Abstract
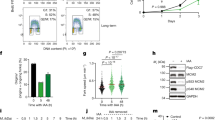
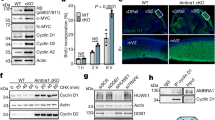
Unicellular organisms such as yeasts require a single cyclin-dependent kinase, Cdk1, to drive cell division1. In contrast, mammalian cells are thought to require the sequential activation of at least four different cyclin-dependent kinases, Cdk2, Cdk3, Cdk4 and Cdk6, to drive cells through interphase, as well as Cdk1 to proceed through mitosis2. This model has been challenged by recent genetic evidence that mice survive in the absence of individual interphase Cdks3,4,5,6,7,8. Moreover, most mouse cell types proliferate in the absence of two or even three interphase Cdks8,9,10. Similar results have been obtained on ablation of some of the activating subunits of Cdks, such as the D-type and E-type cyclins11,12,13,14. Here we show that mouse embryos lacking all interphase Cdks (Cdk2, Cdk3, Cdk4 and Cdk6) undergo organogenesis and develop to midgestation. In these embryos, Cdk1 binds to all cyclins, resulting in the phosphorylation of the retinoblastoma protein pRb and the expression of genes that are regulated by E2F transcription factors. Mouse embryonic fibroblasts derived from these embryos proliferate in vitro, albeit with an extended cell cycle due to inefficient inactivation of Rb proteins. However, they become immortal on continuous passage. We also report that embryos fail to develop to the morula and blastocyst stages in the absence of Cdk1. These results indicate that Cdk1 is the only essential cell cycle Cdk. Moreover, they show that in the absence of interphase Cdks, Cdk1 can execute all the events that are required to drive cell division.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Bloom, J. & Cross, F. R. Multiple levels of cyclin specificity in cell-cycle control. Nature Rev. Mol. Cell Biol. 2, 149–160 (2007)
Malumbres, M. & Barbacid, M. Mammalian cyclin-dependent kinases. Trends Biochem. Sci. 30, 630–641 (2005)
Ye, X., Zhu, C. & Harper, J. W. A premature-termination mutation in the Mus musculus cyclin-dependent kinase 3 gene. Proc. Natl Acad. Sci. USA 98, 1682–1686 (2001)
Rane, S. G. et al. Loss of Cdk4 expression causes insulin-deficient diabetes and Cdk4 activation results in β-cell hyperplasia. Nature Genet. 22, 44–52 (1999)
Tsutsui, T. et al. Targeted disruption of Cdk4 delays cell cycle entry with enhanced p27Kip1 activity. Mol. Cell. Biol. 19, 7011–7019 (1999)
Ortega, S. et al. Cyclin-dependent kinase 2 is essential for meiosis but not for mitotic cell division in mice. Nature Genet. 35, 25–31 (2003)
Berthet, C., Aleem, E., Coppola, V., Tessarollo, L. & Kaldis, P. Cdk2 knockout mice are viable. Curr. Biol. 13, 1775–1785 (2003)
Malumbres, M. et al. Mammalian cells cycle without the D-type Cyclin-dependent kinases Cdk4 and Cdk6. Cell 118, 493–504 (2004)
Berthet, C. et al. Combined loss of Cdk2 and Cdk4 results in embryonic lethality and Rb hypophosphorylation. Dev. Cell 10, 563–573 (2006)
Barriere, C. et al. Mice thrive without Cdk4 and Cdk2. Mol. Oncology 1, 72–83 (2007)
Kozar, K. et al. Mouse development and cell proliferation in the absence of D-cyclins. Cell 118, 477–491 (2004)
Geng, Y. et al. Kinase-independent function of cyclin E. Mol. Cell 25, 127–139 (2007)
Geng, Y. et al. Cyclin E ablation in the mouse. Cell 114, 431–443 (2003)
Parisi, T. et al. Cyclins E1 and E2 are required for endoreplication in placental trophoblast giant cells. EMBO J. 22, 4794–4803 (2003)
Zarkowska, T. & Mittnacht, S. Differential phosphorylation of the retinoblastoma protein by G1/S cyclin-dependent kinases. J. Biol. Chem. 272, 12738–12746 (1997)
Aleem, E., Kiyokawa, H. & Kaldis, P. Cdc2-Cyclin E complexes regulate the G1/S phase transition. Nature Cell Biol. 7, 831–836 (2005)
Saenz-Robles, M. T., Symonds, H., Chen, J. & Van Dyke, T. Induction versus progression of brain tumor development: differential functions for the pRB- and p53-targeting domains of simian virus 40 T antigen. Mol. Cell. Biol. 14, 2686–2698 (1994)
