Abstract
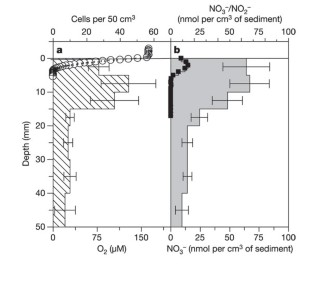
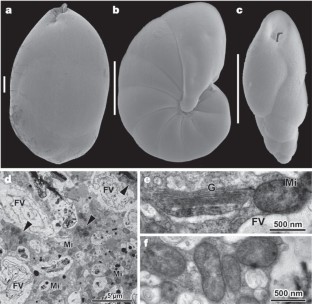
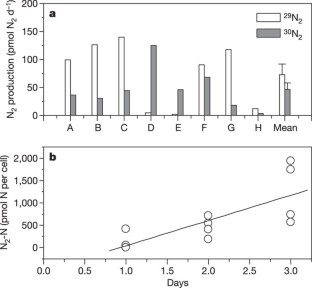
Benthic foraminifera are unicellular eukaryotes found abundantly in many types of marine sediments. Many species survive and possibly reproduce in anoxic habitats1, but sustainable anaerobic metabolism has not been previously described. Here we demonstrate that the foraminifer Globobulimina pseudospinescens accumulates intracellular nitrate stores and that these can be respired to dinitrogen gas. The amounts of nitrate detected are estimated to be sufficient to support respiration for over a month. In a Swedish fjord sediment where G. pseudospinescens is the dominant foraminifer, the intracellular nitrate pool in this species accounted for 20% of the large, cell-bound, nitrate pool present in an oxygen-free zone. Similarly high nitrate concentrations were also detected in foraminifera Nonionella cf. stella and a Stainforthia species, the two dominant benthic taxa occurring within the oxygen minimum zone of the continental shelf off Chile. Given the high abundance of foraminifera in anoxic marine environments1,2,3, these new findings suggest that foraminifera may play an important role in global nitrogen cycling and indicate that our understanding of the complexity of the marine nitrogen cycle is far from complete.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Bernhard, J. M. Microaerophilic and facultative anaerobic benthic foraminifera: a review of experimental and ultrastructural evidence. Rev. de Paléobiol 15, 261–275 (1996)
Gooday, A. J., Bernhard, J. M., Levin, L. A. & Suhr, S. B. Foraminifera in the Arabian Sea oxygen minimum zone and other oxygen-deficient settings: taxonomic composition, diversity, and relation to metazoan faunas. Deep-sea Res. II 47, 25–54 (2000)
Levin, L. et al. Benthic processes on the Peru margin: a transect across the oxygen minimum zone during the 1997–98 El Nino. Prog. Oceanogr. 53, 1–27 (2002)
Fontanier, C. et al. Seasonal and interannual variability of benthic foraminiferal faunas at 550 m depth in the Bay of Biscay. Deep-sea Res. I 50, 457–494 (2003)
Alberts, B., et al. Molecular Biology of the Cell (Garland Science, New York, 2002)
Bernhard, J. M. & Reimers, C. E. Benthic foraminiferal population fluctuations related to anoxia: Santa Barbara Basin. Biogeochemistry 15, 127–149 (1991)
Dalsgaard, T., Canfield, D. E., Petersen, J., Thamdrup, B. & Acuña-González, J. N2 production by the anammox reaction in the anoxic water column of Golfo Dulce, Costa Rica. Nature 422, 606–608 (2003)
Chen, F., Xia, Q. & Ju, L. K. Aerobic denitrification of Pseudomonas aeruginosa monitored by online NAD(P)H fluorescence. Appl. Environ. Microbiol. 69, 6715–6722 (2003)
Finlay, B. J., Span, A. S. W. & Harman, J. M. P. Nitrate respiration in primitive eukaryotes. Nature 303, 333–336 (1983)
Tielens, A. G. M., Rotte, C., van Hellemond, J. J. & Martin, W. Mitochondria as we don't know them. Trends Biochem. Sci. 27, 564–572 (2002)
Geslin, E., Heinz, P., Jorissen, F. & Hemleben, C. Migratory responses of deep-sea benthic foraminifera to variable oxygen conditions: laboratory investigations. Mar. Micropaleontol. 53, 227–243 (2004)
Zopfi, J., Kjaer, T., Nielsen, L. P. & Jorgensen, B. B. Ecology of Thioploca spp.: nitrate and sulfur storage in relation to chemical microgradients and influence of Thioploca spp. on the sedimentary nitrogen cycle. Appl. Environ. Microbiol. 67, 5530–5537 (2001)
Engström, P., Dalsgaard, T., Hulth, S. & Aller, R. C. Anaerobic ammonium oxidation by nitrite (anammox): Implications for N2 production in coastal marine sediments. Geochim. Cosmochim. Acta 69, 2057–2065 (2005)
Huettel, M., Forster, S., Klöser, S. & Fossing, H. Vertical migration in the sediment-dwelling sulfur bacteria Thioploca spp. in overcoming diffusion limitations. Appl. Environ. Microbiol. 62, 1863–1872 (1996)
Larsen, L. H., Kjaer, T. & Revsbech, N. P. A microscale NO-3 biosensor for environmental applications. Anal. Chem. 69, 3527–3531 (1997)
Revsbech, N. P. An oxygen microelectrode with a guard cathode. Limnol. Oceanogr. 34, 474–478 (1989)
