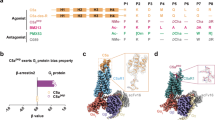
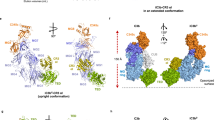
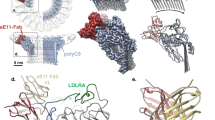
Abstract
The complement system is a key part of the innate immune system, and is required for clearance of pathogens from the bloodstream1. After exposure to pathogens, the third component of the complement system, C3, is cleaved to C3b which, after recruitment of factor B, initiates formation of the alternative pathway convertases2,3,4. CRIg, a complement receptor expressed on macrophages, binds to C3b and iC3b mediating phagocytosis of the particles5, but it is unknown how CRIg selectively recognizes proteolytic C3-fragments and whether binding of CRIg to C3b inhibits convertase activation. Here we present the crystal structure of C3b in complex with CRIg and, using CRIg mutants, provide evidence that CRIg acts as an inhibitor of the alternative pathway of complement. The structure shows that activation of C3 induces major structural rearrangements, including a dramatic movement (>80 Å) of the thioester-bond-containing domain through which C3b attaches to pathogen surfaces. We show that CRIg is not only a phagocytic receptor, but also a potent inhibitor of the alternative pathway convertases. The structure provides insights into the complex macromolecular structural rearrangements that occur during complement activation and inhibition. Moreover, our structure–function studies relating the structural basis of complement activation and the means by which CRIg inhibits the convertases provide important clues to the development of therapeutics that target complement.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Walport, M. J. Complement. First of two parts. N. Engl. J. Med. 344, 1058–1066 (2001)
Pangburn, M. K. & Muller-Eberhard, H. J. Relation of putative thioester bond in C3 to activation of the alternative pathway and the binding of C3b to biological targets of complement. J. Exp. Med. 152, 1102–1114 (1980)
Fishelson, Z., Pangburn, M. K. & Muller-Eberhard, H. J. C3 convertase of the alternative complement pathway. Demonstration of an active, stable C3b, Bb (Ni) complex. J. Biol. Chem. 258, 7411–7415 (1983)
Muller-Eberhard, H. J. & Schreiber, R. D. Molecular biology and chemistry of the alternative pathway of complement. Adv. Immunol. 29, 1–53 (1980)
Helmy, K. Y. et al. CRIg: a macrophage complement receptor required for phagocytosis of circulating pathogens. Cell 124, 915–927 (2006)
Pangburn, M. K., Schreiber, R. D. & Muller-Eberhard, H. J. Formation of the initial C3 convertase of the alternative complement pathway. Acquisition of C3b-like activities by spontaneous hydrolysis of the putative thioester in native C3. J. Exp. Med. 154, 856–867 (1981)
Sahu, A. & Lambris, J. D. Structure and biology of complement protein C3, a connecting link between innate and acquired immunity. Immunol. Rev. 180, 35–48 (2001)
Janssen, B. J. et al. Structures of complement component C3 provide insights into the function and evolution of immunity. Nature 437, 505–511 (2005)
Fredslund, F. et al. The structure of bovine complement component 3 reveals the basis for thioester function. J. Mol. Biol. 361, 115–127 (2006)
Lambris, J. D. et al. Dissection of CR1, factor H, membrane cofactor protein, and factor B binding and functional sites in the third complement component. J. Immunol. 156, 4821–4832 (1996)
Taniguchi-Sidle, A. & Isenman, D. E. Interactions of human complement component C3 with factor B and with complement receptors type 1 (CR1, CD35) and type 3 (CR3, CD11b/CD18) involve an acidic sequence at the N-terminus of C3 alpha'-chain. J. Immunol. 153, 5285–5302 (1994)
Oran, A. E. & Isenman, D. E. Identification of residues within the 727–767 segment of human complement component C3 important for its interaction with factor H and with complement receptor 1 (CR1, CD35). J. Biol. Chem. 274, 5120–5130 (1999)
Liddington, R. & Bankston, L. Structural biology: origins of chemical biodefence. Nature 437, 484–485 (2005)
Gadjeva, M. et al. The covalent binding reaction of complement component C3. J. Immunol. 161, 985–990 (1998)
Langnaese, K., Colleaux, L., Kloos, D. U., Fontes, M. & Wieacker, P. Cloning of Z39Ig, a novel gene with immunoglobulin-like domains located on human chromosome X. Biochim. Biophys. Acta 1492, 522–525 (2000)
Janssen, B. J. & Gros, P. Structural insights into the central complement component C3. Mol. Immunol. (in the press).
