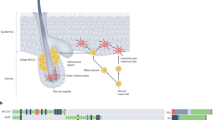
Abstract
Melanocytes are phenotypically prominent but histologically inconspicuous skin cells. They are responsible for the pigmentation of skin and hair, and thereby contribute to the appearance of skin and provide protection from damage by ultraviolet radiation. Pigmentation mutants in various species are highly informative about basic genetic and developmental pathways, and provide important clues to the processes of photoprotection, cancer predisposition and even human evolution. Skin is the most common site of cancer in humans. Continued understanding of melanocyte contributions to skin biology will hopefully provide new opportunities for the prevention and treatment of skin diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Fitzpatrick, T. B. The validity and practicality of sun-reactive skin types I through VI. Arch. Dermatol. 124, 869–871 (1988).
Ziegler, I. The pteridine pathway in zebrafish: regulation and specification during the determination of neural crest cell-fate. Pigment Cell Res. 16, 172–182 (2003).
Rees, J. L. Genetics of hair and skin color. Annu. Rev. Genet. 37, 67–90 (2003).
Searle, A. G. An extension series in the mouse. J. Hered. 59, 341–342 (1968).
Robbins, L. S. et al. Pigmentation phenotypes of variant extension locus alleles result from point mutations that alter MSH receptor function. Cell 72, 827–834 (1993).
Everts, R. E., Rothuizen, J. & van Oost, B. A. Identification of a premature stop codon in the melanocyte-stimulating hormone receptor gene (MC1R) in labrador and golden retrievers with yellow coat colour. Anim. Genet. 31, 194–199 (2000).
Balthasar, N. et al. Divergence of melanocortin pathways in the control of food intake and energy expenditure. Cell 123, 493–505 (2005).
Mountjoy, K. G., Robbins, L. S., Mortrud, M. T. & Cone, R. D. The cloning of a family of genes that encode the melanocortin receptors. Science 257, 1248–1251 (1992).
Levy, C., Khaled, M. & Fisher, D. E. MITF: master regulator of melanocyte development and melanoma oncogene. Trends Mol. Med. 12, 406–414(2006).
D'Orazio, J. A. et al. Topical drug rescue strategy and skin protection based on the role of Mc1r in UV-induced tanning. Nature 443, 340–344 (2006).
Barsh, G., Gunn, T., He, L., Schlossman, S. & Duke-Cohan, J. Biochemical and genetic studies of pigment-type switching. Pigment Cell Res. 13 (Suppl. 8), 48–53 (2000).
Furumura, M., Sakai, C., Abdel-Malek, Z., Barsh, G. S. & Hearing, V. J. The interaction of agouti signal protein and melanocyte stimulating hormone to regulate melanin formation in mammals. Pigment Cell Res. 9, 191–203 (1996).
Kanetsky, P. A. et al. A polymorphism in the agouti signaling protein gene is associated with human pigmentation. Am. J. Hum. Genet. 70, 770–775 (2002).
Rana, B. K. et al. High polymorphism at the human melanocortin 1 receptor locus. Genetics 151, 1547–1557 (1999).
Healy, E. et al. Functional variation of MC1R alleles from red-haired individuals. Hum. Mol. Genet. 10, 2397–2402 (2001).
Ringholm, A. et al. Pharmacological characterization of loss of function mutations of the human melanocortin 1 receptor that are associated with red hair. J. Invest. Dermatol. 123, 917–923 (2004).
Naysmith, L. et al. Quantitative measures of the effect of the melanocortin 1 receptor on human pigmentary status. J. Invest. Dermatol. 122, 423–428 (2004).
Harding, R. M. et al. Evidence for variable selective pressures at MC1R. Am. J. Hum. Genet. 66, 1351–1361 (2000).
Mogil, J. S. et al. The melanocortin-1 receptor gene mediates female-specific mechanisms of analgesia in mice and humans. Proc. Natl Acad. Sci. USA 100, 4867–4872 (2003).
Lamason, R. L. et al. SLC24A5, a putative cation exchanger, affects pigmentation in zebrafish and humans. Science 310, 1782–1786 (2005).
