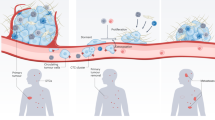
Abstract
Approximately 90% of all cancer deaths arise from the metastatic spread of primary tumours. Of all the processes involved in carcinogenesis, local invasion and the formation of metastases are clinically the most relevant, but they are the least well understood at the molecular level. Revealing their mechanisms is one of the main challenges for exploratory and applied cancer research. Recent experimental progress has identified a number of molecular pathways and cellular mechanisms that underlie the multistage process of metastasis formation: these include tumour invasion, tumour-cell dissemination through the bloodstream or the lymphatic system, colonization of distant organs and, finally, fatal outgrowth of metastases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Grunert, S., Jechlinger, M. & Beug, H. Diverse cellular and molecular mechanisms contribute to epithelial plasticity and metastasis. Nature Rev. Mol. Cell Biol. 4, 657–665 (2003).
Thiery, J. P. Epithelial–mesenchymal transitions in tumour progression. Nature Rev. Cancer 2, 442–454 (2002).
Huber, M. A., Kraut, N. & Beug, H. Molecular requirements for epithelial–mesenchymal transition during tumor progression. Curr. Opin. Cell Biol. 17, 548–558 (2005).
Siegel, P. M. & Massague, J. Cytostatic and apoptotic actions of TGF-β in homeostasis and cancer. Nature Rev. Cancer 3, 807–821 (2003).
Trusolino, L. & Comoglio, P. M. Scatter-factor and semaphorin receptors: cell signalling for invasive growth. Nature Rev. Cancer 2, 289–300 (2002).
Corso, S., Comoglio, P. M. & Giordano, S. Cancer therapy: can the challenge be MET? Trends Mol. Med. 11, 284–292 (2005).
Birchmeier, C., Birchmeier, W., Gherardi, E. & Vande Woude, G. F. Met, metastasis, motility and more. Nature Rev. Mol. Cell Biol. 4, 915–925 (2003).
Boccaccio, C. et al. The MET oncogene drives a genetic programme linking cancer to haemostasis. Nature 434, 396–400 (2005).
Trusolino, L., Bertotti, A. & Comoglio, P. M. A signaling adapter function for α6β4 integrin in the control of HGF-dependent invasive growth. Cell 107, 643–654 (2001).
Conrotto, P., Corso, S., Gamberini, S., Comoglio, P. M. & Giordano, S. Interplay between scatter factor receptors and B plexins controls invasive growth. Oncogene 23, 5131–5137 (2004).
Giordano, S. et al. The semaphorin 4D receptor controls invasive growth by coupling with Met. Nature Cell Biol. 4, 720–724 (2002).
Stella, M. C., Trusolino, L., Pennacchietti, S. & Comoglio, P. M. Negative feedback regulation of Met-dependent invasive growth by Notch. Mol. Cell Biol. 25, 3982–3996 (2005).
Michieli, P. et al. Targeting the tumor and its microenvironment by a dual-function decoy Met receptor. Cancer Cell 6, 61–73 (2004).
Yang, Y. A. et al. Lifetime exposure to a soluble TGF-β antagonist protects mice against metastasis without adverse side effects. J. Clin. Invest. 109, 1607–1615 (2002).
Douma, S. et al. Suppression of anoikis and induction of metastasis by the neurotrophic receptor TrkB. Nature 430, 1034–1039 (2004).
Lopez, T. & Hanahan, D. Elevated levels of IGF-1 receptor convey invasive and metastatic capability in a mouse model of pancreatic islet tumorigenesis. Cancer Cell 1, 339–353 (2002).
Mitsiades, C. S. et al. Inhibition of the insulin-like growth factor receptor-1 tyrosine kinase activity as a therapeutic strategy for multiple myeloma, other hematologic malignancies, and solid tumors. Cancer Cell 5, 221–230 (2004).
Garcia-Echeverria, C. et al. In vivo antitumor activity of NVP-AEW541 — A novel, potent, and selective inhibitor of the IGF-IR kinase. Cancer Cell 5, 231–239 (2004).
