Abstract
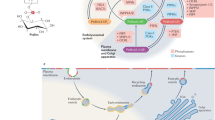
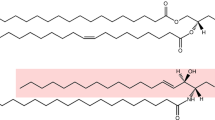
Inositol phospholipids have long been known to have an important regulatory role in cell physiology. The repertoire of cellular processes known to be directly or indirectly controlled by this class of lipids has now dramatically expanded. Through interactions mediated by their headgroups, which can be reversibly phosphorylated to generate seven species, phosphoinositides play a fundamental part in controlling membrane–cytosol interfaces. These lipids mediate acute responses, but also act as constitutive signals that help define organelle identity. Their functions, besides classical signal transduction at the cell surface, include regulation of membrane traffic, the cytoskeleton, nuclear events and the permeability and transport functions of membranes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Hokin, L. E. Receptors and phosphoinositide-generated second messengers. Annu. Rev. Biochem. 54, 205–235 (1985)
Berridge, M. J. & Irvine, R. F. Inositol phosphates and cell signalling. Nature 341, 197–205 (1989)
Lassing, I. & Lindberg, U. Specific interaction between phosphatidylinositol 4,5-bisphosphate and profilactin. Nature 314, 472–474 (1985)
Ma, L., Cantley, L. C., Janmey, P. A. & Kirschner, M. W. Corequirement of specific phosphoinositides and small GTP-binding protein Cdc42 in inducing actin assembly in Xenopus egg extracts. J. Cell Biol. 140, 1125–1136 (1998)
Yin, H. L. & Janmey, P. A. Phosphoinositide regulation of the actin cytoskeleton. Annu. Rev. Physiol. 65, 761–789 (2003)
Whitman, M., Downes, C. P., Keeler, M., Keller, T. & Cantley, L. Type I phosphatidylinositol kinase makes a novel inositol phospholipid, phosphatidylinositol-3-phosphate. Nature 332, 644–646 (1988)
Traynor-Kaplan, A. E., Harris, A. L., Thompson, B. L., Taylor, P. & Sklar, L. A. An inositol tetrakisphosphate-containing phospholipid in activated neutrophils. Nature 334, 353–356 (1988)
Auger, K. R. et al. PDGF-dependent tyrosine phosphorylation stimulates production of novel polyphosphoinositides in intact cells. Cell 57, 167–175 (1989)
Eberhard, D. A., Cooper, C. L., Low, M. G. & Holz, R. W. Evidence that the inositol phospholipids are necessary for exocytosis. Loss of inositol phospholipids and inhibition of secretion in permeabilized cells caused by a bacterial phospholipase C and removal of ATP. Biochem. J. 268, 15–25 (1990)
Schu, P. V. et al. Phosphatidylinositol 3-kinase encoded by yeast VPS34 gene essential for protein sorting. Science 260, 88–91 (1993)
McPherson, P. S. et al. A presynaptic inositol-5-phosphatase. Nature 379, 353–357 (1996)
Martin, T. F. Phosphoinositide lipids as signaling molecules: common themes for signal transduction, cytoskeletal regulation, and membrane trafficking. Annu. Rev. Cell Dev. Biol. 14, 231–264 (1998)
Dove, S. K. et al. Osmotic stress activates phosphatidylinositol-3,5-bisphosphate synthesis. Nature 390, 187–192 (1997)
Rameh, L. E., Tolias, K. F., Duckworth, B. C. & Cantley, L. C. A new pathway for synthesis of phosphatidylinositol-4,5-bisphosphate. Nature 390, 192–196 (1997)
Lemmon, M. A. Phosphoinositide recognition domains. Traffic 4, 201–213 (2003)
Balla, T. Inositol-lipid binding motifs: signal integrators through protein–lipid and protein–protein interactions. J. Cell Sci. 118, 2093–2104 (2005)
Majerus, P. W., Kisseleva, M. V. & Norris, F. A. The role of phosphatases in inositol signaling reactions. J. Biol. Chem. 274, 10669–10672 (1999)
