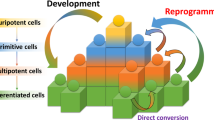
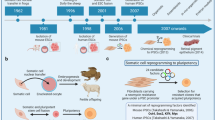
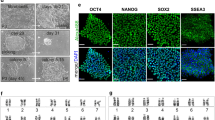
Abstract
The cloning of mammals from differentiated donor cells has refuted the old dogma that development is an irreversible process. It has demonstrated that the oocyte can reprogramme an adult nucleus into an embryonic state that can direct development of a new organism. The prospect of deriving patient-specific embryonic stem cells by nuclear transfer underscores the potential use of this technology in regenerative medicine. The future challenge will be to study alternatives to nuclear transfer in order to recapitulate reprogramming in a Petri dish without the use of oocytes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Gurdon, J. B. & Byrne, J. A. The first half-century of nuclear transplantation. Proc. Natl Acad. Sci. USA 100, 8048–8052 (2003).
Wakayama, T., Perry, A. C., Zuccotti, M., Johnson, K. R. & Yanagimachi, R. Full-term development of mice from enucleated oocytes injected with cumulus cell nuclei. Nature 394, 369–374 (1998).
Wilmut, I., Schnieke, A. E., McWhir, J., Kind, A. J. & Campbell, K. H. Viable offspring derived from fetal and adult mammalian cells. Nature 385, 810–813 (1997).
Hochedlinger, K. et al. Reprogramming of a melanoma genome by nuclear transplantation. Genes Dev. 18, 1875–1885 (2004).
Hochedlinger, K. & Jaenisch, R. Monoclonal mice generated by nuclear transfer from mature B and T donor cells. Nature 415, 1035–1038 (2002).
Eggan, K. et al. Mice cloned from olfactory sensory neurons. Nature 428, 44–49 (2004).
Li, L., Connelly, M. C., Wetmore, C., Curran, T. & Morgan, J. I. Mouse embryos cloned from brain tumors. Cancer Res. 63, 2733–2736 (2003).
Li, J., Ishii, T., Feinstein, P. & Mombaerts, P. Odorant receptor gene choice is reset by nuclear transfer from mouse olfactory sensory neurons. Nature 428, 393–399 (2004).
Rideout, W. M., Hochedlinger, K., Kyba, M., Daley, G. Q. & Jaenisch, R. Correction of a genetic defect by nuclear transplantation and combined cell and gene therapy. Cell 109, 17–27 (2002).
Tamashiro, K. L. et al. Cloned mice have an obese phenotype not transmitted to their offspring. Nature Med. 8, 262–267 (2002).
Ogonuki, N. et al. Early death of mice cloned from somatic cells. Nature Genet. 30, 253–254 (2002).
Briggs, R. & King, T. J. Changes in the nuclei of differentiating endoderm cells as revealed by nuclear transplantation. J. Morphol. 100, 269–311 (1957).
Cheong, H. T., Takahashi, Y. & Kanagawa, H. Birth of mice after transplantation of early cell-cycle-stage embryonic nuclei into enucleated oocytes. Biol. Reprod. 48, 958–963 (1993).
Hiiragi, T. & Solter, D. Reprogramming is essential in nuclear transfer. Mol. Reprod. Dev. 70, 417–421 (2005).
Eggan, K. et al. Hybrid vigor, fetal overgrowth, and viability of mice derived by nuclear cloning and tetraploid embryo complementation. Proc. Natl Acad. Sci. USA 98, 6209–6214 (2001).
Rideout, W. M. et al. Generation of mice from wild-type and targeted ES cells by nuclear cloning. Nature Genet. 24, 109–110 (2000).
Wakayama, T. & Yanagimachi, R. Cloning of male mice from adult tail-tip cells. Nature Genet. 22, 127–128 (1999).
Ogura, A. et al. Production of male cloned mice from fresh, cultured, and cryopreserved immature Sertoli cells. Biol. Reprod. 62, 1579–1584 (2000).
Inoue, K. et al. Generation of cloned mice by direct nuclear transfer from natural killer T cells. Curr. Biol. 15, 1114–1118 (2005).
