McCosker, J. E. & Böhlke, J. E. in Fishes of the Western North Atlantic (ed. Böhlke, E. B.) 104–206 (Sears Foundation for Marine Research Memoir, Yale Univ., New Haven, Connecticut, 1989)
Cundall, D. & Greene, H. W. in Feeding: Form, Function, and Evolution in Tetrapod Vertebrates (ed. Schwenk, K.) 293–333 (Academic, San Diego, 2000)
Kley, N. J. & Brainerd, E. L. Post-cranial prey transport mechanisms in the black pinesnake, Pituophis melanoleucus lodingi: an x-ray videographic study. Zoology 105, 153–164 (2002)
Miller, T. J. Knotting: a previously undescribed feeding behaviour in Muraenid eels. Copeia 1987, 1055–1057 (1987)
Robins, C. R. in Fishes of the Western North Atlantic (ed. Böhlke, E. B.) 9–23 (Sears Foundation for Marine Research Memoir, Yale Univ., New Haven, Connecticut, 1989)
Gilbert, M., Rasmussen, J. B. & Kramer, D. L. Estimating the density and biomass of moray eels (Muraenidae) using a modified visual census method for hole-dwelling reef fauna. Environ. Biol. Fish. 73, 415–426 (2005)
Hixon, M. A. & Beets, J. P. Predation, prey refuges, and the structure of coral-reef fish assemblages. Ecol. Monogr. 63, 77–101 (1993)
Carr, M. H. & Hixon, M. A. Predation effects on early post-settlement survivorship of coral-reef fishes. Mar. Ecol. Prog. Ser. 124, 31–42 (1995)
Alfaro, M. E., Janovetz, J. & Westneat, M. W. Motor control across trophic strategies: muscle activity of biting and suction feeding fishes. Am. Zool. 41, 1266–1279 (2001)
Gillis, G. B. & Lauder, G. V. Kinematics of feeding in bluegill sunfish: is there a general distinction between aquatic capture and transport behaviors? J. Exp. Biol. 198, 709–720 (1995)
Popta, C. M.-L. Les arcs branchiaux de quelques Muraenidae. Ann. Sci. Nat. 19, 367–390 (1909)
Hulsey, C. D., Fraser, G. J. & Streelman, J. T. Evolution and development of complex biomechanical systems: 300 million years of fish jaws. Zebrafish 2, 243–257 (2005)
Lauder, G. V. Functional design and evolution of the pharyngeal jaw apparatus in euteleostean fishes. Zool. J. Linn. Soc. 77, 1–38 (1983)
Carroll, A. M., Wainwright, P. C., Huskey, S. H., Collar, D. C. & Turingan, R. G. Morphology predicts suction feeding performance in centrarchid fishes. J. Exp. Biol. 207, 3873–3881 (2004)
Richard, B. A. & Wainwright, P. C. Scaling of the feeding mechanism of largemouth bass (Micropterus salmoides): kinematics of prey capture. J. Exp. Biol. 198, 419–433 (1995)
Day, S. W., Higham, T. E., Cheer, A. Y. & Wainwright, P. C. Spatial and temporal flow patterns during suction feeding of bluegill sunfish (Lepomis macrochirus) by Particle Image Velocimetry. J. Exp. Biol. 208, 2661–2671 (2005)
Westneat, M. W. Evolution of levers and linkages in the feeding mechanism of fishes. Integr. Comp. Biol. 44, 378–389 (2004)
Schwenk, K. in Feeding: Form, Function and Evolution in Tetrapod Vertebrates (ed., Schwenk, K.) 21–61 (Academic, San Diego, 2000)
Liem, K. F. Evolutionary strategies and morphological innovations: cichlid pharyngeal jaws. Sys. Zool. 22, 425–441 (1973)
Dingerkus, G. & Uhler, L. D. Enzyme clearing of alcian blue stained whole small vertebrates for demonstration of cartilage. Stain Technol. 52, 229–232 (1977)
Acknowledgements
We thank M. E. Alfaro, D. C. Collar, S. W. Day, H. W. Greene, N. J. Kley, A. H. Krakauer, R. D. Mehta, J. T. Redwine, A. Sinsheimer, T. W. Schoener and K. Vanderveen for comments on various drafts of this manuscript. We thank N. J. Kley for discussions regarding prey transport in vertebrates. H. Tran and L. B. Feng provided assistance with video collections. We are grateful to R. E. Pollard, C. Stafford, T. B. Waltzek and E. R. Wisner for help with radiographs and videofluoroscopy. P. Kysar provided technical assistance with scanning electron microscopy. We thank the California Academy of Sciences and the Harvard Museum of Comparative Zoology for specimen loans. Funding was provided by the American Association of University Women and by the National Science Foundation.
Author Contributions R.S.M. designed research, performed research and wrote the manuscript. P.C.W. helped design the study, contributed to the interpretation of anatomy and video, and participated in writing the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at www.nature.com/reprints. The authors declare no competing financial interests.
Supplementary information
Supplementary Video 1
This file contains Supplementary Video 1. This movie shows a portion of prey transport behaviour of the reticulated moray eel, Muraena retifera. Prey transport in lateral view reveals the upper pharyngeal jaw ensnaring a piece of squid prey apprehended by the oral jaws (MOV 6596 kb)
Supplementary Video 2
This file contains Supplementary Video 2 This movie shows a feeding sequence in lateral view of the reticulated moray eel, Muraena retifera. Morays apprehend prey by biting. During prey transport, the upper pharyngeal jaws ensnare squid prey. (MOV 5353 kb)
Supplementary Video 3
This file contains Supplementary Video 3. This movie shows the reticulated moray eel, Muraena retifera transporting a long piece of squid (5 cm). The feeding sequence is in lateral view and reveals how the reticulated moray can use multiple alternating movements of the oral and pharyngeal jaws to transport long prey. (MOV 54761 kb)
Rights and permissions
About this article
Cite this article
Mehta, R., Wainwright, P. Raptorial jaws in the throat help moray eels swallow large prey. Nature 449, 79–82 (2007). https://doi.org/10.1038/nature06062
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature06062
This article is cited by
-
Aggressive behaviour in moray eels versus species identity: do I oust you, or do I eat you?
Marine Biology (2023)
-
Novel gene rearrangement in the mitochondrial genome of Muraenesox cinereus and the phylogenetic relationship of Anguilliformes
Scientific Reports (2021)
-
Ecological redundancy between coral reef sharks and predatory teleosts
Reviews in Fish Biology and Fisheries (2020)
-
A moray’s many knots: knot tying behaviour around bait in two species of Gymnothorax moray eel
Environmental Biology of Fishes (2016)
-
To knot or not? Novel feeding behaviours in moray eels
Marine Biodiversity (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.