Odorizzi, G., Babst, M. & Emr, S. D. Phosphoinositide signaling and the regulation of membrane trafficking in yeast. Trends Biochem. Sci. 25, 229–235 (2000)
Roth, M. G. Phosphoinositides in constitutive membrane traffic. Physiol. Rev. 84, 699–730 (2004)
Hammond, G., Thomas, C. L. & Schiavo, G. Nuclear phosphoinositides and their functions. Curr. Top. Microbiol. Immunol. 282, 177–206 (2004)
Hurley, J. H. & Meyer, T. Subcellular targeting by membrane lipids. Curr. Opin. Cell Biol. 13, 146–152 (2001)
Wenk, M. R. & De Camilli, P. Protein–lipid interactions and phosphoinositide metabolism in membrane traffic: insights from vesicle recycling in nerve terminals. Proc. Natl Acad. Sci. USA 101, 8262–8269 (2004)
Behnia, R. & Munro, S. Organelle identity and the signposts for membrane traffic. Nature 438, 597–604 (2005)
Gaidarov, I. & Keen, J. H. Phosphoinositide–AP-2 interactions required for targeting to plasma membrane clathrin-coated pits. J. Cell Biol. 146, 755–764 (1999)
Wang, Y. J. et al. Phosphatidylinositol 4 phosphate regulates targeting of clathrin adaptor AP-1 complexes to the Golgi. Cell 114, 299–310 (2003)
Honing, S. et al. Phosphatidylinositol-(4,5)-bisphosphate regulates sorting signal recognition by the clathrin-associated adaptor complex AP2. Mol. Cell 18, 519–531 (2005)
Owen, D. J., Collins, B. M. & Evans, P. R. Adaptors for clathrin coats: structure and function. Annu. Rev. Cell Dev. Biol. 20, 153–191 (2004)
Honda, A. et al. Phosphatidylinositol 4-phosphate 5-kinase α is a downstream effector of the small G protein ARF6 in membrane ruffle formation. Cell 99, 521–532 (1999)
Shin, H. W. et al. An enzymatic cascade of Rab5 effectors regulates phosphoinositide turnover in the endocytic pathway. J. Cell Biol. 170, 607–618 (2005)
Cremona, O. et al. Essential role of phosphoinositide metabolism in synaptic vesicle recycling. Cell 99, 179–188 (1999)
Cantley, L. C. The phosphoinositide 3-kinase pathway. Science 296, 1655–1657 (2002)
Czech, M. P. Dynamics of phosphoinositides in membrane retrieval and insertion. Annu. Rev. Physiol. 65, 791–815 (2003)
Katso, R. et al. Cellular function of phosphoinositide 3-kinases: implications for development, homeostasis, and cancer. Annu. Rev. Cell Dev. Biol. 17, 615–675 (2001)
Wishart, M. J. & Dixon, J. E. PTEN and myotubularin phosphatases: from 3-phosphoinositide dephosphorylation to disease. Trends Cell Biol. 12, 579–585 (2002)
Li, J. et al. PTEN, a putative protein tyrosine phosphatase gene mutated in human brain, breast, and prostate cancer. Science 275, 1943–1947 (1997)
Hilgemann, D. W., Feng, S. & Nasuhoglu, C. The complex and intriguing lives of PIP2 with ion channels and transporters. Sci. STKE 2001, RE19, doi:10.1126/stke.2001.111.re19 (2001)
Suh, B. C. & Hille, B. Regulation of ion channels by phosphatidylinositol 4,5-bisphosphate. Curr. Opin. Neurobiol. 15, 370–378 (2005)
Pollard, T. D. & Borisy, G. G. Cellular motility driven by assembly and disassembly of actin filaments. Cell 112, 453–465 (2003)
Rohatgi, R., Ho, H. Y. & Kirschner, M. W. Mechanism of N-WASP activation by CDC42 and phosphatidylinositol 4,5-bisphosphate. J. Cell Biol. 150, 1299–1310 (2000)
Ho, H. Y. et al. Toca-1 mediates Cdc42-dependent actin nucleation by activating the N-WASP–WIP complex. Cell 118, 203–216 (2004)
Di Paolo, G. et al. Recruitment and regulation of phosphatidylinositol phosphate kinase type 1 γ by the FERM domain of talin. Nature 420, 85–89 (2002)
Ling, K. et al. Type I γ phosphatidylinositol phosphate kinase targets and regulates focal adhesions. Nature 420, 89–93 (2002)