Blelloch, R. et al. Reprogramming efficiency following somatic cell nuclear transfer is influenced by the differentiation and methylation state of the donor nucleus. Stem Cells published online 18 May 2006 (doi:10.1634/stemcells.2006-0050).
Inoue, K. et al. Inefficient reprogramming of the haematopoietic stem cell genome following nuclear transfer. J. Cell Sci. 119, 1985–1991 (2006).
Ng, R. K. & Gurdon, J. B. Epigenetic memory of active gene transcription is inherited through somatic cell nuclear transfer. Proc. Natl Acad. Sci. USA 102, 1957–1962 (2005).
Kohda, T. et al. Variation in gene expression and aberrantly regulated chromosome regions in cloned mice. Biol. Reprod. 73, 1302–1311 (2005).
Humpherys, D. et al. Abnormal gene expression in cloned mice derived from embryonic stem cell and cumulus cell nuclei. Proc. Natl Acad. Sci. USA 99, 12889–12894 (2002).
Jaenisch, R. Human cloning — the science and ethics of nuclear transplantation. N. Engl. J. Med. 351, 2787–2791 (2004).
Munsie, M. J. et al. Isolation of pluripotent embryonic stem cells from reprogrammed adult mouse somatic cell nuclei. Curr. Biol. 10, 989–992 (2000).
Brambrink, T., Hochedlinger, K., Bell, G. & Jaenisch, R. ES cells derived from cloned and fertilized blastocysts are transcriptionally and functionally indistinguishable. Proc. Natl Acad. Sci. USA 103, 933–938 (2006).
Cibelli, J. B. et al. Somatic cell nuclear transfer in humans: pronuclear and early embryonic development. J. Regen. Med. 2, 25–31 (2001).
Chen, Y. et al. Embryonic stem cells generated by nuclear transfer of human somatic nuclei into rabbit oocytes. Cell Res. 13, 251–263 (2003).
Dey, R., Barrientos, A. & Moraes, C. T. Functional constraints of nuclear–mitochondrial DNA interactions in xenomitochondrial rodent cell lines. J. Biol. Chem. 275, 31520–31527 (2000).
Simerly, C. et al. Molecular correlates of primate nuclear transfer failures. Science 300, 297 (2003).
Simerly, C. et al. Embryogenesis and blastocyst development after somatic cell nuclear transfer in nonhuman primates: overcoming defects caused by meiotic spindle extraction. Dev. Biol. 276, 237–252 (2004).
Meng, L., Ely, J. J., Stouffer, R. L. & Wolf, D. P. Rhesus monkeys produced by nuclear transfer. Biol. Reprod. 57, 454–459 (1997).
Blau, H. M. & Blakely, B. T. Plasticity of cell fate: insights from heterokaryons. Semin. Cell Dev. Biol. 10, 267–272 (1999).
Miller, R. A. & Ruddle, F. H. Pluripotent teratocarcinoma–thymus somatic cell hybrids. Cell 9, 45–55 (1976).
Tada, M. et al. Pluripotency of reprogrammed somatic genomes in embryonic stem hybrid cells. Dev. Dyn. 227, 504–510 (2003).
Tada, M., Tada, T., Lefebvre, L., Barton, S. C. & Surani, M. A. Embryonic germ cells induce epigenetic reprogramming of somatic nucleus in hybrid cells. EMBO J. 16, 6510–6520 (1997).
Tada, M., Takahama, Y., Abe, K., Nakatsuji, N. & Tada, T. Nuclear reprogramming of somatic cells by in vitro hybridization with ES cells. Curr. Biol. 11, 1553–1558 (2001).
Cowan, C. A., Atienza, J., Melton, D. A. & Eggan, K. Nuclear reprogramming of somatic cells after fusion with human embryonic stem cells. Science 309, 1369–1373 (2005).
Yu, J., Vodyanik, M. A., He, P., Slukvin, I. I. & Thomson, J. A. Human embryonic stem cells reprogram myeloid precursors following cell–cell fusion. Stem Cells 24, 168–176 (2005).
