Abstract
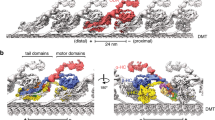
The axoneme, which forms the core of eukaryotic flagella and cilia, is one of the largest macromolecular machines, with a structure that is largely conserved from protists to mammals1. Microtubule doublets are structural components of axonemes that contain a number of proteins besides tubulin, and are usually found in arrays of nine doublets arranged around two singlet microtubules. Coordinated sliding of adjacent doublets, which involves a host of other proteins in the axoneme, produces periodic beating movements of the axoneme. We have obtained a three-dimensional density map of intact microtubule doublets using cryo-electron tomography and image averaging. Our map, with a resolution of about 3 nm, provides insights into locations of particular proteins within the doublets and the structural features of the doublets that define their mechanical properties. We identify likely candidates for several of these non-tubulin components of the doublets. This work offers insight on how tubulin protofilaments and accessory proteins attach together to form the doublets and provides a structural basis for understanding doublet function in axonemes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Kohl, L. & Bastin, P. The flagellum of trypanosomes. Int. Rev. Cytol. 244, 227–285 (2005)
Nogales, E., Wolf, S. G. & Downing, K. H. Structure of the αβ tubulin dimer by electron crystallography. Nature 391, 199–203 (1998)
Witman, G. B., Carlson, K., Berliner, J. & Rosenbaum, J. L. Chlamydomonas flagella. I. Isolation and electrophoretic analysis of microtubules, matrix, membranes, and mastigonemes. J. Cell Biol. 54, 507–539 (1972)
Witman, G. B., Carlson, K. & Rosenbaum, J. L. Chlamydomonas flagella. II. The distribution of tubulins 1 and 2 in the outer doublet microtubules. J. Cell Biol. 54, 540–555 (1972)
Meza, I., Huang, B. & Bryan, J. Chemical heterogeneity of protofilaments forming the outer doublets from sea urchin flagella. Exp. Cell Res. 74, 535–540 (1972)
Linck, R. W. & Norrander, J. M. Protofilament ribbon compartments of ciliary and flagellar microtubules. Protist 154, 299–311 (2003)
Linck, R. W. Flagellar doublet microtubules: fractionation of minor components and α-tubulin from specific regions of the A-tubule. J. Cell Sci. 20, 405–439 (1976)
Linck, R. W. & Langevin, G. L. Structure and chemical composition of insoluble filamentous components of sperm flagellar microtubules. J. Cell Sci. 58, 1–22 (1982)
Linck, R. W., Amos, L. A. & Amos, W. B. Localization of tektin filaments in microtubules of sea urchin sperm flagella by immunoelectron microscopy. J. Cell Biol. 100, 126–135 (1985)
Linck, R. W. & Stephens, R. E. Biochemical characterization of tektins from sperm flagellar doublet microtubules. J. Cell Biol. 104, 1069–1075 (1987)
Chang, X. J. & Piperno, G. Cross-reactivity of antibodies specific for flagellar tektin and intermediate filament subunits. J. Cell Biol. 104, 1563–1568 (1987)
Linck, R. W., Albertini, D. F., Kenney, D. M. & Langevin, G. L. Tektin filaments: chemically unique filaments of sperm flagellar microtubules. Cell Motility Suppl. 1, 127–132 (1982)
Chen, R., Perrone, C. A., Amos, L. A. & Linck, R. W. Tektin B1 from ciliary microtubules: primary structure as deduced from the cDNA sequence and comparison with tektin A1. J. Cell Sci. 106, 909–918 (1993)
Norrander, J. M., Amos, L. A. & Linck, R. W. Primary structure of tektin A1: comparison with intermediate-filament proteins and a model for its association with tubulin. Proc. Natl Acad. Sci. USA 89, 8567–8571 (1992)
Steffen, W. & Linck, R. W. Relationship between tektins and intermediate filament proteins: an immunological study. Cell Motil. Cytoskel. 14, 359–371 (1989)
Pirner, M. A. & Linck, R. W. Tektins are heterodimeric polymers in flagellar microtubules with axial periodicities matching the tubulin lattice. J. Biol. Chem. 269, 31800–31806 (1994)