Marone, R. et al. Memo mediates ErbB2-driven cell motility. Nature Cell Biol. 6, 515–522 (2004).
Karhadkar, S. S. et al. Hedgehog signalling in prostate regeneration, neoplasia and metastasis. Nature 431, 707–712 (2004).
Pennacchietti, S. et al. Hypoxia promotes invasive growth by transcriptional activation of the met protooncogene. Cancer Cell 3, 347–361 (2003).
Matteucci, E., Locati, M. & Desiderio, M. A. Hepatocyte growth factor enhances CXCR4 expression favoring breast cancer cell invasiveness. Exp. Cell Res. 310, 176–185 (2005).
Phillips, R. J. et al. Epidermal growth factor and hypoxia-induced expression of CXC chemokine receptor 4 on non-small cell lung cancer cells is regulated by the phosphatidylinositol 3-kinase/PTEN/AKT/mammalian target of rapamycin signaling pathway and activation of hypoxia inducible factor-1α. J. Biol. Chem. 280, 22473–22481 (2005).
Cavallaro, U. & Christofori, G. Cell adhesion and signalling by cadherins and Ig-CAMs in cancer. Nature Rev. Cancer 4, 118–132 (2004).
Perl, A. K., Wilgenbus, P., Dahl, U., Semb, H. & Christofori, G. A causal role for E-cadherin in the transition from adenoma to carcinoma. Nature 392, 190–193 (1998).
De Craene, B., van Roy, F. & Berx, G. Unraveling signalling cascades for the Snail family of transcription factors. Cell Signal. 17, 535–547 (2005).
Kang, Y. & Massague, J. Epithelial–mesenchymal transitions: twist in development and metastasis. Cell 118, 277–279 (2004).
Peinado, H., Ballestar, E., Esteller, M. & Cano, A. Snail mediates E-cadherin repression by the recruitment of the Sin3A/histone deacetylase 1 (HDAC1)/HDAC2 complex. Mol. Cell. Biol. 24, 306–319 (2004).
Strathdee, G. Epigenetic versus genetic alterations in the inactivation of E-cadherin. Semin. Cancer Biol. 12, 373–379 (2002).
Yook, J. I., Li, X. Y., Ota, I., Fearon, E. R. & Weiss, S. J. Wnt-dependent regulation of the E-cadherin repressor snail. J. Biol. Chem. 280, 11740–11748 (2005).
Lu, Z., Ghosh, S., Wang, Z. & Hunter, T. Downregulation of caveolin-1 function by EGF leads to the loss of E-cadherin, increased transcriptional activity of β-catenin, and enhanced tumor cell invasion. Cancer Cell 4, 499–515 (2003).
Zhou, B. P. et al. Dual regulation of Snail by GSK-3β-mediated phosphorylation in control of epithelial–mesenchymal transition. Nature Cell Biol. 6, 931–940 (2004).
Bachelder, R. E., Yoon, S. O., Franci, C., de Herreros, A. G. & Mercurio, A. M. Glycogen synthase kinase-3 is an endogenous inhibitor of Snail transcription: implications for the epithelial–mesenchymal transition. J. Cell Biol. 168, 29–33 (2005).
Peinado, H. et al. A molecular role for lysyl oxidase-like 2 enzyme in Snail regulation and tumor progression. EMBO J. 24, 3446–3458 (2005).
Fujita, Y. et al. Hakai, a c-Cbl-like protein, ubiquitinates and induces endocytosis of the E-cadherin complex. Nature Cell Biol. 4, 222–231 (2002).
Gavert, N. et al. L1, a novel target of β-catenin signaling, transforms cells and is expressed at the invasive front of colon cancers. J. Cell Biol. 168, 633–642 (2005).
Conacci-Sorrell, M. E. et al. Nr-CAM is a target gene of the β-catenin/LEF-1 pathway in melanoma and colon cancer and its expression enhances motility and confers tumorigenesis. Genes Dev. 16, 2058–2072 (2002).
Perl, A. K. et al. Reduced expression of neural cell adhesion molecule induces metastatic dissemination of pancreatic β tumor cells. Nature Med. 5, 286–291 (1999).
