Abstract
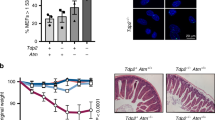
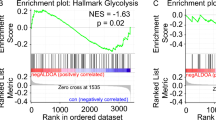
The ATM (ataxia-telangiectasia mutated) protein kinase mediates early cellular responses to DNA double-strand breaks (DSBs) generated during metabolic processes or by DNA-damaging agents1,2,3,4. ATM deficiency leads to ataxia-telangiectasia, a disease marked by lymphopenia, genomic instability and an increased predisposition to lymphoid malignancies with chromosomal translocations involving lymphocyte antigen receptor loci5,6. ATM activates cell-cycle checkpoints and can induce apoptosis in response to DNA DSBs1,2,3,4. However, defects in these pathways of the DNA damage response cannot fully account for the phenotypes of ATM deficiency. Here, we show that ATM also functions directly in the repair of chromosomal DNA DSBs by maintaining DNA ends in repair complexes generated during lymphocyte antigen receptor gene assembly. When coupled with the cell-cycle checkpoint and pro-apoptotic activities of ATM, these findings provide a molecular explanation for the increase in lymphoid tumours with translocations involving antigen receptor loci associated with ataxia-telangiectasia.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Kastan, M. B. & Lim, D. S. The many substrates and functions of ATM. Nature Rev. Mol. Cell Biol. 1, 179–186 (2000)
Pandita, T. K. A multifaceted role for ATM in genome maintenance. Expert Rev. Mol. Med. 5, 1–21 (2003)
Shiloh, Y. ATM and related protein kinases: safeguarding genome integrity. Nature Rev. Cancer 3, 155–168 (2003)
Durocher, D. & Jackson, S. P. DNA-PK, ATM and ATR as sensors of DNA damage: variations on a theme? Curr. Opin. Cell Biol. 13, 225–231 (2001)
Lavin, M. F. & Shiloh, Y. The genetic defect in ataxia-telangiectasia. Annu. Rev. Immunol. 15, 177–202 (1997)
Taylor, A. M., Metcalfe, J. A., Thick, J. & Mak, Y. F. Leukemia and lymphoma in ataxia telangiectasia. Blood 87, 423–438 (1996)
Tonegawa, S. Somatic generation of antibody diversity. Nature 302, 575–581 (1983)
Fugmann, S. D., Lee, A. I., Shockett, P. E., Villey, I. J. & Schatz, D. G. The RAG proteins and V(D)J recombination: complexes, ends, and transposition. Annu. Rev. Immunol. 18, 495–527 (2000)
Oettinger, M. A. V(D)J recombination: on the cutting edge. Curr. Opin. Cell Biol. 11, 325–329 (1999)
Gellert, M. V(D)J recombination: rag proteins, repair factors, and regulation. Annu. Rev. Biochem. 71, 101–132 (2002)
Bassing, C. H. & Alt, F. W. The cellular response to general and programmed DNA double strand breaks. DNA Repair (Amst.) 3, 781–796 (2004)
Matei, I. R., Guidos, C. J. & Danska, J. S. ATM-dependent DNA damage surveillance in T-cell development and leukemogenesis: the DSB connection. Immunol. Rev. 209, 142–158 (2006)
Perkins, E. J. et al. Sensing of intermediates in V(D)J recombination by ATM. Genes Dev. 16, 159–164 (2002)
Liao, M. J. & Van Dyke, T. Critical role for Atm in suppressing V(D)J recombination-driven thymic lymphoma. Genes Dev. 13, 1246–1250 (1999)
Petiniot, L. K. et al. Recombinase-activating gene (RAG) 2-mediated V(D)J recombination is not essential for tumorigenesis in Atm-deficient mice. Proc. Natl Acad. Sci. USA 97, 6664–6669 (2000)
Donehower, L. A. et al. Mice deficient for p53 are developmentally normal but susceptible to spontaneous tumours. Nature 356, 215–221 (1992)
Hirao, A. et al. Chk2 is a tumor suppressor that regulates apoptosis in both an ataxia telangiectasia mutated (ATM)-dependent and an ATM-independent manner. Mol. Cell. Biol. 22, 6521–6532 (2002)
Muljo, S. A. & Schlissel, M. S. A small molecule Abl kinase inhibitor induces differentiation of Abelson virus-transformed pre-B cell lines. Nature Immunol. 4, 31–37 (2003)
