Abstract
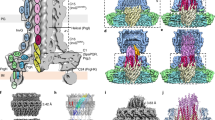
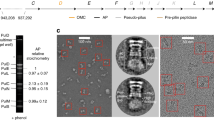
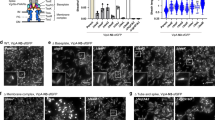
Assembly of multi-component supramolecular machines is fundamental to biology, yet in most cases, assembly pathways and their control are poorly understood. An example is the type III secretion machine, which mediates the transfer of bacterial virulence proteins into host cells1. A central component of this nanomachine is the needle complex or injectisome, an organelle associated with the bacterial envelope that is composed of a multi-ring base, an inner rod, and a protruding needle2. Assembly of this organelle proceeds in sequential steps that require the reprogramming of the secretion machine. Here we provide evidence that, in Salmonella typhimurium, completion of the assembly of the inner rod determines the size of the needle substructure. Assembly of the inner rod, which is regulated by the InvJ protein, triggers conformational changes on the cytoplasmic side of the injectisome, reprogramming the secretion apparatus to stop secretion of the needle protein.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Galán, J. E. & Collmer, A. Type III secretion machines: bacterial devices for protein delivery into host cells. Science 284, 1322–1328 (1999)
Kubori, T. et al. Supramolecular structure of the Salmonella typhimurium type III protein secretion system. Science 280, 602–605 (1998)
Cornelis, G. R. & Van Gijsegem, F. Assembly and function of type III secretory systems. Annu. Rev. Microbiol. 54, 735–774 (2000)
Kubori, T., Sukhan, A., Aizawa, S. I. & Galán, J. E. Molecular characterization and assembly of the needle complex of the Salmonella typhimurium type III protein secretion system. Proc. Natl Acad. Sci. USA 97, 10225–10230 (2000)
Marlovits, T. C. et al. Structural insights into the assembly of the type III secretion needle complex. Science 306, 1040–1042 (2004)
Sukhan, A., Kubori, T., Wilson, J. & Galán, J. E. Genetic analysis of assembly of the Salmonella enterica serovar Typhimurium type III secretion-associated needle complex. J. Bacteriol. 183, 1159–1167 (2001)
Collazo, C. & Galán, J. E. Requirement of exported proteins for secretion through the invasion-associated Type III system in Salmonella typhimurium. Infect. Immun. 64, 3524–3531 (1996)
Journet, L., Agrain, C., Broz, P. & Cornelis, G. R. The needle length of bacterial injectisomes is determined by a molecular ruler. Science 302, 1757–1760 (2003)
Sukhan, A., Kubori, T. & Galan, J. E. Synthesis and localization of the Salmonella SPI-1 type III secretion needle complex proteins PrgI and PrgJ. J. Bacteriol. 185, 3480–3483 (2003)
Galán, J. E. Salmonella interaction with host cells: Type III secretion at work. Annu. Rev. Cell Dev. Biol. 17, 53–86 (2001)
Collazo, C. M., Zierler, M. K. & Galán, J. E. Functional analysis of the Salmonella typhimurium invasion genes invI and invJ and identification of a target of the protein secretion apparatus encoded in the inv locus. Mol. Microbiol. 15, 25–38 (1995)
Macnab, R. How bacteria assemble flagella. Annu. Rev. Microbiol. 57, 77–100 (2003)
Macnab, R. Type III flagellar protein export and flagellar assembly. Biochim. Biophys. Acta 1694, 207–217 (2004)
Wang, R. F. & Kushner, S. R. Construction of versatile low-copy-number vectors for cloning, sequencing and gene expression in Escherichia coli. Gene 100, 195–199 (1991)
Chen, L. M., Kaniga, K. & Galán, J. E. Salmonella spp. are cytotoxic for cultured macrophages. Mol. Microbiol. 21, 1101–1115 (1996)
Galán, J. E. & Curtiss, R. III Cloning and molecular characterization of genes whose products allow Salmonella typhimurium to penetrate tissue culture cells. Proc. Natl Acad. Sci. USA 86, 6383–6387 (1989)
Acknowledgements
We thank members of the Galán laboratory for critical reviews of this manuscript. We are also grateful to the Yale School of Medicine for the support of the Cryo Electron Microscopy Core Facility. Molecular graphic images were produced using the Chimera package from the Computer Graphics Laboratory, University of California, San Francisco (supported by the NIH). This work was supported by grants from the National Institutes of Health (to V.M.U. and J.E.G.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The three-dimensional density maps have been deposited into the EBI-MSD EMD database under the accession code EMD-1214. Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Supplementary Methods
This file contains additional description of the methods used in this study. (DOC 2536 kb)
Suplementary Figures
This file contains Supplementary Figures 1–3 and accompanying legends. (DOC 2784 kb)
Rights and permissions
About this article
Cite this article
Marlovits, T., Kubori, T., Lara-Tejero, M. et al. Assembly of the inner rod determines needle length in the type III secretion injectisome. Nature 441, 637–640 (2006). https://doi.org/10.1038/nature04822
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature04822
This article is cited by
-
Type three secretion system in Salmonella Typhimurium: the key to infection
Genes & Genomics (2020)
-
Control of type III protein secretion using a minimal genetic system
Nature Communications (2017)
-
Identification of a novel type III secretion-associated outer membrane-bound protein from Xanthomonas campestris pv. campestris
Scientific Reports (2017)
-
Assembly, structure, function and regulation of type III secretion systems
Nature Reviews Microbiology (2017)
-
HrcU and HrpP are pathogenicity factors in the fire blight pathogen Erwinia amylovora required for the type III secretion of DspA/E
BMC Microbiology (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.