Cavallaro, U., Niedermeyer, J., Fuxa, M. & Christofori, G. N-CAM modulates tumour-cell adhesion to matrix by inducing FGF-receptor signalling. Nature Cell Biol. 3, 650–657 (2001).
Hood, J. D. & Cheresh, D. A. Role of integrins in cell invasion and migration. Nature Rev. Cancer 2, 91–100 (2002).
Hynes, R. O. Integrins: bidirectional, allosteric signaling machines. Cell 110, 673–687 (2002).
Guo, W. & Giancotti, F. G. Integrin signalling during tumour progression. Nature Rev. Mol. Cell Biol. 5, 816–826 (2004).
Bao, S. et al. Periostin potently promotes metastatic growth of colon cancer by augmenting cell survival via the Akt/PKB pathway. Cancer Cell 5, 329–339 (2004).
Bissell, M. J. & Labarge, M. A. Context, tissue plasticity, and cancer: are tumor stem cells also regulated by the microenvironment? Cancer Cell 7, 17–23 (2005).
Hayward, S. W. et al. Malignant transformation in a nontumorigenic human prostatic epithelial cell line. Cancer Res. 61, 8135–8142 (2001).
Orimo, A. et al. Stromal fibroblasts present in invasive human breast carcinomas promote tumor growth and angiogenesis through elevated SDF-1/CXCL12 secretion. Cell 121, 335–348 (2005).
Bhowmick, N. A. et al. TGF-β signaling in fibroblasts modulates the oncogenic potential of adjacent epithelia. Science 303, 848–851 (2004).
de Visser, K. E., Korets, L. V. & Coussens, L. M. De novo carcinogenesis promoted by chronic inflammation is B lymphocyte dependent. Cancer Cell 7, 411–423 (2005).
Pollard, J. W. Tumour-educated macrophages promote tumour progression and metastasis. Nature Rev. Cancer 4, 71–78 (2004).
Joyce, J. A. Therapeutic targeting of the tumor microenvironment. Cancer Cell 7, 513–520 (2005).
Dranoff, G. Cytokines in cancer pathogenesis and cancer therapy. Nature Rev. Cancer 4, 11–22 (2004).
Coussens, L. M., Tinkle, C. L., Hanahan, D. & Werb, Z. MMP-9 supplied by bone marrow-derived cells contributes to skin carcinogenesis. Cell 103, 481–490 (2000).
Lin, E. Y., Nguyen, A. V., Russell, R. G. & Pollard, J. W. Colony-stimulating factor 1 promotes progression of mammary tumors to malignancy. J. Exp. Med. 193, 727–740 (2001).
Aharinejad, S. et al. Colony-stimulating factor-1 antisense treatment suppresses growth of human tumor xenografts in mice. Cancer Res. 62, 5317–5324 (2002).
De Palma, M. et al. Tie2 identifies a hematopoietic lineage of proangiogenic monocytes required for tumor vessel formation and a mesenchymal population of pericyte progenitors. Cancer Cell 8, 211–226 (2005).
Radisky, D. C. et al. Rac1b and reactive oxygen species mediate MMP-3-induced EMT and genomic instability. Nature 436, 123–127 (2005).
Boire, A. et al. PAR1 is a matrix metalloprotease-1 receptor that promotes invasion and tumorigenesis of breast cancer cells. Cell 120, 303–313 (2005).
Bergers, G. & Benjamin, L. E. Tumorigenesis and the angiogenic switch. Nature Rev. Cancer 3, 401–410 (2003).
Folkman, J. Role of angiogenesis in tumor growth and metastasis. Semin. Oncol. 29, 15–18 (2002).
Achen, M. G., McColl, B. K. & Stacker, S. A. Focus on lymphangiogenesis in tumor metastasis. Cancer Cell 7, 121–127 (2005).
Saharinen, P., Tammela, T., Karkkainen, M. J. & Alitalo, K. Lymphatic vasculature: development, molecular regulation and role in tumor metastasis and inflammation. Trends Immunol. 25, 387–395 (2004).