Slominski, A., Tobin, D. J., Shibahara, S. & Wortsman, J. Melanin pigmentation in mammalian skin and its hormonal regulation. Physiol. Rev. 84, 1155–1228 (2004).
Land, E. J. & Riley, P. A. Spontaneous redox reactions of dopaquinone and the balance between the eumelanic and phaeomelanic pathways. Pigment Cell Res. 13, 273–277 (2000).
Oetting, W. S., Fryer, J. P., Shriram, S. & King, R. A. Oculocutaneous albinism type 1: the last 100 years. Pigment Cell Res. 16, 307–311 (2003).
Wei, M. L. Hermansky–Pudlak syndrome: a disease of protein trafficking and organelle function. Pigment Cell Res. 19, 19–42 (2006).
Boissy, R. E. Melanosome transfer to and translocation in the keratinocyte. Exp. Dermatol. 12 (Suppl. 2), 5–12 (2003).
Heenen, M., Giacomoni, P. U. & Golstein, P. Individual variations in the correlation between erythemal threshold, UV-induced DNA damage and sun-burn cell formation. J. Photochem. Photobiol. B 63, 84–87 (2001).
Tsatmali, M., Ancans, J., Yukitake, J. & Thody, A. J. Skin POMC peptides: their actions at the human MC-1 receptor and roles in the tanning response. Pigment Cell Res. 13 (Suppl. 8), 125–129 (2000).
Paus, R. et al. The skin POMC system (SPS). Leads and lessons from the hair follicle. Ann. NY Acad. Sci. 885, 350–363 (1999).
Schauer, E. et al. Proopiomelanocortin-derived peptides are synthesized and released by human keratinocytes. J. Clin. Invest. 93, 2258–2262 (1994).
Krude, H. et al. Severe early-onset obesity, adrenal insufficiency and red hair pigmentation caused by POMC mutations in humans. Nature Genet. 19, 155–157 (1998).
Eller, M. S., Yaar, M. & Gilchrest, B. A. DNA damage and melanogenesis. Nature 372, 413–414 (1994).
Eller, M. S., Ostrom, K. & Gilchrest, B. A. DNA damage enhances melanogenesis. Proc. Natl Acad. Sci. USA 93, 1087–1092 (1996).
Corre, S. et al. UV-induced expression of key component of the tanning process, the POMC and MC1R genes, is dependent on the p-38-activated upstream stimulating factor-1 (USF-1). J. Biol. Chem. 279, 51226–51233 (2004).
Smalley, K. & Eisen, T. The involvement of p38 mitogen-activated protein kinase in the alpha-melanocyte stimulating hormone (alpha-MSH)-induced melanogenic and anti-proliferative effects in B16 murine melanoma cells. FEBS Lett. 476, 198–202 (2000).
Ancans, J., Flanagan, N., Hoogduijn, M. J. & Thody, A. J. P-locus is a target for the melanogenic effects of MC-1R signaling: a possible control point for facultative pigmentation. Ann. NY Acad. Sci. 994, 373–377 (2003).
Suzuki, K., Ojima, M., Kodama, S. & Watanabe, M. Radiation-induced DNA damage and delayed induced genomic instability. Oncogene 22, 6988–6993 (2003).
Ziegler, A. et al. Sunburn and p53 in the onset of skin cancer. Nature 372, 773–776 (1994).
Kamb, A. Sun protection factor p53. Nature 372, 730–731 (1994).
Kaidbey, K. H., Agin, P. P., Sayre, R. M. & Kligman, A. M. Photoprotection by melanin — a comparison of black and Caucasian skin. J. Am. Acad. Dermatol. 1, 249–260 (1979).
Kollias, N., Sayre, R. M., Zeise, L. & Chedekel, M. R. Photoprotection by melanin. J. Photochem. Photobiol. B 9, 135–160 (1991).
Kennedy, C. et al. Melanocortin 1 receptor (MC1R) gene variants are associated with an increased risk for cutaneous melanoma which is largely independent of skin type and hair color. J. Invest. Dermatol. 117, 294–300 (2001).
Scott, M. C. et al. Human melanocortin 1 receptor variants, receptor function and melanocyte response to UV radiation. J. Cell Sci. 115, 2349–2355 (2002).