Meyer, R. L., Kjaer, T. & Revsbech, N. P. Use of NOx- microsensors to estimate the activity of sediment nitrification and NOx- consumption along an estuarine salinity, nitrate, and light gradient. Aquat. Microb. Ecol. 26, 181–193 (2001)
Sayama, M. Presence of nitrate-accumulating sulfur bacteria and their influence on nitrogen cycling in a shallow coastal marine sediment. Appl. Environ. Microbiol. 67, 3481–3487 (2001)
Braman, R. S. & Hendrix, S. A. Nanogram nitrite and nitrate determination in environmental and biological-materials by vanadium(III) reduction with chemi-luminescence detection. Anal. Chem. 61, 2715–2718 (1989)
Kuypers, M. M. M. et al. Anaerobic ammonium oxidation by anammox bacteria in the Black Sea. Nature 422, 608–611 (2003)
Raghoebarsing, A. A. et al. Methanotrophic symbionts provide carbon for photosynthesis in peat bogs. Nature 436, 1153–1156 (2005)
Risgaard-Petersen, N. & Rysgaard, S. in Methods in Applied Soil Microbiology and Biochemistry (eds Alef, K. & Nannipieri, P.) 287–296 (Academic, London, 1995)
Risgaard-Petersen, N., Rysgaard, S. & Revsbech, N. P. A sensitive assay for determination of 14N/15N isotope distribution in NO3-. J. Microbiol. Meth. 17, 155–164 (1993)
Vester, F. & Ingvorsen, K. Improved most-probable-number method to detect sulfate-reducing bacteria with natural media and a radiotracer. Appl. Environ. Microbiol. 64, 1700–1707 (1998)
Bernhard, J. M. Distinguishing live from dead foraminifera: methods review and proper applications. Micropaleontology 46, 38–46 (2000)
Nielsen, L. P. Denitrification in sediments determined from nitrogen isotope pairing. FEMS Microbiol. Ecol. 86, 357–362 (1992)
Acknowledgements
We thank P. Engström, J. Brandsma, V. A. Gallardo, B. B. Jørgensen, E. Frandsen, G. Ittman, S. Petersen, K. L. Knudsen, G. J. Janssen, P. Sørensen and L. Pierson for their support. The work was supported by the Carlsberg Foundation, Denmark (N.R.-P.); the National Science Research Foundation, Denmark (N.R.-P., T.C. and N.P.R.); the Netherlands Organization for Scientific Research (NWO-ALW biogeosphere; G.J.v.d.Z. and M.S.M.J.) and the Swedish Research Council (S.P.E.). Author Contributions A.M.L., M.C.S., N.R.-P., S.I., L.P.N. and N.P.R. performed the sampling. A.M.L, G.J.v.d.Z and T.C. carried out the foraminifer identification. N.R.-P., N.P.R., E.P.-O., S.I., L.P.N. and A.M.L. performed the nitrate and denitrification rate measurements; J.W.M.D., H.J.M.O.d.C., T.C. and M.C.S. performed the microscopy. The research was conceived by N.R.-P., S.P.E., G.J.v.d.Z., M.S.M.J., H.J.M.O.d.C., A.M.L., N.P.R, L.P.N. and S.I. All authors contributed to interpreting the data and writing the paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at www.nature.com/reprints. The authors declare no competing financial interests.
Supplementary information
Supplementary Methods
This file contains methodology used for measuring denitrification in a foraminifer and the procedure used for calculation of the denitrification activity for a bacterial cell. There is also one figure. (PDF 59 kb)
Rights and permissions
About this article
Cite this article
Risgaard-Petersen, N., Langezaal, A., Ingvardsen, S. et al. Evidence for complete denitrification in a benthic foraminifer. Nature 443, 93–96 (2006). https://doi.org/10.1038/nature05070
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature05070
This article is cited by
-
A deep-learning automated image recognition method for measuring pore patterns in closely related bolivinids and calibration for quantitative nitrate paleo-reconstructions
Scientific Reports (2023)
-
The Peruvian oxygen minimum zone was similar in extent but weaker during the Last Glacial Maximum than Late Holocene
Communications Earth & Environment (2022)
-
Kleptoplast distribution, photosynthetic efficiency and sequestration mechanisms in intertidal benthic foraminifera
The ISME Journal (2022)
-
Diversified crop rotation improves continuous monocropping eggplant production by altering the soil microbial community and biochemical properties
Plant and Soil (2022)
-
Intracellular nitrate storage by diatoms can be an important nitrogen pool in freshwater and marine ecosystems
Communications Earth & Environment (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.