Muller, A. et al. Involvement of chemokine receptors in breast cancer metastasis. Nature 410, 50–56 (2001).
Zeelenberg, I. S., Ruuls- Van Stalle, L. & Roos, E. The chemokine receptor CXCR4 is required for outgrowth of colon carcinoma micrometastases. Cancer Res. 63, 3833–3839 (2003).
Kang, Y. et al. A multigenic program mediating breast cancer metastasis to bone. Cancer Cell 3, 537–549 (2003).
Minn, A. J. et al. Genes that mediate breast cancer metastasis to lung. Nature 436, 518–524 (2005).
Lynch, C. C. et al. MMP-7 promotes prostate cancer-induced osteolysis via the solubilization of RANKL. Cancer Cell 7, 485–496 (2005).
Brown, D. M. & Ruoslahti, E. Metadherin, a cell surface protein in breast tumors that mediates lung metastasis. Cancer Cell 5, 365–374 (2004).
Kaplan, R. N. et al. VEGFR1-positive haematopoietic bone marrow progenitors initiate the pre-metastatic niche. Nature 438, 820–827 (2005).
Chambers, A. F., Groom, A. C. & MacDonald, I. C. Dissemination and growth of cancer cells in metastatic sites. Nature Rev. Cancer 2, 563–572 (2002).
Suyama, K., Shapiro, I., Guttman, M. & Hazan, R. B. A signaling pathway leading to metastasis is controlled by N-cadherin and the FGF receptor. Cancer Cell 2, 301–314 (2002).
Marambaud, P. et al. A CBP binding transcriptional repressor produced by the PS1/epsilon-cleavage of N-cadherin is inhibited by PS1 FAD mutations. Cell 114, 635–645 (2003).
Huber, M. A. et al. NF-κB is essential for epithelial–mesenchymal transition and metastasis in a model of breast cancer progression. J. Clin. Invest. 114, 569–581 (2004).
Ozdamar, B. et al. Regulation of the polarity protein Par6 by TGFβ receptors controls epithelial cell plasticity. Science 307, 1603–1609 (2005).
Orian-Rousseau, V., Chen, L., Sleeman, J. P., Herrlich, P. & Ponta, H. CD44 is required for two consecutive steps in HGF/c-Met signaling. Genes Dev. 16, 3074–3086 (2002).
van der Voort, R. et al. Heparan sulfate-modified CD44 promotes hepatocyte growth factor/scatter factor-induced signal transduction through the receptor tyrosine kinase c-Met. J. Biol. Chem. 274, 6499–6506 (1999).
Acknowledgements
I apologize to all colleagues whose important work could not be cited due to space limitations. I am grateful to F. Lehembre for providing parts of Fig. 1. Experimental work in the author's laboratory is supported by NCCR Oncology, Swiss National Science Foundation, EU-FP6 framework programme LYMPHANGIOGENOMICS LSHG-CT-2004-50357, EU-FP6 framework programme BRECOSM LSHC-CT-2004-503224, Swiss Bridge Award, Krebsliga Beider Basel, and Roche Research Foundation.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The author declares no competing financial interests.
Additional information
Author Information Reprints and permissions information is available at npg.nature.com/reprintsandpermissions.
Rights and permissions
About this article
Cite this article
Christofori, G. New signals from the invasive front. Nature 441, 444–450 (2006). https://doi.org/10.1038/nature04872
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature04872
This article is cited by
-
Prostate cancer metastasis and health disparities: a systematic review
Prostate Cancer and Prostatic Diseases (2023)
-
The clinical significance of cyclin B1 (CCNB1) in invasive breast cancer with emphasis on its contribution to lymphovascular invasion development
Breast Cancer Research and Treatment (2023)
-
Aquaporin-mediated dysregulation of cell migration in disease states
Cellular and Molecular Life Sciences (2023)
-
CircN4BP2L2 promotes colorectal cancer growth and metastasis through regulation of the miR-340-5p/CXCR4 axis
Laboratory Investigation (2022)
-
Catulin reporter marks a heterogeneous population of invasive breast cancer cells with some demonstrating plasticity and participating in vascular mimicry
Scientific Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.