Pangburn, M. K. & Muller-Eberhard, H. J. The C3 convertase of the alternative pathway of human complement. Enzymic properties of the bimolecular proteinase. Biochem. J. 235, 723–730 (1986)
Vogt, W., Schmidt, G., Von Buttlar, B. & Dieminger, L. A new function of the activated third component of complement: binding to C5, an essential step for C5 activation. Immunology 34, 29–40 (1978)
Rawal, N. & Pangburn, M. K. Functional role of the noncatalytic subunit of complement C5 convertase. J. Immunol. 164, 1379–1385 (2000)
Seelen, M. A. et al. Functional analysis of the classical, alternative, and MBL pathways of the complement system: standardization and validation of a simple ELISA. J. Immunol. Methods 296, 187–198 (2005)
Kostavasili, I. et al. Mechanism of complement inactivation by glycoprotein C of herpes simplex virus. J. Immunol. 158, 1763–1771 (1997)
Rawal, N. & Pangburn, M. K. C5 convertase of the alternative pathway of complement. Kinetic analysis of the free and surface-bound forms of the enzyme. J. Biol. Chem. 273, 16828–16835 (1998)
Krych-Goldberg, M. et al. Decay accelerating activity of complement receptor type 1 (CD35). Two active sites are required for dissociating C5 convertases. J. Biol. Chem. 274, 31160–31168 (1999)
Acknowledgements
We thank B. de Vos, A. Chan, F. Bazan and H. Spits for critically reading the manuscript, and J. Chinn and B. Appleton for important contributions. We acknowledge the support staff at beamline 5.0.2 at the Advanced Light Source. The Advanced Light Source is supported by the Director, Office of Science, Office of Basic Energy Sciences, Materials Sciences Division of the US Department of Energy at Lawrence Berkeley National Laboratory. Author Contributions K.J.K., JP.Y., K.Y.H. and M.S. contributed equally to this work. K.J.K., L.E. and L.DF. performed the C3 convertase and haemolysis assay, S.A.MC. and W.J.F. contributed to mutant design, K.Y.H. generated CRIg mutants and performed the microwell assays, M.S. and P.E.H. purified the complement proteins, JP.Y. generated CRIg for crystallography, purified the complexes and grew the crystals, C.W. solved the structures and wrote the paper together with M.v.L.C.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Coordinates and structure factors have been deposited at the RCSB Protein Databank (2ICC, 2ICE, and 2ICF). Reprints and permission information is available at www.nature.com/reprints. The authors declare no competing financial interests.
Supplementary information
Supplementary Notes
Structure of complement receptor CRIg bound to C3b provides insight into the regulation of complement activation. This file contains Supplementary Methods, Supplementary Table and Supplementary Figure Legends. (DOC 94 kb)
Supplementary Figure 1
Differences in domain arrangement between C3 and C3b. (PDF 152 kb)
Supplementary Figure 2
CRIg has similar affinities for C3b and C3c. (PDF 25 kb)
Supplementary Figure 3a
Sequence alignment of CRIg from different species. (PDF 164 kb)
Supplementary Figure 3b
Sequence alignment of human and mouse C3 (PDF 562 kb)
Supplementary Figure 4
Local differences between C3 and C3b in the vicinity of the CRIg binding site. (PDF 266 kb)
Supplementary Figure 5
CRIg inhibits cleavage of C3 in a fluid-phase C3 convertase assay. (PDF 99 kb)
Supplementary Figure 6
CRIg does not accelerate decay of the C3 convertase. (PDF 93 kb)
Supplementary Figure 7
CRIg does not inhibit the CP convertase. (PDF 23 kb)
Rights and permissions
About this article
Cite this article
Wiesmann, C., Katschke, K., Yin, J. et al. Structure of C3b in complex with CRIg gives insights into regulation of complement activation. Nature 444, 217–220 (2006). https://doi.org/10.1038/nature05263
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature05263
This article is cited by
-
CRIg on liver macrophages clears pathobionts and protects against alcoholic liver disease
Nature Communications (2021)
-
How novel structures inform understanding of complement function
Seminars in Immunopathology (2018)
-
Molecular insights into the surface-specific arrangement of complement C5 convertase enzymes
BMC Biology (2015)
-
Structure of a bacterial α2-macroglobulin reveals mimicry of eukaryotic innate immunity
Nature Communications (2014)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.