Hill, H. Z. & Hill, G. J. UVA, pheomelanin and the carcinogenesis of melanoma. Pigment Cell Res. 13 (Suppl. 8), 140–144 (2000).
Takeuchi, S. et al. Melanin acts as a potent UVB photosensitizer to cause an atypical mode of cell death in murine skin. Proc. Natl Acad. Sci. USA 101, 15076–15081 (2004).
Le Douarin, N. M., Creuzet, S., Couly, G. & Dupin, E. Neural crest cell plasticity and its limits. Development 131, 4637–4650 (2004).
Mackenzie, M. A., Jordan, S. A., Budd, P. S. & Jackson, I. J. Activation of the receptor tyrosine kinase Kit is required for the proliferation of melanoblasts in the mouse embryo. Dev. Biol. 192, 99–107 (1997).
Price, E. R. & Fisher, D. E. Sensorineural deafness and pigmentation genes: melanocytes and the Mitf transcriptional network. Neuron 30, 15–18 (2001).
Steingrimsson, E., Copeland, N. G. & Jenkins, N. A. Melanocytes and the microphthalmia transcription factor network. Annu. Rev. Genet. 38, 365–411 (2004).
Hershey, C. L. & Fisher, D. E. Mitf and Tfe3: members of a b-HLH-ZIP transcription factor family essential for osteoclast development and function. Bone 34, 689–696 (2004).
Chin, L., Garraway, L. A. & Fisher, D. E. Malignant melanoma: genetics and therapeutics in the genomic era. Genes Dev. 20, 2149–2182 (2006).
Christiansen, J. H., Coles, E. G. & Wilkinson, D. G. Molecular control of neural crest formation, migration and differentiation. Curr. Opin. Cell Biol. 12, 719–724 (2000).
Larue, L. & Delmas, V. The WNT/Beta-catenin pathway in melanoma. Front. Biosci. 11, 733–742 (2006).
Lee, H. O., Levorse, J. M. & Shin, M. K. The endothelin receptor-B is required for the migration of neural crest-derived melanocyte and enteric neuron precursors. Dev. Biol. 259, 162–175 (2003).
Wehrle-Haller, B. The role of Kit-ligand in melanocyte development and epidermal homeostasis. Pigment Cell Res. 16, 287–296 (2003).
Hemesath, T. J., Price, E. R., Takemoto, C., Badalian, T. & Fisher, D. E. MAP kinase links the transcription factor Microphthalmia to c-Kit signalling in melanocytes. Nature 391, 298–301 (1998).
Ruan, H. B., Zhang, N. & Gao, X. Identification of a novel point mutation of mouse proto-oncogene c-kit through N-ethyl-N-nitrosourea mutagenesis. Genetics 169, 819–831 (2005).
Spritz, R. A. & Beighton, P. Piebaldism with deafness: molecular evidence for an expanded syndrome. Am. J. Med. Genet. 75, 101–103 (1998).
Shears, D. et al. Molecular heterogeneity in two families with auditory pigmentary syndromes: the role of neuroimaging and genetic analysis in deafness. Clin. Genet. 65, 384–389 (2004).
Steel, K. P., Davidson, D. R. & Jackson, I. J. TRP-2/DT, a new early melanoblast marker, shows that steel growth factor (c-kit ligand) is a survival factor. Development 115, 1111–1119 (1992).
Jordan, S. A. & Jackson, I. J. MGF (KIT ligand) is a chemokinetic factor for melanoblast migration into hair follicles. Dev. Biol. 225, 424–436 (2000).
Legros, L., Cassuto, J. P. & Ortonne, J. P. Imatinib mesilate (Glivec): a systemic depigmenting agent for extensive vitiligo? Br. J. Dermatol. 153, 691–692 (2005).
Perez-Losada, J. et al. Zinc-finger transcription factor Slug contributes to the function of the stem cell factor c-kit signaling pathway. Blood 100, 1274–1286 (2002).
Sanchez-Martin, M. et al. Deletion of the SLUG (SNAI2) gene results in human piebaldism. Am. J. Med. Genet. A 122, 125–132 (2003).
Gupta, P. B. et al. The melanocyte differentiation program predisposes to metastasis after neoplastic transformation. Nature Genet. 37, 1047–1054 (2005).