Rousset, J. P., Bucchini, D. & Jami, J. Hybrids between F9 nullipotent teratocarcinoma and thymus cells produce multidifferentiated tumors in mice. Dev. Biol. 96, 331–336 (1983).
Oshima, R. G., McKerrow, J. & Cox, D. Murine embryonal carcinoma hybrids: decreased ability to spontaneously differentiate as a dominant trait. J. Cell Physiol. 109, 195–204 (1981).
Do, J. T. & Scholer, H. R. Nuclei of embryonic stem cells reprogram somatic cells. Stem Cells 22, 941–949 (2004).
Byrne, J. A., Simonsson, S., Western, P. S. & Gurdon, J. B. Nuclei of adult mammalian somatic cells are directly reprogrammed to oct-4 stem cell gene expression by amphibian oocytes. Curr. Biol. 13, 1206–1213 (2003).
Simonsson, S. & Gurdon, J. DNA demethylation is necessary for the epigenetic reprogramming of somatic cell nuclei. Nature Cell Biol. 6, 984–990 (2004).
Kikyo, N., Wade, P. A., Guschin, D., Ge, H. & Wolffe, A. P. Active remodeling of somatic nuclei in egg cytoplasm by the nucleosomal ATPase ISWI. Science 289, 2360–2362 (2000).
Hansis, C., Barreto, G., Maltry, N. & Niehrs, C. Nuclear reprogramming of human somatic cells by Xenopus egg extract requires BRG1. Curr. Biol. 14, 1475–1480 (2004).
Lemaitre, J. M., Danis, E., Pasero, P., Vassetzky, Y. & Mechali, M. Mitotic remodeling of the replicon and chromosome structure. Cell 123, 787–801 (2005).
Gurdon, J. The developmental capacity of nuclei taken from intestinal epithelium cells of feeding tadpoles. J. Embryol. Exp. Morphol. 10, 622–640 (1962).
Taranger, C. K. et al. Induction of dedifferentiation, genomewide transcriptional programming, and epigenetic reprogramming by extracts of carcinoma and embryonic stem cells. Mol. Biol. Cell 16, 5719–5735 (2005).
Hakelien, A. M., Landsverk, H. B., Robl, J. M., Skalhegg, B. S. & Collas, P. Reprogramming fibroblasts to express T-cell functions using cell extracts. Nature Biotechnol. 20, 460–466 (2002).
Raff, M. Adult stem cell plasticity: fact or artifact? Annu. Rev. Cell Dev. Biol. 19, 1–22 (2003).
Andrews, P. W. From teratocarcinomas to embryonic stem cells. Phil. Trans. R. Soc. Lond. B 357, 405–417 (2002).
Evans, M. J. & Kaufman, M. H. Establishment in culture of pluripotential cells from mouse embryos. Nature 292, 154–156 (1981).
Martin, G. R. Isolation of a pluripotent cell line from early mouse embryos cultured in medium conditioned by teratocarcinoma stem cells. Proc. Natl Acad. Sci. USA 78, 7634–7638 (1981).
Matsui, Y., Zsebo, K. & Hogan, B. L. Derivation of pluripotential embryonic stem cells from murine primordial germ cells in culture. Cell 70, 841–847 (1992).
Resnick, J. L., Bixler, L. S., Cheng, L. & Donovan, P. J. Long-term proliferation of mouse primordial germ cells in culture. Nature 359, 550–551 (1992).
Labosky, P. A., Barlow, D. P. & Hogan, B. L. Mouse embryonic germ (EG) cell lines: transmission through the germline and differences in the methylation imprint of insulin-like growth factor 2 receptor (Igf2r) gene compared with embryonic stem (ES) cell lines. Development 120, 3197–3204 (1994).
Tada, T. et al. Epigenotype switching of imprintable loci in embryonic germ cells. Dev. Genes Evol. 207, 551–561 (1998).
Rossant, J. & McBurney, M. W. The developmental potential of a euploid male teratocarcinoma cell line after blastocyst injection. J. Embryol. Exp. Morphol. 70, 99–112 (1982).
Blelloch, R. H. et al. Nuclear cloning of embryonal carcinoma cells. Proc. Natl Acad. Sci. USA 101, 13985–13990 (2004).
