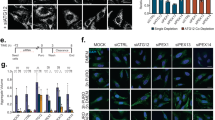
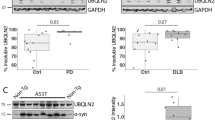
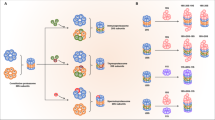
Abstract
Many late-onset neurodegenerative diseases, including Parkinson's disease and Huntington's disease, are associated with the formation of intracellular aggregates by toxic proteins. It is therefore crucial to understand the factors that regulate the steady-state levels of these 'toxins', at both the synthetic and degradation stages. The degradation pathways acting on such aggregate-prone cytosolic proteins include the ubiquitin–proteasome system and macroautophagy. Dysfunction of the ubiquitin–proteasome or macroautophagy pathways might contribute to the pathology of various neurodegenerative conditions. However, enhancing macroautophagy with drugs such as rapamycin could offer a tractable therapeutic strategy for a number of these diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Taylor, J. P., Hardy, J. & Fischbeck, K. H. Toxic proteins in neurodegenerative disease. Science 296, 1991–1995 (2002).
Ross, C. A. & Poirier, M. A. What is the role of protein aggregation in neurodegeneration? Nature Rev. Mol. Cell Biol. 6, 891–898 (2005).
Perutz, M. F. & Windle, A. H. Cause of neural death in neurodegenerative diseases attributable to expansion of glutamine repeats. Nature 412, 143–144 (2001).
Pangalos, M. N., Jacobsen, S. J. & Reinhart, P. H. Disease modifying strategies for the treatment of Alzheimer's disease targeted at modulating levels of the β-amyloid peptide. Biochem. Soc. Trans. 33, 553–558 (2005).
Wellington, C. L. et al. Caspase cleavage of mutant huntingtin precedes neurodegeneration in Huntington's disease. J. Neurosci. 22, 7862–7872 (2002).
Luo, S., Vacher, C., Davies, J. E. & Rubinsztein, D. C. Cdk5 phosphorylation of huntingtin reduces its cleavage by caspases: implications for mutant huntingtin toxicity. J. Cell Biol. 169, 647–656 (2005).
Wellington, C. L. et al. Caspase cleavage of gene products associated with triplet expansion disorders generates truncated fragments containing the polyglutamine tract. J. Biol. Chem. 273, 9158–9167 (1998).
Ciechanover, A. The ubiquitin proteolytic system: from a vague idea, through basic mechanisms, and onto human diseases and drug targeting. Neurology 66, S7–S19 (2006).
Richly, H. et al. A series of ubiquitin binding factors connects CDC48/p97 to substrate multiubiquitylation and proteasomal targeting. Cell 120, 73–84 (2005).
Weihl, C. C., Dalal, S., Pestronk, A. & Hanson, P. I. Inclusion body myopathy-associated mutations in p97/VCP impair endoplasmic reticulum-associated degradation. Hum. Mol. Genet. 15, 189–199 (2006).
Ravikumar, B., Duden, R. & Rubinsztein, D. C. Aggregate-prone proteins with polyglutamine and polyalanine expansions are degraded by autophagy. Hum. Mol. Genet. 11, 1107–1117 (2002).
Webb, J. L., Ravikumar, B., Atkins, J., Skepper, J. N. & Rubinsztein, D. C. α-Synuclein is degraded by both autophagy and the proteasome. J. Biol. Chem. 278, 25009–25013 (2003).
Verhoef, L. G., Lindsten, K., Masucci, M. G. & Dantuma, N. P. Aggregate formation inhibits proteasomal degradation of polyglutamine proteins. Hum. Mol. Genet. 11, 2689–2700 (2002).
Venkatraman, P., Wetzel, R., Tanaka, M., Nukina, N. & Goldberg, A. L. Eukaryotic proteasomes cannot digest polyglutamine sequences and release them during degradation of polyglutamine-containing proteins. Mol. Cell 14, 95–104 (2004).
Holmberg, C. I., Staniszewski, K. E., Mensah, K. N., Matouschek, A. & Morimoto, R. I. Inefficient degradation of truncated polyglutamine proteins by the proteasome. EMBO J. 23, 4307–4318 (2004).
