Abstract
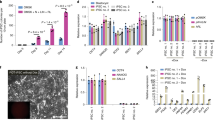
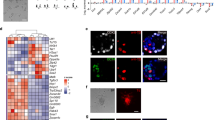
The application of human embryonic stem (ES) cells in medicine and biology has an inherent reliance on understanding the starting cell population. Human ES cells differ from mouse ES cells and the specific embryonic origin of both cell types is unclear. Previous work suggested that mouse ES cells could only be obtained from the embryo before implantation in the uterus1,2,3,4,5. Here we show that cell lines can be derived from the epiblast, a tissue of the post-implantation embryo that generates the embryo proper. These cells, which we refer to as EpiSCs (post-implantation epiblast-derived stem cells), express transcription factors known to regulate pluripotency, maintain their genomic integrity, and robustly differentiate into the major somatic cell types as well as primordial germ cells. The EpiSC lines are distinct from mouse ES cells in their epigenetic state and the signals controlling their differentiation. Furthermore, EpiSC and human ES cells share patterns of gene expression and signalling responses that normally function in the epiblast. These results show that epiblast cells can be maintained as stable cell lines and interrogated to understand how pluripotent cells generate distinct fates during early development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Evans, M. J. & Kaufman, M. H. Establishment in culture of pluripotential cells from mouse embryos. Nature 292, 154–156 (1981)
Martin, G. R. Isolation of a pluripotent cell line from early mouse embryos cultured in medium conditioned by teratocarcinoma stem cells. Proc. Natl Acad. Sci. USA 78, 7634–7638 (1981)
Brook, F. A. & Gardner, R. L. The origin and efficient derivation of embryonic stem cells in the mouse. Proc. Natl Acad. Sci. USA 94, 5709–5712 (1997)
Chung, Y. et al. Embryonic and extraembryonic stem cell lines derived from single mouse blastomeres. Nature 439, 216–219 (2006)
Tesar, P. J. Derivation of germ-line-competent embryonic stem cell lines from preblastocyst mouse embryos. Proc. Natl Acad. Sci. USA 102, 8239–8244 (2005)
Gardner, R. L. Origin and differentiation of extraembryonic tissues in the mouse. Int. Rev. Exp. Pathol. 24, 63–133 (1983)
Surani, M. A., Hayashi, K. & Hajkova, P. Genetic and epigenetic regulators of pluripotency. Cell 128, 747–762 (2007)
Pelton, T. A., Sharma, S., Schulz, T. C., Rathjen, J. & Rathjen, P. D. Transient pluripotent cell populations during primitive ectoderm formation: correlation of in vivo and in vitro pluripotent cell development. J. Cell Sci. 115, 329–339 (2002)
Azuara, V. et al. Chromatin signatures of pluripotent cell lines. Nature Cell Biol. 8, 532–538 (2006)
Bernstein, B. E. et al. A bivalent chromatin structure marks key developmental genes in embryonic stem cells. Cell 125, 315–326 (2006)
Boyer, L. A. et al. Core transcriptional regulatory circuitry in human embryonic stem cells. Cell 122, 947–956 (2005)
Loh, Y. H. et al. The Oct4 and Nanog transcription network regulates pluripotency in mouse embryonic stem cells. Nature Genet. 38, 431–440 (2006)
Yeom, Y. I. et al. Germline regulatory element of Oct-4 specific for the totipotent cycle of embryonal cells. Development 122, 881–894 (1996)
Lawson, K. A., Meneses, J. J. & Pedersen, R. A. Clonal analysis of epiblast fate during germ layer formation in the mouse embryo. Development 113, 891–911 (1991)
Gardner, R. L., Lyon, M. F., Evans, E. P. & Burtenshaw, M. D. Clonal analysis of X-chromosome inactivation and the origin of the germ line in the mouse embryo. J. Embryol. Exp. Morphol. 88, 349–363 (1985)
Kubo, A. et al. Development of definitive endoderm from embryonic stem cells in culture. Development 131, 1651–1662 (2004)
Cai, J. et al. Assessing self-renewal and differentiation in human embryonic stem cell lines. Stem Cells 24, 516–530 (2006)
Liu, Y. et al. Genome wide profiling of human embryonic stem cells (hESCs), their derivatives and embryonal carcinoma cells to develop base profiles of U.S. Federal government approved hESC lines. BMC Dev. Biol. 6, 20 (2006)
Wang, J. et al. A protein interaction network for pluripotency of embryonic stem cells. Nature 444, 364–368 (2006)
Yu, R. N., Ito, M., Saunders, T. L., Camper, S. A. & Jameson, J. L. Role of Ahch in gonadal development and gametogenesis. Nature Genet. 20, 353–357 (1998)
Hayashi, K., de Sousa Lopes, S. M. & Surani, M. A. Germ cell specification in mice. Science 316, 394–396 (2007)
Lawson, K. A. et al. Bmp4 is required for the generation of primordial germ cells in the mouse embryo. Genes Dev. 13, 424–436 (1999)
Camus, A., Perea-Gomez, A., Moreau, A. & Collignon, J. Absence of Nodal signaling promotes precocious neural differentiation in the mouse embryo. Dev. Biol. 295, 743–755 (2006)
