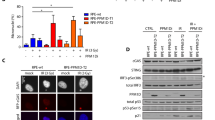
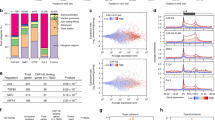
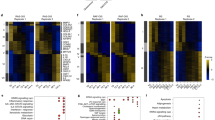
Abstract
Early tumorigenesis is associated with the engagement of the DNA-damage checkpoint response (DDR)1,2. Cell proliferation and transformation induced by oncogene activation are restrained by cellular senescence3,4,5,6. It is unclear whether DDR activation and oncogene-induced senescence (OIS) are causally linked. Here we show that senescence, triggered by the expression of an activated oncogene (H-RasV12) in normal human cells, is a consequence of the activation of a robust DDR. Experimental inactivation of DDR abrogates OIS and promotes cell transformation. DDR and OIS are established after a hyper-replicative phase occurring immediately after oncogene expression. Senescent cells arrest with partly replicated DNA and with DNA replication origins having fired multiple times. In vivo DNA labelling and molecular DNA combing reveal that oncogene activation leads to augmented numbers of active replicons and to alterations in DNA replication fork progression. We also show that oncogene expression does not trigger a DDR in the absence of DNA replication. Last, we show that oncogene activation is associated with DDR activation in a mouse model in vivo. We propose that OIS results from the enforcement of a DDR triggered by oncogene-induced DNA hyper-replication.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Bartkova, J. et al. DNA damage response as a candidate anti-cancer barrier in early human tumorigenesis. Nature 434, 864–870 (2005)
Gorgoulis, V. G. et al. Activation of the DNA damage checkpoint and genomic instability in human precancerous lesions. Nature 434, 907–913 (2005)
Michaloglou, C. et al. BRAFE600-associated senescence-like cell cycle arrest of human naevi. Nature 436, 720–724 (2005)
Chen, Z. et al. Crucial role of p53-dependent cellular senescence in suppression of Pten-deficient tumorigenesis. Nature 436, 725–730 (2005)
Braig, M. et al. Oncogene-induced senescence as an initial barrier in lymphoma development. Nature 436, 660–665 (2005)
Collado, M. et al. Tumour biology: senescence in premalignant tumours. Nature 436, 642 (2005)
Mathon, N. F. & Lloyd, A. C. Cell senescence and cancer. Nature Rev. Cancer 1, 203–213 (2001)
Serrano, M., Lin, A. W., McCurrach, M. E., Beach, D. & Lowe, S. W. Oncogenic ras provokes premature cell senescence associated with accumulation of p53 and p16INK4a. Cell 88, 593–602 (1997)
Kastan, M. B. & Bartek, J. Cell-cycle checkpoints and cancer. Nature 432, 316–323 (2004)
Narita, M. et al. Rb-mediated heterochromatin formation and silencing of E2F target genes during cellular senescence. Cell 113, 703–716 (2003)
Seger, Y. R. et al. Transformation of normal human cells in the absence of telomerase activation. Cancer Cell 2, 401–413 (2002)
Vaziri, C. et al. A p53-dependent checkpoint pathway prevents rereplication. Mol. Cell 11, 997–1008 (2003)
Melixetian, M. et al. Loss of Geminin induces rereplication in the presence of functional p53. J. Cell Biol. 165, 473–482 (2004)
Lebofsky, R. & Bensimon, A. Single DNA molecule analysis: applications of molecular combing. Brief. Funct. Genomic. Proteomic. 1, 385–396 (2003)
Takeda, D. Y., Shibata, Y., Parvin, J. D. & Dutta, A. Recruitment of ORC or CDC6 to DNA is sufficient to create an artificial origin of replication in mammalian cells. Genes Dev. 19, 2827–2836 (2005)
Duursma, A. & Agami, R. p53-dependent regulation of Cdc6 protein stability controls cellular proliferation. Mol. Cell. Biol. 25, 6937–6947 (2005)
Casper, A. M., Nghiem, P., Arlt, M. F. & Glover, T. W. ATR regulates fragile site stability. Cell 111, 779–789 (2002)
Quintanilla, M., Brown, K., Ramsden, M. & Balmain, A. Carcinogen-specific mutation and amplification of Ha-ras during mouse skin carcinogenesis. Nature 322, 78–80 (1986)
d’Adda di Fagagna, F. et al. A DNA damage checkpoint response in telomere-initiated senescence. Nature 426, 194–198 (2003)
Herbig, U., Jobling, W. A., Chen, B. P., Chen, D. J. & Sedivy, J. M. Telomere shortening triggers senescence of human cells through a pathway involving ATM, p53, and p21CIP1, but not p16INK4a. Mol. Cell 14, 501–513 (2004)
Denko, N. C., Giaccia, A. J., Stringer, J. R. & Stambrook, P. J. The human Ha-ras oncogene induces genomic instability in murine fibroblasts within one cell cycle. Proc. Natl Acad. Sci. USA 91, 5124–5128 (1994)
Williams, G. H. et al. Improved cervical smear assessment using antibodies against proteins that regulate DNA replication. Proc. Natl Acad. Sci. USA 95, 14932–14937 (1998)
Karakaidos, P. et al. Overexpression of the replication licensing regulators hCdt1 and hCdc6 characterizes a subset of non-small-cell lung carcinomas: Synergistic effect with mutant p53 on tumor growth and chromosomal instability—evidence of E2F-1 transcriptional control over hCdt1. Am. J. Pathol. 165, 1351–1365 (2004)
Bartkova, J. et al. Oncogene-induced senescence is part of the tumorigenesis barrier imposed by DNA damage checkpoints. Nature doi: 10.1038/nature05268 (this issue).