Fung, T. K. & Poon, R. Y. A roller coaster ride with the mitotic cyclins. Semin. Cell Dev. Biol. 16, 335–342 (2005)
Sutherland, H. G. et al. Large-scale identification of mammalian proteins localized to nuclear sub-compartments. Hum. Mol. Genet. 10, 1995–2011 (2001)
Schnutgen, F. et al. Genomewide production of multipurpose alleles for the functional analysis of the mouse genome. Proc. Natl Acad. Sci. USA 102, 7221–7226 (2005)
Murphy, M. et al. Delayed early embryonic lethality following disruption of the murine cyclin A2 gene. Nature Genet. 15, 83–86 (1997)
Brandeis, M. et al. Cyclin B2-null mice develop normally and are fertile whereas cyclin B1-null mice die in utero. Proc. Natl Acad. Sci. USA 95, 4344–4349 (1998)
Martin, A. et al. Cell cycle inhibition and tumor suppression by p21Cip1 and p27Kip1 are independent of Cdk2. Cancer Cell 7, 591–598 (2005)
Livak, K. J. & Schmittgen, T. D. Analysis of relative gene expression data using real-time quantitative PCR and the 2 - Δ Δ C T method. Methods 25, 402–408 (2001)
Acknowledgements
This work is dedicated to the memory of our colleagues L. Gómez García and J. López Navarro. We thank R. González, M. San Román, B. Velasco and R. Villar for technical assistance, A. García for help with the cytometer, and A. Swat and S. Mouron for advice on quantitative RT–PCR. We also value the support provided by the Transgenic and Comparative Pathology Units of the CNIO. We thank M. Puyol for the Cdk2 and Cdk6 antibodies. This work was supported by grants from the Plan Nacional de Investigación Científica (D.S., M.M. and M.B.), the OncoCycle programme from the Comunidad de Madrid (M.M. and M.B.), Fondo de Investigación Sanitaria (D.S.), V Framework Programme of the European Union (M.B.) and INSERM and Association pour la Recherche contre le Cancer (P.D.). C.T., C.B. and A.C. were supported by fellowships from FEBS, la Ligue contre le Cancer (Comité de la Dordogne) and FPI (Ministerio de Educación y Ciencia), respectively.
Author Contributions M.B. supervised the entire project. M.B., D.S. and M.M. conceived and designed the experiments, and wrote the manuscript with comments from co-authors. C.B. generated the mouse strains and carried out most cell culture experiments and part of the protein analysis. A.C. was responsible for most protein analysis and part of the cell culture experiments. S.H. and C.T. characterized Cdc2a mutant mice. P.D. was responsible for histopathological analysis. J.F.C. and K.N. generated one of the Cdc2a mutant embryonic stem cell clones.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at www.nature.com/reprints. The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Figures S1-S3 with Legends and Supplementary Table S1. (PDF 801 kb)
Rights and permissions
About this article
Cite this article
Santamaría, D., Barrière, C., Cerqueira, A. et al. Cdk1 is sufficient to drive the mammalian cell cycle. Nature 448, 811–815 (2007). https://doi.org/10.1038/nature06046
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature06046
This article is cited by
-
CDCA8 Facilitates Tumor Proliferation and Predicts a Poor Prognosis in Hepatocellular Carcinoma
Applied Biochemistry and Biotechnology (2024)
-
Knockdown of MRPL35 promotes cell apoptosis and inhibits cell proliferation in non-small-cell lung cancer
BMC Pulmonary Medicine (2023)
-
HADHA promotes ovarian cancer outgrowth via up-regulating CDK1
Cancer Cell International (2023)
-
PIN1 and CDK1 cooperatively govern pVHL stability and suppressive functions
Cell Death & Differentiation (2023)
-
Loss of phosphatase CTDNEP1 potentiates aggressive medulloblastoma by triggering MYC amplification and genomic instability
Nature Communications (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.