Yorimitsu, T. & Klionsky, D. J. Autophagy: molecular machinery for self-eating. Cell Death Differ. 12, 1542–1552 (2005).
Shibata, M. et al. Regulation of intracellular accumulation of mutant Huntingtin by Beclin 1. J. Biol. Chem. 281, 14474–14485 (2006).
Iwata, A. et al. Increased susceptibility of cytoplasmic over nuclear polyglutamine aggregates to autophagic degradation. Proc. Natl Acad. Sci. USA 102, 13135–13140 (2005).
Qin, Z. H. et al. Autophagy regulates the processing of amino terminal huntingtin fragments. Hum. Mol. Genet. 12, 3231–3244 (2003).
Berger, Z. et al. Rapamycin alleviates toxicity of different aggregate-prone proteins. Hum. Mol. Genet. 15, 433–442 (2006).
Ravikumar, B. et al. mTOR inhibition induces autophagy and reduces toxicity of the Huntington's disease mutation in Drosophila and mouse models. Nature Genet. 36, 585–595 (2004).
Komatsu, M. et al. Impairment of starvation-induced and constitutive autophagy in Atg7-deficient mice. J. Cell Biol. 169, 425–434 (2005).
Ravikumar, B. et al. Raised intracellular glucose concentrations reduce aggregation and cell death caused by mutant huntingtin exon 1 by decreasing mTOR phosphorylation and inducing autophagy. Hum. Mol. Genet. 12, 985–994 (2003).
Rideout, H., Lang-Rollin, I. & Stefanis, L. Involvement of macroautophagy in the dissolution of neuronal inclusions. Int. J. Biochem. Cell Biol. 36, 2551–2562 (2004).
Massey, A., Kiffin, R. & Cuervo, A. M. Pathophysiology of chaperone-mediated autophagy. Int. J. Biochem. Cell Biol. 36, 2420–2434 (2004).
McNaught, K. S., Belizaire, R., Isacson, O., Jenner, P. & Olanow, C. W. Altered proteasomal function in sporadic Parkinson's disease. Exp. Neurol. 179, 38–46 (2003).
Seo, H., Sonntag, K. C. & Isacson, O. Generalized brain and skin proteasome inhibition in Huntington's disease. Ann. Neurol. 56, 319–328 (2004).
Bence, N. F., Sampat, R. M. & Kopito, R. R. Impairment of the ubiquitin–proteasome system by protein aggregation. Science 292, 1552–1555 (2001).
Bowman, A. B., Yoo, S. Y., Dantuma, N. P. & Zoghbi, H. Y. Neuronal dysfunction in a polyglutamine disease model occurs in the absence of ubiquitin–proteasome system impairment and inversely correlates with a degree of nuclear inclusion formation. Hum. Mol. Genet. 14, 679–691 (2005).
Diaz-Hernandez, M. et al. Neuronal induction of the immunoproteasome in Huntington's disease. J. Neurosci. 23, 11653–11661 (2003).
Sun, X. M. et al. Caspase activation inhibits proteasome function during apoptosis. Mol. Cell 14, 81–93 (2004).
Zhang, Y. et al. Parkin functions as an E2-dependent ubiquitin–protein ligase and promotes the degradation of the synaptic vesicle-associated protein, CDCrel-1. Proc. Natl Acad. Sci. USA 97, 13354–13359 (2000).
Shimura, H. et al. Familial Parkinson disease gene product, parkin, is a ubiquitin–protein ligase. Nature Genet. 25, 302–305 (2000).
Imai, Y., Soda, M. & Takahashi, R. Parkin suppresses unfolded protein stress-induced cell death through its E3 ubiquitin–protein ligase activity. J. Biol. Chem. 275, 35661–35664 (2000).
Leroy, E. et al. The ubiquitin pathway in Parkinson's disease. Nature 395, 451–452 (1998).
Moore, D. J., West, A. B., Dawson, V. L. & Dawson, T. M. Molecular pathophysiology of Parkinson's disease. Annu. Rev. Neurosci. 28, 57–87 (2005).
Healy, D. G. et al. UCHL-1 is not a Parkinson's disease susceptibility gene. Ann. Neurol. 59, 627–633 (2006).