Vallier, L., Reynolds, D. & Pedersen, R. A. Nodal inhibits differentiation of human embryonic stem cells along the neuroectodermal default pathway. Dev. Biol. 275, 403–421 (2004)
James, D., Levine, A. J., Besser, D. & Hemmati-Brivanlou, A. TGFβ/activin/nodal signaling is necessary for the maintenance of pluripotency in human embryonic stem cells. Development 132, 1273–1282 (2005)
Niwa, H., Burdon, T., Chambers, I. & Smith, A. Self-renewal of pluripotent embryonic stem cells is mediated via activation of STAT3. Genes Dev. 12, 2048–2060 (1998)
Daheron, L. et al. LIF/STAT3 signaling fails to maintain self-renewal of human embryonic stem cells. Stem Cells 22, 770–778 (2004)
Androutsellis-Theotokis, A. et al. Notch signalling regulates stem cell numbers in vitro and in vivo. Nature 442, 823–826 (2006)
Ogawa, K. et al. Activin–Nodal signaling is involved in propagation of mouse embryonic stem cells. J. Cell Sci. 120, 55–65 (2007)
Keller, G. Embryonic stem cell differentiation: emergence of a new era in biology and medicine. Genes Dev. 19, 1129–1155 (2005)
Downs, K. M. In vitro methods for studying vascularization of the murine allantois and allantoic union with the chorion. Methods. Mol. Med. 121, 241–72 (2006)
Beddington, R. S. Isolation, culture and manipulation of post-implantation mouse embryos (ed. Monk, M.) (IRL Press, Oxford, 1987)
Nagy, A. Manipulating the mouse embryo: a laboratory manual (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, 2003)
Zvetkova, I. et al. Global hypomethylation of the genome in XX embryonic stem cells. Nat. Genet. 37, 1274–1279 (2005)
Robertson, E. J. Teratocarcinomas and embryonic stem cells: a practical approach (IRL, Oxford; Washington DC, 1987)
Deome, K. B., Faulkin, L. J., Bern, H. A. & Blair, P. B. Development of mammary tumors from hyperplastic alveolar nodules transplanted into gland-free mammary fat pads of female C3H mice. Cancer. Res. 19, 515–520 (1959)
Hirst, C. E. et al. Transcriptional profiling of mouse and human ESCs identifies SLAIN1, a novel stem cell gene. Dev. Biol. 293, 90–103 (2006)
Kent, W. J. et al. The human genome browser at UCSC. Genome. Res. 12, 996–1006 (2002)
Acknowledgements
We thank J. Battey and Y. Ninomiya for comments on the manuscript and C. Graham, P. Fairchild, J. Pickel, K. Downs, W. Ferguson, and members of the McKay and Gardner labs and the NIH Stem Cell Unit for their contributions to this work. This research was supported by the Intramural Research Programs of the NIH–NINDS and NIH–NCI and by the Wellcome Trust. R.L.G. acknowledges the Royal Society for support. P.J.T. is an NIH–Oxford Biomedical Research Scholar.
Author Contributions P.J.T. derived the EpiSC lines. J.G.C. and P.J.T. carried out in vitro experiments. F.A.B., R.L.G. and P.J.T. derived the mouse ES cells. P.J.T., F.A.B. and T.J.D. performed chimera experiments. P.J.T. and D.L.M. performed teratoma experiments. P.J.T. and E.P.E. performed karyotypic analysis. P.J.T., J.G.C., and R.D.G.M. analysed the data and wrote the paper.
Microarray data are available at the Gene Expression Omnibus website (www.ncbi.nlm.nih.gov/geo) under accession number GSE7902.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
Microarray data are available at the Gene Expression Omnibus website (www.ncbi.nlm.nih.gov/geo) under accession number GSE7902. Reprints and permissions information is available at www.nature.com/reprints. The authors declare no competing financial interests.
Supplementary information
Supplementary Information 1
This file contains Supplementary Figures 1-9 with Legends, Supplementary Table 1, and additional references. (PDF 3597 kb)
Rights and permissions
About this article
Cite this article
Tesar, P., Chenoweth, J., Brook, F. et al. New cell lines from mouse epiblast share defining features with human embryonic stem cells. Nature 448, 196–199 (2007). https://doi.org/10.1038/nature05972
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature05972
This article is cited by
-
Establishment of African pygmy mouse induced pluripotent stem cells using defined doxycycline inducible transcription factors
Scientific Reports (2024)
-
3D Enhancer–promoter networks provide predictive features for gene expression and coregulation in early embryonic lineages
Nature Structural & Molecular Biology (2024)
-
c-Jun as a one-way valve at the naive to primed interface
Cell & Bioscience (2023)
-
Modelling in vitro gametogenesis using induced pluripotent stem cells: a review
Cell Regeneration (2023)
-
Cellular population dynamics shape the route to human pluripotency
Nature Communications (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.