Oikawa, T. et al. PtdIns(3,4,5)P3 binding is necessary for WAVE2-induced formation of lamellipodia. Nature Cell Biol. 6, 420–426 (2004)
Iijima, M., Huang, Y. E. & Devreotes, P. Temporal and spatial regulation of chemotaxis. Dev. Cell 3, 469–478 (2002)
Golub, T. & Caroni, P. PI(4,5)P2-dependent microdomain assemblies capture microtubules to promote and control leading edge motility. J. Cell Biol. 169, 151–165 (2005)
Klopfenstein, D. R. & Vale, R. D. The lipid binding pleckstrin homology domain in UNC-104 kinesin is necessary for synaptic vesicle transport in Caenorhabditis elegans. Mol. Biol. Cell 15, 3729–3739 (2004)
Bai, J., Tucker, W. C. & Chapman, E. R. PIP2 increases the speed of response of synaptotagmin and steers its membrane-penetration activity toward the plasma membrane. Nature Struct. Mol. Biol. 11, 36–44 (2004)
Rothman, J. E. The protein machinery of vesicle budding and fusion. Protein Sci. 5, 185–194 (1996)
Milosevic, I. et al. Plasmalemmal phosphatidylinositol-4,5-bisphosphate level regulates the releasable vesicle pool size in chromaffin cells. J. Neurosci. 25, 2557–2565 (2005)
Gong, L. W. et al. Phosphatidylinositol phosphate kinase type I γ regulates dynamics of large dense-core vesicle fusion. Proc. Natl Acad. Sci. USA 102, 5204–5209 (2005)
Di Paolo, G. et al. Impaired PtdIns(4,5)P2 synthesis in nerve terminals produces defects in synaptic vesicle trafficking. Nature 431, 415–422 (2004)
Lackner, M. R., Nurrish, S. J. & Kaplan, J. M. Facilitation of synaptic transmission by EGL-30 Gqα and EGL-8 PLCβ: DAG binding to UNC-13 is required to stimulate acetylcholine release. Neuron 24, 335–346 (1999)
Rhee, J. S. et al. β phorbol ester- and diacylglycerol-induced augmentation of transmitter release is mediated by Munc13s and not by PKCs. Cell 108, 121–133 (2002)
Engqvist-Goldstein, A. E. & Drubin, D. G. Actin assembly and endocytosis: from yeast to mammals. Annu. Rev. Cell Dev. Biol. 19, 287–332 (2003)
Stefan, C. J., Audhya, A. & Emr, S. D. The yeast synaptojanin-like proteins control the cellular distribution of phosphatidylinositol (4,5)-bisphosphate. Mol. Biol. Cell 13, 542–557 (2002)
Botelho, R. J., Scott, C. C. & Grinstein, S. Phosphoinositide involvement in phagocytosis and phagosome maturation. Curr. Top. Microbiol. Immunol. 282, 1–30 (2004)
Pizarro-Cerda, J. & Cossart, P. Subversion of phosphoinositide metabolism by intracellular bacterial pathogens. Nature Cell Biol. 6, 1026–1033 (2004)
Niebuhr, K. et al. Conversion of PtdIns(4,5)P2 into PtdIns(5)P by the S. flexneri effector IpgD reorganizes host cell morphology. EMBO J. 21, 5069–5078 (2002)
Terebiznik, M. R. et al. Elimination of host cell PtdIns(4,5)P2 by bacterial SigD promotes membrane fission during invasion by Salmonella. Nature Cell Biol. 4, 766–773 (2002)
Vergne, I. et al. Mechanism of phagolysosome biogenesis block by viable Mycobacterium tuberculosis. Proc. Natl Acad. Sci. USA 102, 4033–4038 (2005)
Ungewickell, A. et al. The identification and characterization of two phosphatidylinositol-4,5-bisphosphate 4-phosphatases. Proc. Natl Acad. Sci. USA 102, 18854–18859 (2005)
Gaynor, E. C., Chen, C. Y., Emr, S. D. & Graham, T. R. ARF is required for maintenance of yeast Golgi and endosome structure and function. Mol. Biol. Cell 9, 653–670 (1998)
De Matteis, M. A., Di Campli, A. & Godi, A. The role of the phosphoinositides at the Golgi complex. Biochim. Biophys. Acta 1744, 396–405 (2005)
Guo, S., Stolz, L. E., Lemrow, S. M. & York, J. D. SAC1-like domains of yeast SAC1, INP52, and INP53 and of human synaptojanin encode polyphosphoinositide phosphatases. J. Biol. Chem. 274, 12990–12995 (1999)
