Abstract
Muscle contraction is triggered by the opening of acetylcholine receptors at the vertebrate nerve–muscle synapse1,2,3,4. The M2 helix of this allosteric membrane protein lines the channel, and contains a ‘gate’ that regulates the flow of ions through the pore. We used single-molecule kinetic analysis to probe the transition state of the gating conformational change and estimate the relative timing of M2 motions in the α-subunit of the murine acetylcholine receptor5. This analysis produces a ‘Φ-value’ for a given residue that reflects its open-like versus closed-like character at the transition state. Here we show that most of the residues throughout the length of M2 have a Φ-value of ∼0.64 but that some near the middle have lower Φ-values of 0.52 or 0.31, suggesting that αM2 moves in three discrete steps. The core of the channel serves both as a gate that regulates ion flow and as a hub that directs the propagation of the gating isomerization through the membrane domain of the acetylcholine receptor.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Edelstein, S. & Changeux, J.-P. Allosteric transitions of the acetylcholine receptor. Adv. Protein Chem. 51, 121–184 (1998)
Karlin, A. Emerging structure of the nicotinic acetylcholine receptors. Nature Rev. Neurosci. 3, 102–114 (2002)
Lester, H. A., Dibas, M. I., Dahan, D. S., Leite, J. F. & Dougherty, D. A. Cys-loop receptors: New twists and turns. Trends Neurosci. 27, 329–336 (2004)
Sine, S. M. & Engel, A. G. Recent advances in Cys-loop receptor structure and function. Nature 440, 448–455 (2006)
Zhou, Y., Pearson, J. E. & Auerbach, A. Φ-Value analysis of a linear, sequential reaction mechanism: Theory and application to ion channel gating. Biophys. J. 89, 3680–3685 (2005)
Miyazawa, A., Fujiyoshi, Y. & Unwin, N. Structure and gating mechanism of the acetylcholine receptor pore. Nature 423, 949–955 (2003)
Unwin, N. Refined structure of the nicotinic acetylcholine receptor at 4 Å resolution. J. Mol. Biol. 346, 967–989 (2005)
Akabas, M. H., Kaufmann, C., Archdeacon, P. & Karlin, A. Identification of acetylcholine receptor channel-lining residues in the entire M2 segment of the α-subunit. Neuron 13, 919–927 (1994)
Wilson, G. G. & Karlin, A. The location of the gate in the acetylcholine receptor channel. Neuron 20, 1269–1281 (1998)
Paas, Y. et al. Pore conformations and gating mechanism of a Cys-loop receptor. Proc. Natl Acad. Sci. USA 102, 15877–15882 (2005)
Bali, M. & Akabas, H. The location of closed channel gate in Cys-loop receptor channels. J. Gen. Physiol. (in the press).
Panicker, S., Cruz, H., Arrabit, C. & Slesinger, P. A. Evidence for a centrally located gate in the pore of a serotonin-gated ion channel. J. Neurosci. 22, 1629–1639 (2002)
Beckstein, O. & Sansom, M. S. P. A hydrophobic gate in an ion channel: The closed state of the nicotinic acetylcholine receptor. Phys. Biol. 3, 147–159 (2006)
Labarca, C. et al. Channel gating governed symmetrically by conserved leucine residues in the M2 domain of nicotinic receptors. Nature 376, 514–516 (1995)
Pascual, J. M. & Karlin, A. State-dependent accessibility and electrostatic potential in the channel of the acetylcholine receptor. inferences from rates of reaction of thiosulfonates with substituted cysteines in the M2 segment of the α subunit. J. Gen. Physiol. 111, 717–739 (1998)
Leffler, J. E. & Grunwald, E. Rates and Equilibria of Organic Reactions as Treated by Statistical, Thermodynamic, and Extrathermodynamic Methods (Wiley, New York, 1963)
Mitra, A., Cymes, G. D. & Auerbach, A. Dynamics of the acetylcholine receptor pore at the gating transition state. Proc. Natl Acad. Sci. USA 102, 15069–15074 (2005)
Cymes, G. D., Grosman, C. & Auerbach, A. Structure of the transition state of gating in the acetylcholinereceptor channel pore: A Φ-value analysis. Biochemistry 41, 5548–5555 (2002)
Villarroel, A., Herlitze, S., Koenen, M. & Sakmann, B. Location of a threonine residue in the α-subunit M2 transmembrane segment that determines the ion flow through the acetylcholine receptor channel. Proc. R. Soc. Lond. B 243, 69–74 (1991)
Zhang, H. & Karlin, A. Contribution of the beta subunit M2 segment to the ion-conducting pathway of the acetylcholine receptor. Biochemistry 37, 7952–7964 (1998)
Chakrapani, S., Bailey, T. D. & Auerbach, A. The role of loop 5 in acetylcholine receptor channel gating. J. Gen. Physiol. 122, 521–539 (2003)
Cadugan, D. J. & Auerbach, A. Conformational dynamics of the αM3 transmembrane helix during acetylcholine receptor channel gating. Biophys. J. (in the press).
