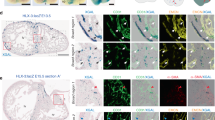
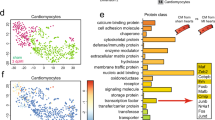
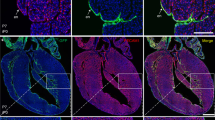
Abstract
Cardiac failure has a principal underlying aetiology of ischaemic damage arising from vascular insufficiency. Molecules that regulate collateral growth in the ischaemic heart also regulate coronary vasculature formation during embryogenesis. Here we identify thymosin β4 (Tβ4) as essential for all aspects of coronary vessel development in mice, and demonstrate that Tβ4 stimulates significant outgrowth from quiescent adult epicardial explants, restoring pluripotency and triggering differentiation of fibroblasts, smooth muscle cells and endothelial cells. Tβ4 knockdown in the heart is accompanied by significant reduction in the pro-angiogenic cleavage product N-acetyl-seryl-aspartyl-lysyl-proline (AcSDKP). Although injection of AcSDKP was unable to rescue Tβ4 mutant hearts, it significantly enhanced endothelial cell differentiation from adult epicardially derived precursor cells. This study identifies Tβ4 and AcSDKP as potent stimulators of coronary vasculogenesis and angiogenesis, and reveals Tβ4-induced adult epicardial cells as a viable source of vascular progenitors for continued renewal of regressed vessels at low basal level or sustained neovascularization following cardiac injury.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Smart, N., Hill, A. A., Cross, J. C. & Riley, P. R. A differential screen for putative targets of the bHLH transcription factor Hand1 in cardiac morphogenesis. Mech. Dev. 119, S65–S71 (2002)
Bock-Marquette, I., Saxena, A., White, M. D., Dimaio, J. M. & Srivastava, D. Thymosin β4 activates integrin-linked kinase and promotes cardiac cell migration, survival and cardiac repair. Nature 432, 466–472 (2004)
Moses, K., DeMayo, F., Braun, R., Reecy, J. & Schwartz, R. Embryonic expression of an Nkx2–5/Cre gene using ROSA26 reporter mice. Genesis 31, 176–180 (2001)
Zeisberg, E. M. et al. Morphogenesis of the right ventricle requires myocardial expression of Gata4. J. Clin. Invest. 115, 1522–1531 (2005)
Chen, J. et al. Selective requirement of myosin light chain 2v in embryonic heart function. J. Biol. Chem. 273, 1252–1256 (1998)
von Kodolitsch, Y. et al. Coronary artery anomalies Part I: Recent insights from molecular embryology. Z. Kardiol. 93, 929–937 (2004)
Ward, N. L. & Dumont, D. J. The angiopoietins and Tie2/Tek: adding to the complexity of cardiovascular development. Semin. Cell Dev. Biol. 13, 19–27 (2002)
Merki, E. et al. Epicardial retinoid X receptor α is required for myocardial growth and coronary artery formation. Proc. Natl Acad. Sci. USA 102, 18455–18460 (2005)
Yamashita, J. et al. Flk1-positive cells derived from embryonic stem cells serve as vascular progenitors. Nature 408, 92–96 (2000)
Carmeliet, P. Angiogenesis in health and disease. Nature Med. 9, 653–660 (2003)
Tevosian, S. G. et al. FOG-2, a cofactor for GATA transcription factors, is essential for heart morphogenesis and development of coronary vessels from epicardium. Cell 101, 729–739 (2000)
Giordano, F. J. et al. A cardiac myocyte vascular endothelial growth factor paracrine pathway is required to maintain cardiac function. Proc. Natl Acad. Sci. USA 98, 5780–5785 (2001)
Chen, T. H. et al. Epicardial induction of fetal cardiomyocyte proliferation via a retinoic acid-inducible trophic factor. Dev. Biol. 250, 198–207 (2002)
Morabito, C. J., Dettman, R. W., Kattan, J., Collier, J. M. & Bristow, J. Positive and negative regulation of epicardial-mesenchymal transformation during avian heart development. Dev. Biol. 234, 204–215 (2001)
Poelmann, R. E., Lie-Venema, H. & Gittenberger-de Groot, A. C. The role of the epicardium and neural crest as extracardiac contributors to coronary vascular development. Tex. Heart Inst. J. 29, 255–261 (2002)
Luttun, A. & Carmeliet, P. De novo vasculogenesis in the heart. Cardiovasc. Res. 58, 378–389 (2003)
Kamihata, H. et al. Implantation of bone marrow mononuclear cells into ischemic myocardium enhances collateral perfusion and regional function via side supply of angioblasts, angiogenic ligands, and cytokines. Circulation 104, 1046–1052 (2001)