Stewart, C. L., Gadi, I. & Bhatt, H. Stem cells from primordial germ cells can reenter the germ line. Dev. Biol. 161, 626–628 (1994).
Stewart, T. A. & Mintz, B. Recurrent germ-line transmission of the teratocarcinoma genome from the METT-1 culture line to progeny in vivo. J. Exp. Zool. 224, 465–469 (1982).
Durcova-Hills, G., Adams, I. R., Barton, S. C., Surani, M. A. & McLaren, A. The role of exogenous FGF-2 on the reprogramming of primordial germ cells into pluripotent stem cells. Stem Cells (in the press).
Kehler, J. et al. Oct4 is required for primordial germ cell survival. EMBO Rep. 5, 1078–1083 (2004).
Niwa, H., Miyazaki, J. & Smith, A. G. Quantitative expression of Oct-3/4 defines differentiation, dedifferentiation or self-renewal of ES cells. Nature Genet. 24, 372–376 (2000).
Chung, Y. et al. Embryonic and extraembryonic stem cell lines derived from single mouse blastomeres. Nature 439, 216–219 (2005).
Kanatsu-Shinohara, M. et al. Generation of pluripotent stem cells from neonatal mouse testis. Cell 119, 1001–1012 (2004).
Guan, K. et al. Pluripotency of spermatogonial stem cells from adult mouse testis. Nature 440, 1199–1203 (2006).
Zwaka, T. P. & Thomson, J. A. A germ cell origin of embryonic stem cells? Development 132, 227–233 (2005).
Jiang, Y. et al. Pluripotency of mesenchymal stem cells derived from adult marrow. Nature 418, 41–49 (2002).
Kogler, G. et al. A new human somatic stem cell from placental cord blood with intrinsic pluripotent differentiation potential. J. Exp. Med. 200, 123–135 (2004).
Snow, M. H. L. Gastrulation in the mouse: growth and regionalization of the epiblast. J. Embryol. Exp. Morphol. 42, 293–303 (1977).
Yamazaki, Y. et al. Adult mice cloned from migrating primordial germ cells. Proc. Natl Acad. Sci. USA 102, 11361–11366 (2005).
Hajkova, P. et al. Epigenetic reprogramming in mouse primordial germ cells. Mech. Dev. 117, 15–23 (2002).
Lee, J. et al. Erasing genomic imprinting memory in mouse clone embryos produced from day 11.5 primordial germ cells. Development 129, 1807–1817 (2002).
Hernandez, L., Kozlov, S., Piras, G. & Stewart, C. L. Paternal and maternal genomes confer opposite effects on proliferation, cell-cycle length, senescence, and tumor formation. Proc. Natl Acad. Sci. USA 100, 13344–13349 (2003).
Holm, T. M. et al. Global loss of imprinting leads to widespread tumorigenesis in adult mice. Cancer Cell 8, 275–285 (2005).
Chambers, I. & Smith, A. Self-renewal of teratocarcinoma and embryonic stem cells. Oncogene 23, 7150–7160 (2004).
Li, X., Kato, Y. & Tsunoda, Y. Comparative analysis of development-related gene expression in mouse preimplantation embryos with different developmental potential. Mol. Reprod. Dev. 72, 152–160 (2005).
Bortvin, A. et al. Incomplete reactivation of Oct4-related genes in mouse embryos cloned from somatic nuclei. Development 130, 1673–1680 (2003).
Boiani, M., Eckardt, S., Scholer, H. R. & McLaughlin, K. J. Oct4 distribution and level in mouse clones: consequences for pluripotency. Genes Dev. 16, 1209–1219 (2002).
Boyer, L. A. et al. Core transcriptional regulatory circuitry in human embryonic stem cells. Cell 122, 947–956 (2005).
Loh, Y. H. et al. The Oct4 and Nanog transcription network regulates pluripotency in mouse embryonic stem cells. Nature Genet. 38, 431–440 (2006).
Ringrose, L. & Paro, R. Epigenetic regulation of cellular memory by the Polycomb and Trithorax group proteins. Annu. Rev. Genet. 38, 413–443 (2004).