Roy, A. & Levine, T. P. Multiple pools of phosphatidylinositol 4-phosphate detected using the pleckstrin homology domain of Osh2p. J. Biol. Chem. 279, 44683–44689 (2004)
Attree, O. et al. The Lowe's oculocerebrorenal syndrome gene encodes a protein highly homologous to inositol polyphosphate-5-phosphatase. Nature 358, 239–242 (1992)
Birkeland, H. C. & Stenmark, H. Protein targeting to endosomes and phagosomes via FYVE and PX domains. Curr. Top. Microbiol. Immunol. 282, 89–115 (2004)
Zerial, M. & McBride, H. Rab proteins as membrane organizers. Nature Rev. Mol. Cell Biol. 2, 107–117 (2001)
Michell, R. H., Heath, V. L., Lemmon, M. A. & Dove, S. K. Phosphatidylinositol 3,5-bisphosphate: metabolism and cellular functions. Trends Biochem. Sci., 52–63 (2005)
Laporte, J. et al. A gene mutated in X-linked myotubular myopathy defines a new putative tyrosine phosphatase family conserved in yeast. Nature Genet. 13, 175–182 (1996)
Bolino, A. et al. Charcot-Marie-Tooth type 4B is caused by mutations in the gene encoding myotubularin-related protein-2. Nature Genet. 25, 17–19 (2000)
Li, S. et al. Mutations in PIP5K3 are associated with Francois-Neetens mouchetee fleck corneal dystrophy. Am. J. Hum. Genet. 77, 54–63 (2005)
McLaughlin, S. & Murray, D. Plasma membrane phosphoinositide organization by protein electrostatics. Nature 438, 605–611 (2005)
Bader, A. G., Kang, S., Zhao, L. & Vogt, P. K. Oncogenic PI3K deregulates transcription and translation. Nature Rev. Cancer 5, 921–929 (2005)
Halstead, J. R., Jalink, K. & Divecha, N. An emerging role for PtdIns(4,5)P2-mediated signalling in human disease. Trends Pharmacol. Sci. 26, 654–660 (2005)
Acknowledgements
We thank N. Borgese, V. Haucke and O. Cremona for critical reading of the manuscript. We also thank B. Chang for providing the fluorescence images in Fig. 1 and for her comments on the manuscript. DNA constructs were gifts from H. Stenmark, T. Meyer and A. De Matteis. We apologize to all the scientists whose original studies and reviews were not quoted in our manuscript owing to space limitations. G.D.P. is funded by grants from the National Institute of Health. P.D.C is funded by the HHMI and by grants from the National Institute of Health, the Yale Center for Genomics and Proteomics, the Yale/NIDA Neuroproteomics Center and the G. Harold and Leila Y. Mathers Charitable Foundation.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
Reprints and permissions information is available at www.nature.com/reprints. The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Di Paolo, G., De Camilli, P. Phosphoinositides in cell regulation and membrane dynamics. Nature 443, 651–657 (2006). https://doi.org/10.1038/nature05185
Issue Date:
DOI: https://doi.org/10.1038/nature05185
This article is cited by
-
Rab GTPases and phosphoinositides fine-tune SNAREs dependent targeting specificity of intracellular vesicle traffic
Nature Communications (2024)
-
Lipid kinase PIP5Kα contributes to Hippo pathway activation via interaction with Merlin and by mediating plasma membrane targeting of LATS1
Cell Communication and Signaling (2023)
-
Metabolism, metabolites, and macrophages in cancer
Journal of Hematology & Oncology (2023)
-
Spatiotemporal expression patterns of genes coding for plasmalemmal chloride transporters and channels in neurological diseases
Molecular Brain (2023)
-
Long-term depression in neurons involves temporal and ultra-structural dynamics of phosphatidylinositol-4,5-bisphosphate relying on PIP5K, PTEN and PLC
Communications Biology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.