Hinchcliffe, E. H. & Linck, R. W. Two proteins isolated from sea urchin sperm flagella: structural components common to the stable microtubules of axonemes and centrioles. J. Cell Sci. 111, 585–595 (1998)
Patel-King, R. S., Benashski, S. E. & King, S. M. A bipartite Ca2 + -regulated nucleoside-diphosphate kinase system within the Chlamydomonas flagellum. The regulatory subunit p72. J. Biol. Chem. 277, 34271–34279 (2002)
Ikeda, K. et al. Rib72, a conserved protein associated with the ribbon compartment of flagellar A-microtubules and potentially involved in the linkage between outer doublet microtubules. J. Biol. Chem. 278, 7725–7734 (2003)
Lindemann, C. B. Testing the geometric clutch hypothesis. Biol. Cell. 96, 681–690 (2004)
Bozkurt, H. H. & Woolley, D. M. Morphology of nexin links in relation to interdoublet sliding in the sperm flagellum. Cell Motil. Cytoskel. 24, 109–118 (1993)
Woolley, D. M. Studies on the eel sperm flagellum. I. The structure of the inner dynein arm complex. J. Cell Sci. 110, 85–94 (1997)
Huang, B., Ramanis, Z. & Luck, D. J. Suppressor mutations in Chlamydomonas reveal a regulatory mechanism for flagellar function. Cell 28, 115–124 (1982)
Gardner, L. C., O'Toole, E., Perrone, C. A., Giddings, T. & Porter, M. E. Components of a “dynein regulatory complex” are located at the junction between the radial spokes and the dynein arms in Chlamydomonas flagella. J. Cell Biol. 127, 1311–1325 (1994)
Piperno, G., Mead, K., LeDizet, M. & Moscatelli, A. Mutations in the “dynein regulatory complex” alter the ATP-insensitive binding sites for inner arm dyneins in Chlamydomonas axonemes. J. Cell Biol. 125, 1109–1117 (1994)
Piperno, G., Mead, K. & Shestak, W. The inner dynein arms I2 interact with a “dynein regulatory complex” in Chlamydomonas flagella. J. Cell Biol. 118, 1455–1463 (1992)
Kar, S., Fan, J., Smith, M. J., Goedert, M. & Amos, L. A. Repeat motifs of tau bind to the insides of microtubules in the absence of taxol. EMBO J. 22, 70–77 (2003)
Waterman-Storer, C. M. in Current Protocols in Cell Biology (eds Bonifacino, J. S., Dasso, M., Harford, J. B., Lippincott-Schwartz, J. & Yamada, K. M.) 13.1.6–13.1.7 (John Wiley, New York, 1998)
Hoops, H. J. & Witman, G. B. Outer doublet heterogeneity reveals structural polarity related to beat direction in Chlamydomonas flagella. J. Cell Biol. 97, 902–908 (1983)
Mastronarde, D. N., O'Toole, E. T., McDonald, K. L., McIntosh, J. R. & Porter, M. E. Arrangement of inner dynein arms in wild-type and mutant flagella of Chlamydomonas. J. Cell Biol. 118, 1145–1162 (1992)
Acknowledgements
We thank A. Killilea for sea urchin collection, K. Gull for discussions on protofilament numbering, H. Li, B. Rockel and D. Typke for discussions and help with image processing, B. Heymann and C. Yang for discussions and help with BSOFT library usage, and R. Glaeser and M. Auer for suggestions and encouragement. This work is supported by NIH grants and by the US Department of Energy.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Supplementary Notes
This file contains Supplementary Figures 1–3, Supplementary Methods and additional references. (PDF 1739 kb)
Rights and permissions
About this article
Cite this article
Sui, H., Downing, K. Molecular architecture of axonemal microtubule doublets revealed by cryo-electron tomography. Nature 442, 475–478 (2006). https://doi.org/10.1038/nature04816
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature04816
This article is cited by
-
In situ structure determination at nanometer resolution using TYGRESS
Nature Methods (2020)
-
The molecular structure of mammalian primary cilia revealed by cryo-electron tomography
Nature Structural & Molecular Biology (2020)
-
Self-organization of self-clearing beating patterns in an array of locally interacting ciliated cells formulated as an adaptive Boolean network
Theory in Biosciences (2020)
-
A lumenal interrupted helix in human sperm tail microtubules
Scientific Reports (2018)
-
The Developmental Process of the Growing Motile Ciliary Tip Region
Scientific Reports (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.