Mitra, A., Bailey, T. D. & Auerbach, A. L. Structural dynamics of the M4 transmembrane segment during acetylcholine receptor gating. Structure 12, 1909–1918 (2004)
Auerbach, A. Gating of acetylcholine receptor channels: Brownian motion across a broad transition state. Proc. Natl Acad. Sci. USA 102, 1408–1412 (2005)
Grosman, C., Zhou, M. & Auerbach, A. Mapping the conformational wave of acetylcholine receptor channel gating. Nature 403, 773–776 (2000)
Chakrapani, S., Bailey, T. D. & Auerbach, A. Gating dynamics of the acetylcholine receptor extracellular domain. J. Gen. Physiol. 123, 341–356 (2004)
Lummis, S. C. R. et al. Cis-trans isomerization at a proline opens the pore of a neurotransmitter-gated ion channel. Nature 438, 248–252 (2005)
Lee, W. Y. & Sine, S. M. Principal pathway coupling agonist binding to channel gating in nicotinic receptors. Nature 438, 243–247 (2005)
Chakrapani, S. & Auerbach, A. A speed limit for conformational change of an allosteric membrane protein. Proc. Natl Acad. Sci. USA 102, 87–92 (2005)
Acknowledgements
We thank C. Grosman, P. Gottlieb and F. Sachs for discussions, and C. Nicolai, B. Steidl, M. Merritt and M. Teeling for technical assistance. This work was supported by the NIH.
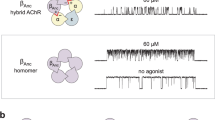
Author Contributions All of the experiments and data analyses shown in Fig. 2 were performed by P.P. except for those for positions 4′ and 9′, which were performed by A.M.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at www.nature.com/reprints. The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Methods, Supplementary Tables 1-2, Supplementary Figures 1-2 with Legends and additional references. (PDF 298 kb)
Rights and permissions
About this article
Cite this article
Purohit, P., Mitra, A. & Auerbach, A. A stepwise mechanism for acetylcholine receptor channel gating. Nature 446, 930–933 (2007). https://doi.org/10.1038/nature05721
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature05721
This article is cited by
-
Molecular mechanisms underlying menthol binding and activation of TRPM8 ion channel
Nature Communications (2020)
-
Thermophoretic analysis of ligand-specific conformational states of the inhibitory glycine receptor embedded in copolymer nanodiscs
Scientific Reports (2020)
-
The conformational wave in capsaicin activation of transient receptor potential vanilloid 1 ion channel
Nature Communications (2018)
-
Understand spiciness: mechanism of TRPV1 channel activation by capsaicin
Protein & Cell (2017)
-
Mechanical coupling maintains the fidelity of NMDA receptor–mediated currents
Nature Neuroscience (2014)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.