Abstract
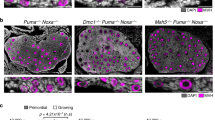
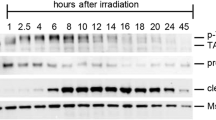
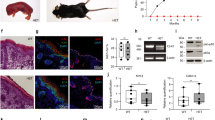
Meiosis in the female germ line of mammals is distinguished by a prolonged arrest in prophase of meiosis I between homologous chromosome recombination and ovulation1. How DNA damage is detected in these arrested oocytes is poorly understood, but it is variably thought to involve p53, a central tumour suppressor in mammals2,3,4. While the function of p53 in monitoring the genome of somatic cells is clear, a consensus for the importance of p53 for germ line integrity has yet to emerge. Here we show that the p53 homologue p63 (refs 5, 6), and specifically the TAp63 isoform, is constitutively expressed in female germ cells during meiotic arrest and is essential in a process of DNA damage-induced oocyte death not involving p53. We also show that DNA damage induces both the phosphorylation of p63 and its binding to p53 cognate DNA sites and that these events are linked to oocyte death. Our data support a model whereby p63 is the primordial member of the p53 family and acts in a conserved process of monitoring the integrity of the female germ line, whereas the functions of p53 are restricted to vertebrate somatic cells for tumour suppression. These findings have implications for understanding female germ line fidelity, the regulation of fertility and the evolution of tumour suppressor mechanisms.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Eppig, J. J. Coordination of nuclear and cytoplasmic oocyte maturation in Eutherian mammals. Reprod. Fertil. Dev. 8, 485–489 (1996)
Hainaut, P. & Wiman, K. 25 Years of p53 Research (Springer, Berlin, 2005)
Lane, D. P. et al. On the regulation of the p53 tumour suppressor, and its role in the cellular response to DNA damage. Phil. Trans. R. Soc. Lond. B 347, 83–87 (1995)
Harris, S. L. & Levine, A. J. The p53 pathway: positive and negative feedback loops. Oncogene 24, 2899–2908 (2005)
Yang, A. et al. p63, a p53 homolog at 3q27–29, encodes multiple products with transactivating, death-inducing, and dominant-negative activities. Mol. Cell 2, 305–316 (1998)
Yang, A., Kaghad, M., Caput, D. & McKeon, F. On the shoulders of giants: p63, p73 and the rise of p53. Trends Genet. 18, 90–95 (2002)
Yang, A. et al. Relationships between p63 binding, DNA sequence, transcription activity, and biological function in human cells. Mol. Cell (in the press)
Yang, A. et al. p63 is essential for regenerative proliferation in limb, craniofacial and epithelial development. Nature 398, 714–718 (1999)
Yang, A. et al. p73-deficient mice have neurological, pheromonal and inflammatory defects but lack spontaneous tumours. Nature 404, 99–103 (2000)
Di Iorio, E. et al. Isoforms of ΔNp63 and the migration of ocular limbal cells in human corneal regeneration. Proc. Natl Acad. Sci. USA 102, 9523–9528 (2005)
Peters, H. Migration of gonocytes into the mammalian gonad and their differentiation. Phil. Trans. R. Soc. Lond. B 259, 91–101 (1970)
Yu, J., Hecht, N. B. & Schultz, R. M. Expression of MSY2 in mouse oocytes and preimplantation embryos. Biol. Reprod. 65, 1260–1270 (2001)
Peters, H. & Levy, E. Effect of irradiation in infancy on the mouse ovary; a quantitative study of oocyte sensitivity. J. Reprod. Fertil. 7, 37–45 (1964)
Budihardjo, I., Oliver, H., Lutter, M., Luo, X. & Wang, X. Biochemical pathways of caspase activation during apoptosis. Annu. Rev. Cell Dev. Biol. 15, 269–290 (1999)
Mahadevaiah, S. K. et al. Recombinational DNA double-strand breaks in mice precede synapsis. Nature Genet. 27, 271–276 (2001)
Ma, L. et al. A plausible model for the digital response of p53 to DNA damage. Proc. Natl Acad. Sci. USA 102, 14266–14271 (2005)
Serber, Z. et al. A C-terminal inhibitory domain controls the activity of p63 by an intramolecular mechanism. Mol. Cell. Biol. 22, 8601–8611 (2002)
Derry, W. B., Putzke, A. P. & Rothman, J. H. Caenorhabditis elegans p53: role in apoptosis, meiosis, and stress resistance. Science 294, 591–595 (2001)
Schumacher, B., Hofmann, K., Boulton, S. & Gartner, A. The C. elegans homolog of the p53 tumor suppressor is required for DNA damage-induced apoptosis. Curr. Biol. 11, 1722–1727 (2001)