Liang, H. E. et al. The “dispensable” portion of RAG2 is necessary for efficient V-to-DJ rearrangement during B and T cell development. Immunity 17, 639–651 (2002)
Lewis, S. M., Hesse, J. E., Mizuuchi, K. & Gellert, M. Novel strand exchanges in V(D)J recombination. Cell 55, 1099–1107 (1988)
Morzycka-Wroblewska, E., Lee, F. E. & Desiderio, S. V. Unusual immunoglobulin gene rearrangement leads to replacement of recombinational signal sequences. Science 242, 261–263 (1988)
Hickson, I. et al. Identification and characterization of a novel and specific inhibitor of the ataxia-telangiectasia mutated kinase ATM. Cancer Res. 64, 9152–9159 (2004)
Roth, D. B., Menetski, J. P., Nakajima, P. B., Bosma, M. J. & Gellert, M. V(D)J recombination: broken DNA molecules with covalently sealed (hairpin) coding ends in scid mouse thymocytes. Cell 70, 983–991 (1992)
Franco, S. et al. H2AX prevents DNA breaks from progressing to chromosome breaks and translocations. Mol. Cell 21, 201–214 (2006)
Agrawal, A. & Schatz, D. G. RAG1 and RAG2 form a stable postcleavage synaptic complex with DNA containing signal ends in V(D)J recombination. Cell 89, 43–53 (1997)
Hiom, K. & Gellert, M. Assembly of a 12/23 paired signal complex: a critical control point in V(D)J recombination. Mol. Cell 1, 1011–1019 (1998)
Riballo, E. et al. A pathway of double-strand break rejoining dependent upon ATM, Artemis, and proteins locating to γ-H2AX foci. Mol. Cell 16, 715–724 (2004)
Sekiguchi, J. A., Whitlow, S. & Alt, F. W. Increased accumulation of hybrid V(D)J joins in cells expressing truncated versus full-length RAGs. Mol. Cell 8, 1383–1390 (2001)
Mills, K. D., Ferguson, D. O. & Alt, F. W. The role of DNA breaks in genomic instability and tumorigenesis. Immunol. Rev. 194, 77–95 (2003)
Acknowledgements
We thank M. Schlissel for pMX-RSS-GFP/IRES-hCD4. STI571 was provided by Novartis Pharmaceuticals and KU-55933 was provided by G. Smith at KuDOS Pharmaceuticals. We thank H. Virgin, K. Murphy and M. Krangel for critical review of the manuscript. This work is supported in part by grants from the National Institutes of Health (to B.P.S. and T.K.P.) and the Department of Army (T.K.P.). C.H.B. is a Pew Scholar in the Biomedical Sciences and a recipient of a Pilot Project Award from the American Cancer Society. B.P.S. is a recipient of a Research Scholar Award from the American Cancer Society. C.-Y.H. is supported by a post-doctoral training grant and A.L.B. a pre-doctoral training grant from the NIH.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Supplementary Notes
This file contains Supplementary Figures 1–19, Supplementary Methods and Supplementary Notes. The Supplementary Figures show the setup of the experimental system, data from additional cell lines, and controls for Figures 1-4. (PDF 2604 kb)
Rights and permissions
About this article
Cite this article
Bredemeyer, A., Sharma, G., Huang, CY. et al. ATM stabilizes DNA double-strand-break complexes during V(D)J recombination. Nature 442, 466–470 (2006). https://doi.org/10.1038/nature04866
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature04866
This article is cited by
-
AGO2 silences mobile transposons in the nucleus of quiescent cells
Nature Structural & Molecular Biology (2023)
-
An Igh distal enhancer modulates antigen receptor diversity by determining locus conformation
Nature Communications (2023)
-
The role of chromatin loop extrusion in antibody diversification
Nature Reviews Immunology (2022)
-
Subclonal heterogeneity sheds light on the transformation trajectory in IGLV3-21R110 chronic lymphocytic leukemia
Blood Cancer Journal (2022)
-
T Cell Repertoire Abnormality in Immunodeficiency Patients with DNA Repair and Methylation Defects
Journal of Clinical Immunology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.