Imokawa, G. Autocrine and paracrine regulation of melanocytes in human skin and in pigmentary disorders. Pigment Cell Res. 17, 96–110 (2004).
Otsuka, T. et al. c-Met autocrine activation induces development of malignant melanoma and acquisition of the metastatic phenotype. Cancer Res. 58, 5157–5167 (1998).
Noonan, F. P. et al. Neonatal sunburn and melanoma in mice. Nature 413, 271–272 (2001).
Nishimura, E. K., Yoshida, H., Kunisada, T. & Nishikawa, S. I. Regulation of E- and P-cadherin expression correlated with melanocyte migration and diversification. Dev. Biol. 215, 155–166 (1999).
Fitch, K. R. et al. Genetics of dark skin in mice. Genes Dev. 17, 214–228 (2003).
Van Raamsdonk, C. D., Fitch, K. R., Fuchs, H., de Angelis, M. H. & Barsh, G. S. Effects of G-protein mutations on skin color. Nature Genet. 36, 961–968 (2004).
Shin, M. K., Levorse, J. M., Ingram, R. S. & Tilghman, S. M. The temporal requirement for endothelin receptor-B signalling during neural crest development. Nature 402, 496–501 (1999).
Lerner, A. B. et al. A mouse model for vitiligo. J. Invest. Dermatol. 87, 299–304 (1986).
McGill, G. G. et al. Bcl2 regulation by the melanocyte master regulator Mitf modulates lineage survival and melanoma cell viability. Cell 109, 707–718 (2002).
Veis, D. J., Sorenson, C. M., Shutter, J. R. & Korsmeyer, S. J. Bcl-2-deficient mice demonstrate fulminant lymphoid apoptosis, polycystic kidneys, and hypopigmented hair. Cell 75, 229–240 (1993).
Du, J. et al. Critical role of CDK2 for melanoma growth linked to its melanocyte-specific transcriptional regulation by MITF. Cancer Cell 6, 565–576 (2004).
McGill, G. G., Haq, R., Nishimura, E. K. & Fisher, D. E. c-Met expression is regulated by Mitf in the melanocyte lineage. J. Biol. Chem. 281, 10365–10373 (2006).
Moore, K. A. & Lemischka, I. R. Stem cells and their niches. Science 311, 1880–1885 (2006).
Nishimura, E. K., Granter, S. R. & Fisher, D. E. Mechanisms of hair graying: incomplete melanocyte stem cell maintenance in the niche. Science 307, 720–724 (2005).
Mak, S. S., Moriyama, M., Nishioka, E., Osawa, M. & Nishikawa, S. Indispensable role of Bcl2 in the development of the melanocyte stem cell. Dev. Biol. 291, 144–153 (2006).
Paus, R. et al. A comprehensive guide for the recognition and classification of distinct stages of hair follicle morphogenesis. J. Invest. Dermatol. 113, 523–532 (1999).
Tsao, A. S., Kantarjian, H., Cortes, J., O'Brien, S. & Talpaz, M. Imatinib mesylate causes hypopigmentation in the skin. Cancer 98, 2483–2487 (2003).
Fukamachi, S., Shimada, A. & Shima, A. Mutations in the gene encoding B, a novel transporter protein, reduce melanin content in medaka. Nature Genet. 28, 381–385 (2001).
Olson, E. The rub on sunscreen. New York Times, 19 June 2006.
Wang, S. Q. et al. Ultraviolet A and melanoma: a review. J. Am. Acad. Dermatol. 44, 837–846 (2001).
Berwick, M. et al. Sun exposure and mortality from melanoma. J. Natl Cancer Inst. 97, 195–199 (2005).
Sollitto, R. B., Kraemer, K. H. & DiGiovanna, J. J. Normal vitamin D levels can be maintained despite rigorous photoprotection: six years' experience with xeroderma pigmentosum. J. Am. Acad. Dermatol. 37, 942–947 (1997).
Weinstock, M. A., Stampfer, M. J., Lew, R. A., Willett, W. C. & Sober, A. J. Case-control study of melanoma and dietary vitamin D: implications for advocacy of sun protection and sunscreen use. J. Invest. Dermatol. 98, 809–811 (1992).
