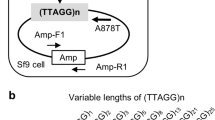
Abstract
Long interspersed element-1 (LINE-1 or L1) elements are abundant, non-long-terminal-repeat (non-LTR) retrotransposons that comprise ∼17% of human DNA1. The average human genome contains ∼80–100 retrotransposition-competent L1s (ref. 2), and they mobilize by a process that uses both the L1 endonuclease and reverse transcriptase, termed target-site primed reverse transcription3,4,5. We have previously reported an efficient, endonuclease-independent L1 retrotransposition pathway (ENi) in certain Chinese hamster ovary (CHO) cell lines that are defective in the non-homologous end-joining (NHEJ) pathway of DNA double-strand-break repair6. Here we have characterized ENi retrotransposition events generated in V3 CHO cells, which are deficient in DNA-dependent protein kinase catalytic subunit (DNA-PKcs) activity and have both dysfunctional telomeres and an NHEJ defect. Notably, ∼30% of ENi retrotransposition events insert in an orientation-specific manner adjacent to a perfect telomere repeat (5′-TTAGGG-3′). Similar insertions were not detected among ENi retrotransposition events generated in controls or in XR-1 CHO cells deficient for XRCC4, an NHEJ factor that is required for DNA ligation but has no known function in telomere maintenance. Furthermore, transient expression of a dominant-negative allele of human TRF2 (also called TERF2) in XRCC4-deficient XR-1 cells, which disrupts telomere capping, enables telomere-associated ENi retrotransposition events. These data indicate that L1s containing a disabled endonuclease can use dysfunctional telomeres as an integration substrate. The findings highlight similarities between the mechanism of ENi retrotransposition and the action of telomerase, because both processes can use a 3′ OH for priming reverse transcription at either internal DNA lesions or chromosome ends7,8. Thus, we propose that ENi retrotransposition is an ancestral mechanism of RNA-mediated DNA repair associated with non-LTR retrotransposons that may have been used before the acquisition of an endonuclease domain.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
International Human Genome Sequencing Consortium. Initial sequencing and analysis of the human genome. Nature 409, 860–921 (2001)
Brouha, B. et al. Hot L1s account for the bulk of retrotransposition in the human population. Proc. Natl Acad. Sci. USA 100, 5280–5285 (2003)
Luan, D. D., Korman, M. H., Jakubczak, J. L. & Eickbush, T. H. Reverse transcription of R2Bm RNA is primed by a nick at the chromosomal target site: a mechanism for non-LTR retrotransposition. Cell 72, 595–605 (1993)
Moran, J. V. et al. High frequency retrotransposition in cultured mammalian cells. Cell 87, 917–927 (1996)
Feng, Q., Moran, J. V., Kazazian, H. H. & Boeke, J. D. Human L1 retrotransposon encodes a conserved endonuclease required for retrotransposition. Cell 87, 905–916 (1996)
Morrish, T. A. et al. DNA repair mediated by endonuclease-independent LINE-1 retrotransposition. Nature Genet. 31, 159–165 (2002)
Pardue, M. L. & DeBaryshe, P. G. Retrotransposons provide an evolutionarily robust non-telomerase mechanism to maintain telomeres. Annu. Rev. Genet. 37, 485–511 (2003)
Lingner, J. et al. Reverse transcriptase motifs in the catalytic subunit of telomerase. Science 276, 561–567 (1997)
Goytisolo, F. A., Samper, E., Edmonson, S., Taccioli, G. E. & Blasco, M. A. The absence of the DNA-dependent protein kinase catalytic subunit in mice results in anaphase bridges and in increased telomeric fusions with normal telomere length and G-strand overhang. Mol. Cell. Biol. 21, 3642–3651 (2001)
Gilley, D. et al. DNA-PKcs is critical for telomere capping. Proc. Natl Acad. Sci. USA 98, 15084–15088 (2001)
Bailey, S. M. et al. DNA double-strand break repair proteins are required to cap the ends of mammalian chromosomes. Proc. Natl Acad. Sci. USA 96, 14899–14904 (1999)
Bailey, S. M., Cornforth, M. N., Kurimasa, A., Chen, D. J. & Goodwin, E. H. Strand-specific postreplicative processing of mammalian telomeres. Science 293, 2462–2465 (2001)
Takai, H., Smogorzewska, A. & de Lange, T. DNA damage foci at dysfunctional telomeres. Curr. Biol. 13, 1549–1556 (2003)
Hao, L. Y., Strong, M. A. & Greider, C. W. Phosphorylation of H2AX at short telomeres in T cells and fibroblasts. J. Biol. Chem. 279, 45148–45154 (2004)
de Lange, T. Shelterin: the protein complex that shapes and safeguards human telomeres. Genes Dev. 19, 2100–2110 (2005)
Gilbert, N., Lutz-Prigge, S. & Moran, J. V. Genomic deletions created upon LINE-1 retrotransposition. Cell 110, 315–325 (2002)
Gilbert, N., Lutz, S., Morrish, T. A. & Moran, J. V. Multiple fates of l1 retrotransposition intermediates in cultured human cells. Mol. Cell. Biol. 25, 7780–7795 (2005)
Faravelli, M. et al. Molecular organization of internal telomeric sequences in Chinese hamster chromosomes. Gene 283, 11–16 (2002)
Taccioli, G. E. et al. Targeted disruption of the catalytic subunit of the DNA-PK gene in mice confers severe combined immunodeficiency and radiosensitivity. Immunity 9, 355–366 (1998)