Liu, Y., Fallon, L., Lashuel, H. A., Liu, Z. & Lansbury, P. T. The UCH-L1 gene encodes two opposing enzymatic activities that affect α-synuclein degradation and Parkinson's disease susceptibility. Cell 111, 209–218 (2002).
Saigoh, K. et al. Intragenic deletion in the gene encoding ubiquitin carboxy-terminal hydrolase in gad mice. Nature Genet. 23, 47–51 (1999).
Watts, G. D. et al. Inclusion body myopathy associated with Paget disease of bone and frontotemporal dementia is caused by mutant valosin-containing protein. Nature Genet. 36, 377–381 (2004).
McNaught, K. S., Perl, D. P., Brownell, A. L. & Olanow, C. W. Systemic exposure to proteasome inhibitors causes a progressive model of Parkinson's disease. Ann. Neurol. 56, 149–162 (2004).
Kuma, A. et al. The role of autophagy during the early neonatal starvation period. Nature 432, 1032–1036 (2004).
Boya, P. et al. Inhibition of macroautophagy triggers apoptosis. Mol. Cell Biol. 25, 1025–1040 (2005).
Ravikumar, B., Berger, Z., Vacher, C., O'Kane, C. J. & Rubinsztein, D. C. Rapamycin pre-treatment protects against apoptosis. Hum. Mol. Genet. 15, 1209–1216 (2006).
Komatsu, M. et al. Loss of autophagy in the central nervous system causes neurodegeneration in mice. Nature 441, 880–884 (2006).
Hara, T. et al. Suppression of basal autophagy in neural cells causes neurodegenerative disease in mice. Nature 441, 885–889 (2006).
Ravikumar, B. et al. Dynein mutations impair autophagic clearance of aggregate-prone proteins. Nature Genet. 37, 771–776 (2005).
Levy, J. R. et al. A motor neuron disease-associated mutation in p150Glued perturbs dynactin function and induces protein aggregation. J. Cell Biol. 172, 733–745 (2006).
Puls, I. et al. Mutant dynactin in motor neuron disease. Nature Genet. 33, 455–456 (2003).
Hafezparast, M. et al. Mutations in dynein link motor neuron degeneration to defects in retrograde transport. Science 300, 808–812 (2003).
Kieran, D. et al. A mutation in dynein rescues axonal transport defects and extends the life span of ALS mice. J. Cell Biol. 169, 561–567 (2005).
Ligon, L. A. et al. Mutant superoxide dismutase disrupts cytoplasmic dynein in motor neurons. Neuroreport 16, 533–536 (2005).
Cuervo, A. M., Stefansi, L., Fredenburg, R., Lansbury, P. T. & Sulzer, D. Impaired degradation of mutant α-synuclein by chaperone-mediated autophagy. Science 305, 1292–1295 (2004).
Harper, S. Q. et al. RNA interference improves motor and neuropathological abnormalities in a Huntington's disease mouse model. Proc. Natl Acad. Sci. USA 102, 5820–5825 (2005).
Rodriguez-Lebron, E. & Paulson, H. L. Allele-specific RNA interference for neurological disease. Gene. Ther. 13, 576–581 (2006).
Davies, J. E., Sarkar, S. & Rubinsztein, D. C. Trehalose reduces aggregate formation and delays pathology in a transgenic mouse model of oculopharyngeal muscular dystrophy. Hum. Mol. Genet. 15, 23–31 (2006).
Sanchez, I., Mahlke, C. & Yuan, Y. Pivotal role of oligomerization in expanded polyglutamine neurodegenerative disorders. Nature 421, 373–379 (2003).
Martin-Aparicio, E. et al. Proteasomal-dependent aggregate reversal and absence of cell death in a conditional mouse model of Huntington's disease. J. Neurosci. 21, 8772–8781 (2001).
Yamamoto, A., Cremona, M. L. & Rothman, J. E. Autophagy-mediated clearance of huntingtin aggregates triggered by the insulin-signaling pathway. J. Cell Biol. 172, 719–731 (2006).
DiFiglia, M. et al. Aggregation of huntingtin in neuronal intranuclear inclusions and dystrophic neurites in brain. Science 277, 1990–1993 (1997).
Kiffin, R., Bandyopadhyay, U. & Cuervo, A. Oxidative stress and autophagy. Antioxid. Redox Signal. 8, 152–162 (2006).