Saiura, A., Sata, M., Hirata, Y., Nagai, R. & Makuuchi, M. Circulating smooth muscle progenitor cells contribute to atherosclerosis. Nature Med. 7, 382–383 (2001)
Grillon, C. et al. Involvement of thymosin β4 and endoproteinase Asp-N in the biosynthesis of the tetrapeptide AcSerAspLysPro a regulator of the hematopoietic system. FEBS Lett. 274, 30–34 (1990)
Rieger, K. J. et al. Involvement of human plasma angiotensin I-converting enzyme in the degradation of the haemoregulatory peptide N-acetyl-seryl-aspartyl-lysyl-proline. Biochem. J. 296, 373–378 (1993)
Goldstein, A. L., Hannappel, E. & Kleinman, H. K. Thymosin β4: actin-sequestering protein moonlights to repair injured tissues. Trends Mol. Med. 11, 421–429 (2005)
Wang, D. et al. N-acetyl-seryl-aspartyl-lysyl-proline stimulates angiogenesis in vitro and in vivo. Am. J. Physiol. Heart Circ. Physiol. 287, H2099–H2105 (2004)
Rasoul, S. et al. Antifibrotic effect of Ac-SDKP and angiotensin-converting enzyme inhibition in hypertension. J. Hypertens. 22, 593–603 (2004)
Pokharel, S. et al. Increased myocardial collagen content in transgenic rats overexpressing cardiac angiotensin-converting enzyme is related to enhanced breakdown of N-acetyl-ser-asp-lys-pro and increased phosphorylation of Smad2/3. Circulation 110, 3129–3135 (2004)
Peng, H. et al. Angiotensin-converting enzyme inhibitors: A new mechanism of action. Circulation 112, 2436–2445 (2005)
Tomanek, R., Zheng, W. & Yue, X. Growth factor activation in myocardial vascularization: Therapeutic implications. Mol. Cell. Biochem. 264, 3–11 (2004)
Kunath, T. et al. Transgenic RNA interference in ES cell-derived embryos recapitulates a genetic null phenotype. Nature Biotechnol. 21, 559–561 (2003)
Acknowledgements
This research was supported by the British Heart Foundation and the Medical Research Council. We acknowledge the collaboration of K. Thiam and S. Millet of genOway (France) in generating the Tβ4shRNAflox line and G. Gish for providing RasGAPshRNA plasmid. We thank J. Clark for providing adult myocardial infarction heart samples and S. Bhattacharya for comments. Author Contributions N.S. carried out the majority of experimental work and data analyses with contributions from C.A.R., A.A.D.M. and P.R.R. K.M. and R.J.S. provided the Nkx2.5Cre mouse strain and K.R.C. provided the MLC2vCre mouse strain. P.R.R. devised and planned the project and the manuscript was written by P.R.R. and N.S.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at www.nature.com/reprints. The authors declare no competing financial interests.
Supplementary information
Supplementary Notes
This file contains Supplementary Methods detailing all the protocols used in the paper and Supplementary Figure Legends. (PDF 220 kb)
Supplementary Figures
Supplementary Figures 1–9. (PDF 1070 kb)
Supplementary Table 1
A table summarising the penetrance and severity of the Tβ4 knockdown embryo phenotype. (PPT 29 kb)
Rights and permissions
About this article
Cite this article
Smart, N., Risebro, C., Melville, A. et al. Thymosin β4 induces adult epicardial progenitor mobilization and neovascularization. Nature 445, 177–182 (2007). https://doi.org/10.1038/nature05383
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature05383
This article is cited by
-
The role of cardiac pericytes in health and disease: therapeutic targets for myocardial infarction
Nature Reviews Cardiology (2024)
-
Regeneration of the heart: from molecular mechanisms to clinical therapeutics
Military Medical Research (2023)
-
Thymosin beta-4 improves endothelial function and reparative potency of diabetic endothelial cells differentiated from patient induced pluripotent stem cells
Stem Cell Research & Therapy (2022)
-
Hypoxia promotes a perinatal-like progenitor state in the adult murine epicardium
Scientific Reports (2022)
-
Epicardial slices: an innovative 3D organotypic model to study epicardial cell physiology and activation
npj Regenerative Medicine (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.