Li, G., Tron, V. & Ho, V. Induction of squamous cell carcinoma in p53-deficient mice after ultraviolet irradiation. J. Invest. Dermatol. 110, 72–75 (1998).
Green, A. et al. Daily sunscreen application and betacarotene supplementation in prevention of basal-cell and squamous-cell carcinomas of the skin: a randomised controlled trial. Lancet 354, 723–729 (1999).
Vainio, H., Miller, A. B. & Bianchini, F. An international evaluation of the cancer-preventive potential of sunscreens. Int. J. Cancer 88, 838–842 (2000).
Gallagher, R. P. et al. Broad-spectrum sunscreen use and the development of new nevi in white children: a randomized controlled trial. J. Am. Med. Assoc. 283, 2955–2960 (2000).
Pfahlberg, A. et al. Monitoring of nevus density in children as a method to detect shifts in melanoma risk in the population. Prev. Med. 38, 382–387 (2004).
Dennis, L. K. et al. Constitutional factors and sun exposure in relation to nevi: a population-based cross-sectional study. Am. J. Epidemiol. 143, 248–256 (1996).
Mones, J. M. & Ackerman, A. B. Melanomas in prepubescent children: review comprehensively, critique historically, criteria diagnostically, and course biologically. Am. J. Dermatopathol. 25, 223–238 (2003).
Wolf, P., Quehenberger, F., Mullegger, R., Stranz, B. & Kerl, H. Phenotypic markers, sunlight-related factors and sunscreen use in patients with cutaneous melanoma: an Austrian case-control study. Melanoma Res. 8, 370–378 (1998).
Autier, P. et al. Melanoma and use of sunscreens: an EORTC case-control study in Germany, Belgium and France. The EORTC Melanoma Cooperative Group. Int. J. Cancer 61, 749–755 (1995).
Dennis, L. K., Beane Freeman, L. E. & VanBeek, M. J. Sunscreen use and the risk for melanoma: a quantitative review. Ann. Intern. Med. 139, 966–978 (2003).
Huncharek, M. & Kupelnick, B. Use of topical sunscreens and the risk of malignant melanoma: a meta-analysis of 9067 patients from 11 case-control studies. Am. J. Public Health 92, 1173–1177 (2002).
Palmer, J. S. et al. Melanocortin-1 receptor polymorphisms and risk of melanoma: is the association explained solely by pigmentation phenotype? Am. J. Hum. Genet. 66, 176–186 (2000).
Wenczl, E. et al. (Pheo)melanin photosensitizes UVA-induced DNA damage in cultured human melanocytes. J. Invest. Dermatol. 111, 678–682 (1998).
Acknowledgements
We thank S. R. Granter, M. E. Bigby, H. A. Haynes, A. B. Kimball, J. Rees, A. J. Sober, R. Stern and H. Tsao for useful comments and discussions. This work was supported by grants from the NIH and Doris Duke Charitable Foundation, and a Ruth L. Kirschstein National Research Service Award (J.Y.L.). D.E.F. is Distinguished Clinical Investigator of the Doris Duke Charitable Foundation and Jan and Charles Nirenberg Fellow in Pediatric Oncology at the Dana-Farber Cancer Institute.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
David Fisher discloses equity and consulting relationships with Magen Biosciences in Cambridge, Massachusetts.
Rights and permissions
About this article
Cite this article
Lin, J., Fisher, D. Melanocyte biology and skin pigmentation. Nature 445, 843–850 (2007). https://doi.org/10.1038/nature05660
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature05660
This article is cited by
-
Revealing mitf functions and visualizing allografted tumor metastasis in colorless and immunodeficient Xenopus tropicalis
Communications Biology (2024)
-
Deciphering the molecular mechanisms of stem cell dynamics in hair follicle regeneration
Experimental & Molecular Medicine (2024)
-
Skin cancer - what general dental practitioners should look for
British Dental Journal (2024)
-
Fluorometric Assay of Tyrosinase and Atrazine Based on the Use of Carbon Dots and the Inhibition of Tyrosinase Activity
Journal of Fluorescence (2024)
-
Bioengineered skin organoids: from development to applications
Military Medical Research (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.