Arrasate, M., Mitra, S., Schweitzer, E. S., Segal, M. R. & Finkbeiner, S. Inclusion body formation reduces levels of mutant huntingtin and the risk of neuronal death. Nature 431, 805–810 (2004).
Lee, V. W. & Chapman, J. R. Sirolimus: its role in nephrology. Nephrology (Carlton) 10, 606–614 (2005).
Bjornsti, M. A. & Houghton, P. J. The TOR pathway: a target for cancer therapy. Nature Rev. Cancer 4, 335–348 (2004).
Galanis, E. et al. Phase II trial of temsirolimus (CCI-779) in recurrent glioblastoma multiforme: a North Central Cancer Treatment Group Study. J. Clin. Oncol. 23, 5294–5304 (2005).
Wullschleger, S., Loewith, R. & Hall, M. N. TOR signaling in growth and metabolism. Cell 124, 471–484 (2006).
Sarkar, S. et al. Lithium induces autophagy by inhibiting inositol monophosphatase. J. Cell Biol. 170, 1101–1111 (2005).
Murphy, M. P. How understanding the control of energy metabolism can help investigation of mitochondrial dysfunction, regulation and pharmacology. Biochim. Biophys. Acta 1504, 1–11 (2001).
Pyo, J. et al. Essential roles of Atg5 and FADD in autophagic cell death: dissection of autophagic cell death into vacuole formation and cell death. J. Biol. Chem. 280, 20722–20728 (2005).
Nasir, J. et al. Targeted disruption of the Huntington's disease gene results in embryonic lethality and behavioural and morphological changes in heterozygotes. Cell 81, 811–823 (1995).
Zeitlin, S. et al. Increased apoptosis and early embryonic lethality in mice nullizygous for the Huntington's disease gene homolog. Nature Genet. 11, 155–163 (1995).
Duyao, M. P. et al. Inactivation of the mouse Huntington's disease gene homolog Hdh. Science 269, 407–410 (1995).
Harper, P. S. Huntington's Disease 2nd edn (WB Saunders, London, 1996).
Rubinsztein, D. C. Lessons from animal models of Huntington's disease. Trends Genet. 18, 202–209 (2002).
Ordway, J. M. et al. Ectopically expressed CAG repeats cause intranuclear inclusions and a progressive late onset neurological phenotype in the mouse. Cell 91, 753–763 (1997).
Cattaneo, E., Zuccato, C. & Tartari, M. Normal huntingtin function: an alternative approach to Huntington's disease. Nature Rev. Neurosci. 6, 919–930 (2005).
Acknowledgements
I am grateful to B. Ravikumar and M. Futter for critical comments on the manuscript and L. Smith for help with manuscript preparation. The work in my laboratory covered by this review has been funded by a Wellcome Trust Senior Fellowship in Clinical Science, a Medical Research Council (MRC) Programme Grant, Wyeth, and European Union Framework VI (EUROSCA).
Author information
Authors and Affiliations
Ethics declarations
Competing interests
D.C.R. is an inventor on patents relating to the use of autophagy induction for treating neurodegenerative diseases. His laboratory has received grant funding from Wyeth, which makes rapamycins.
Additional information
Author Information Reprints and permissions information is available at npg.nature.com/reprintsandpermissions.
Rights and permissions
About this article
Cite this article
Rubinsztein, D. The roles of intracellular protein-degradation pathways in neurodegeneration. Nature 443, 780–786 (2006). https://doi.org/10.1038/nature05291
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature05291
This article is cited by
-
Bilirubin metabolism: delving into the cellular and molecular mechanisms to predict complications
The Egyptian Journal of Internal Medicine (2024)
-
Evaluation of the mTORC activity in the presence of Toxoplasma gondii and azathioprine in human monocyte cell line
BMC Microbiology (2023)
-
ACSL1-induced ferroptosis and platinum resistance in ovarian cancer by increasing FSP1 N-myristylation and stability
Cell Death Discovery (2023)
-
Tubular lysosome induction couples animal starvation to healthy aging
Nature Aging (2023)
-
Mitochondrial Hydrogen Peroxide Activates PTEN and Inactivates Akt Leading to Autophagy Inhibition-Dependent Cell Death in Neuronal Models of Parkinson’s Disease
Molecular Neurobiology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.