Ollmann, M. et al. Drosophila p53 is a structural and functional homolog of the tumor suppressor p53. Cell 101, 91–101 (2000)
Brodsky, M. H. et al. Drosophila p53 binds a damage response element at the reaper locus. Cell 101, 103–113 (2000)
Jin, S. et al. Identification and characterization of a p53 homologue in Drosophila melanogaster. Proc. Natl Acad. Sci. USA 97, 7301–7306 (2000)
Sogame, N., Kim, M. & Abrams, J. M. Drosophila p53 preserves genomic stability by regulating cell death. Proc. Natl Acad. Sci. USA 100, 4696–4701 (2003)
Brodsky, M. H. et al. Drosophila melanogaster MNK/Chk2 and p53 regulate multiple DNA repair and apoptotic pathways following DNA damage. Mol. Cell. Biol. 24, 1219–1231 (2004)
Schumacher, B. et al. Translational repression of C. elegans p53 by GLD-1 regulates DNA damage-induced apoptosis. Cell 120, 357–368 (2005)
Hunt, P. A. & Hassold, T. J. Sex matters in meiosis. Science 296, 2181–2183 (2002)
Haldane, J. B. S. The rate of mutation of the gene for hemophilia and its segregation in males and females. Ann. Eugen. 13, 262–271 (1947)
Ko, L. J. & Prives, C. p53: puzzle and paradigm. Genes Dev. 10, 1054–1072 (1996)
Bourdon, J. C. et al. p53 isoforms can regulate p53 transcriptional activity. Genes Dev. 19, 2122–2137 (2005)
el-Deiry, W. S., Kern, S. E., Pietenpol, J. A., Kinzler, K. W. & Vogelstein, B. Definition of a consensus binding site for p53. Nature Genet. 1, 45–49 (1992)
Acknowledgements
We thank R. Schultz, B. Spyropoulos, P. Moens and M. De Luca for gifts of antibodies; A. Lee, G. Perez, C. Morton and Y. Lee for advice and assistance with technical procedures; and V. Doetsch, M. Senoo, F. Pinto, H. Green, A. Sharpe and M. Colaiacova for discussions. A.Y. was supported by a fellowship from the Department of Defence Program in Breast Cancer. This work was supported by grants from the NCI and the NIH to C.P.C. and F.M. Author Contributions E.K.S. established the ovary experimental system, performed the DNA damage experiments with the wild-type, p53-null, and p63-null mice in vivo and in vitro, identified the TAp63 phosphorylation-dependent mobility shift and performed the kinetics linking it to oocyte death. A.Y. generated the anti-TAp63 monoclonal antibodies, designed the TAp63 targeting and genotyping strategies and assisted in the generation of the TAp63 knockout mice. A.K. generated the TAp63 knockout mice and performed the DNA damage experiments on these strains. C.B. performed the TAp63 DNA binding experiments and statistical analyses of data presented in this paper. A.M. generated and characterized the anti-TAp63 antibodies with A.Y. Z.Z. and A.K. performed the bioinformatics analysis of the p53 gene family. J.E. made early contributions on TAp63 expression in human oocytes. R.B. supervised the histopathology analysis. C.C. and F.M. directed the project, and F.M. wrote the manuscript with help from A.Y., A.K. and E.K.S.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at www.nature.com/reprints. The authors declare no competing financial interests.
Supplementary information
Supplementary Notes
This file contains Supplementary Figures 1–9, Supplementary Methods and additional references. (PDF 1669 kb)
Rights and permissions
About this article
Cite this article
Suh, EK., Yang, A., Kettenbach, A. et al. p63 protects the female germ line during meiotic arrest. Nature 444, 624–628 (2006). https://doi.org/10.1038/nature05337
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature05337
This article is cited by
-
Distinct characteristics of the DNA damage response in mammalian oocytes
Experimental & Molecular Medicine (2024)
-
The Dominant Mechanism of Cyclophosphamide-Induced Damage to Ovarian Reserve: Premature Activation or Apoptosis of Primordial Follicles?
Reproductive Sciences (2024)
-
Tamoxifen decreases ovarian toxicity without compromising cancer treatment in a rat model of mammary cancer
BMC Genomics (2023)
-
Mechanisms of ovarian aging in women: a review
Journal of Ovarian Research (2023)
-
Menstrual blood-derived endometrial stem cell, a unique and promising alternative in the stem cell-based therapy for chemotherapy-induced premature ovarian insufficiency
Stem Cell Research & Therapy (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.