Gonzalez, S. et al. Oncogenic activity of Cdc6 through repression of the INK4/ARF locus. Nature 440, 702–706 (2006)
Hirao, A. et al. Chk2 is a tumor suppressor that regulates apoptosis in both an ataxia telangiectasia mutated (ATM)-dependent and an ATM-independent manner. Mol. Cell. Biol. 22, 6521–6532 (2002)
Vafa, O. et al. c-Myc can induce DNA damage, increase reactive oxygen species, and mitigate p53 function: a mechanism for oncogene-induced genetic instability. Mol. Cell 9, 1031–1044 (2002)
Puc, J. et al. Lack of PTEN sequesters CHK1 and initiates genetic instability. Cancer Cell 7, 193–204 (2005)
Dimri, G. P. et al. A biomarker that identifies senescent human cells in culture and in aging skin in vivo.. Proc. Natl Acad. Sci. USA 92, 9363–9367 (1995)
Mirzoeva, O. K. & Petrini, J. H. J. DNA damage-dependent nuclear dynamics of the Mre11 complex. Mol. Cell. Biol. 21, 281–288 (2001)
Acknowledgements
We thank P. Transidico for confocal analysis; IFOM Cell Biology, DNA Sequencing, Imaging and Microarrays Units for support; J. Campisi, T. D. Halazonetis, K. Helin, S. P. Jackson, Y. Shiloh, T. Mak, C. Marine, S. Pece and A. Tocchetti for sharing reagents; and M. Alcalay, B. Amati, A. Ballabeni, M. Barbacid, M. Foiani and H. Muller for discussions. F.d.A.d.F., R.M. and P.G.P. are supported by AIRC (Associazione Italiana per la Ricerca sul Cancro). R.D.M. and M.F. are SEMM (European School of Molecular Medicine) students. Author Contributions M.F. generated Fig. 4f and Supplementary Figs 4c, 7 and 17; A.C. generated Supplementary Figs S1a, b and 4a, b and helped with initial retroviral transduction experiments; S.P. and R.M. generated and analysed data in Fig. 3 and Supplementary Fig. 11; P.G. generated Fig. 4b and Supplementary Fig. 14; C.L. and P.G.N. generated and analysed data in Fig. 4g; C.S. and A.B. generated and analysed data in Fig. 4c and Supplementary Fig. 15; M.G. helped with confocal analysis; P.G.P. discussed the results; R.D.M. generated all remaining figures and contributed to experimental design; F.d.A.d.F. generated Fig. 4e, planned the project and wrote the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at www.nature.com/reprints. The authors declare no competing financial interests.
Supplementary information
Supplementary Notes
This file contains Supplementary Figure Legends, Supplementary Methods and additional references. (RTF 36 kb)
Supplementary Figures
This file contains Supplementary Figures 1–18. (PDF 1419 kb)
Supplementary Data
This file contains information on the genes and proteins used in this study. (DOC 22 kb)
Rights and permissions
About this article
Cite this article
Di Micco, R., Fumagalli, M., Cicalese, A. et al. Oncogene-induced senescence is a DNA damage response triggered by DNA hyper-replication. Nature 444, 638–642 (2006). https://doi.org/10.1038/nature05327
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature05327
This article is cited by
-
Role of cellular senescence in inflammation and regeneration
Inflammation and Regeneration (2024)
-
A GATA2-CDC6 axis modulates androgen receptor blockade-induced senescence in prostate cancer
Journal of Experimental & Clinical Cancer Research (2023)
-
DNA replication stress and mitotic catastrophe mediate sotorasib addiction in KRASG12C-mutant cancer
Journal of Biomedical Science (2023)
-
Suppression of tumor growth and apoptosis induction by pomegranate seed nano-emulsion in mice bearing solid Ehrlich carcinoma cells
Scientific Reports (2023)
-
Genotoxicity and oxidative stress induction by calcium hydroxide, calcium titanate or/and yttrium oxide nanoparticles in mice
Scientific Reports (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.