Lee, T. I. et al. Control of developmental regulators by polycomb in human embryonic stem cells. Cell 125, 301–313 (2006).
Boyer, L. A. et al. Polycomb complexes repress developmental regulators in murine embryonic stem cells. Nature 441, 349–353 (2006).
Bernstein, B. E. et al. Epigenetic landscape in embryonic stem cells. Cell 125, 315–326 (2006).
Xie, H., Ye, M., Feng, R. & Graf, T. Stepwise reprogramming of B cells into macrophages. Cell 117, 663–676 (2004).
Nutt, S. L., Heavey, B., Rolink, A. G. & Busslinger, M. Commitment to the B-lymphoid lineage depends on the transcription factor Pax5. Nature 401, 556–562 (1999).
Baba, Y., Garrett, K. P. & Kincade, P. W. Constitutively active β-catenin confers multilineage differentiation potential on lymphoid and myeloid progenitors. Immunity 23, 599–609 (2005).
Kondo, T. & Raff, M. Oligodendrocyte precursor cells reprogrammed to become multipotential CNS stem cells. Science 289, 1754–1757 (2000).
Bachoo, R. M. et al. Epidermal growth factor receptor and Ink4a/Arf: convergent mechanisms governing terminal differentiation and transformation along the neural stem cell to astrocyte axis. Cancer Cell 1, 269–277 (2002).
Hochedlinger, K., Yamada, Y., Beard, C. & Jaenisch, R. Ectopic expression of Oct-4 blocks progenitor-cell differentiation and causes dysplasia in epithelial tissues. Cell 121, 465–477 (2005).
Chambers, I. et al. Functional expression cloning of Nanog, a pluripotency sustaining factor in embryonic stem cells. Cell 113, 643–655 (2003).
Li, E., Bestor, T. H. & Jaenisch, R. Targeted mutation of the DNA methyltransferase gene results in embryonic lethality. Cell 69, 915–926 (1992).
Wakayama, S. et al. Establishment of male and female nuclear transfer embryonic stem cell lines from different mouse strains and tissues. Biol. Reprod. 72, 932–936 (2005).
Kennedy, D. Editorial retraction. Science 311, 335 (2006).
Hubner, K. et al. Derivation of oocytes from mouse embryonic stem cells. Science 300, 1251–1256 (2003).
Lacham-Kaplan, O., Chy, H. & Trounson, A. Testicular cell conditioned medium supports differentiation of embryonic stem cells into ovarian structures containing oocytes. Stem Cells 24, 266–273 (2006).
Acknowledgements
We would like to thank B. Hogan, L. Looijenga, A. Wutz, N. Geijsen, M. W. Lensch and members of the Jaenisch laboratory for discussion and critical reading of the manuscript. Support to K.H. came from a Genzyme postdoctoral fellowship. R.J. is supported by grants from the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Author Information Reprints and permissions information is available at npg.nature.com/reprintsandpermissions.
Rights and permissions
About this article
Cite this article
Hochedlinger, K., Jaenisch, R. Nuclear reprogramming and pluripotency. Nature 441, 1061–1067 (2006). https://doi.org/10.1038/nature04955
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature04955
This article is cited by
-
MYC transcription activation mediated by OCT4 as a mechanism of resistance to 13-cisRA-mediated differentiation in neuroblastoma
Cell Death & Disease (2020)
-
Effects of Different Concentrations of Reversine on Plasticity of Mesenchymal Stem Cells
Indian Journal of Clinical Biochemistry (2020)
-
Induced pluripotent stem cells (iPSCs) as model to study inherited defects of neurotransmission in inborn errors of metabolism
Journal of Inherited Metabolic Disease (2018)
-
Integrated analysis of hematopoietic differentiation outcomes and molecular characterization reveals unbiased differentiation capacity and minor transcriptional memory in HPC/HSC-iPSCs
Stem Cell Research & Therapy (2017)
-
Genome-wide gene expression analyses reveal unique cellular characteristics related to the amenability of HPC/HSCs into high-quality induced pluripotent stem cells
Stem Cell Research & Therapy (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.