Blunt, T. et al. Identification of a nonsense mutation in the carboxyl-terminal region of DNA-dependent protein kinase catalytic subunit in the scid mouse. Proc. Natl Acad. Sci. USA 93, 10285–10290 (1996)
Whitmore, G. F., Varghese, A. J. & Gulyas, S. Cell cycle responses of two X-ray sensitive mutants defective in DNA repair. Int. J. Radiat. Biol. 56, 657–665 (1989)
Blunt, T. et al. Defective DNA-dependent protein kinase activity is linked to V(D)J recombination and DNA repair defects associated with the murine scid mutation. Cell 80, 813–823 (1995)
Meek, K., Gupta, S., Ramsden, D. A. & Lees-Miller, S. P. The DNA-dependent protein kinase: the director at the end. Immunol. Rev. 200, 132–141 (2004)
Beamish, H. J. et al. The C-terminal conserved domain of DNA-PKcs, missing in the SCID mouse, is required for kinase activity. Nucleic Acids Res. 28, 1506–1513 (2000)
van Steensel, B., Smogorzewska, A. & de Lange, T. TRF2 protects human telomeres from end-to-end fusions. Cell 92, 401–413 (1998)
Smogorzewska, A., Karlseder, J., Holtgreve-Grez, H., Jauch, A. & de Lange, T. DNA ligase IV-dependent NHEJ of deprotected mammalian telomeres in G1 and G2. Curr. Biol. 12, 1635–1644 (2002)
Celli, G. B. & de Lange, T. DNA processing is not required for ATM-mediated telomere damage response after TRF2 deletion. Nature Cell Biol. 7, 712–718 (2005)
Kulpa, D. A. & Moran, J. V. Cis-preferential LINE-1 reverse transcriptase activity in ribonucleoprotein particles. Nature Struct. Mol. Biol. 13, 655–660 (2006)
Wei, W., Morrish, T. A., Alisch, R. S. & Moran, J. V. A transient assay reveals that cultured human cells can accommodate multiple LINE-1 retrotransposition events. Anal. Biochem. 284, 435–438 (2000)
Dombroski, B. A., Mathias, S. L., Nanthakumar, E., Scott, A. F. & Kazazian, H. H. Isolation of an active human transposable element. Science 254, 1805–1808 (1991)
Acknowledgements
We thank A. M. DesLauriers for help with flow cytometry and cell sorting; R. Lyons for help with sequencing; T. de Lange for discussions and the dominant-negative TRF2 expression construct; M. Abe for the murine DNA-PKcs expression construct; G. Hammer and T. Else for help with the metaphase analysis; and C. Greider for discussions and use of equipment and reagents for immunocytochemistry experiments. We thank T. Else, T. Glover, T. Glaser, N. Howlett, T. Wilson and current members of the Moran laboratory for discussions during the course of this work. T.A.M. was supported in part by a NIH training grant and is now a Leukemia and Lymphoma Society Fellow. J.L.G.-P. was supported in part by a MEC/Fulbright postdoctoral grant. G.E.T. is partially supported by a grant from the Human Frontier Science Program. T.D.S was supported in part by a grant from the National Institutes of Health. J.S. was supported in part by a grant from the Pew Foundation. J.V.M was supported in part by grants from the W. M. Keck Foundation and the National Institutes of Health.
Author Contributions T.A.M is co-first author and is a corresponding author. She contributed to the original concept, designed and performed experiments, analysed the data, and wrote the first draft of the manuscript. J.L.G.-P. is co-first author. He contributed to the concept, designed and performed experiments, analysed the data, and helped write and revise the manuscript. T.D.S and G.E.T. contributed reagents, helped with data interpretation and provided helpful comments during the course of this study. J.S. contributed the murine DNA-PKcs complemented cell lines, performed functional analyses on those cell lines, helped with data analysis, and helped revise the manuscript. J.V.M. is the senior author. He contributed to the original concept, analysed the data, revised the manuscript and provided financial support.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
Reprints and permissions information is available at www.nature.com/reprints. The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-4 with Legends, Supplementary Methods, Supplementary Tables 1-3 and additional references (PDF 1675 kb)
Rights and permissions
About this article
Cite this article
Morrish, T., Garcia-Perez, J., Stamato, T. et al. Endonuclease-independent LINE-1 retrotransposition at mammalian telomeres. Nature 446, 208–212 (2007). https://doi.org/10.1038/nature05560
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature05560
This article is cited by
-
Telomeric retrotransposons show propensity to form G-quadruplexes in various eukaryotic species
Mobile DNA (2023)
-
Young LINE-1 transposon 5′ UTRs marked by elongation factor ELL3 function as enhancers to regulate naïve pluripotency in embryonic stem cells
Nature Cell Biology (2023)
-
Fanconi anemia DNA crosslink repair factors protect against LINE-1 retrotransposition during mouse development
Nature Structural & Molecular Biology (2023)
-
From parasites to partners: exploring the intricacies of host-transposon dynamics and coevolution
Functional & Integrative Genomics (2023)
-
A pilot study of LINE-1 copy number and telomere length with aging in human sperm
Journal of Assisted Reproduction and Genetics (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.