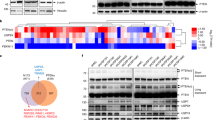
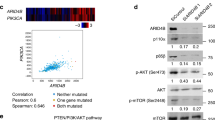
Abstract
The proto-oncogene AKT (also known as PKB) is activated in many human cancers, mostly owing to loss of the PTEN tumour suppressor1. In such tumours, AKT becomes enriched at cell membranes where it is activated by phosphorylation. Yet many targets inhibited by phosphorylated AKT (for example, the FOXO transcription factors) are nuclear; it has remained unclear how relevant nuclear phosphorylated AKT (pAKT) function is for tumorigenesis. Here we show that the PMLtumour suppressor prevents cancer by inactivating pAKT inside the nucleus. We find in a mouse model that Pml loss markedly accelerates tumour onset, incidence and progression in Pten-heterozygous mutants, and leads to female sterility with features that recapitulate the phenotype of Foxo3a knockout mice2. We show that Pml deficiency on its own leads to tumorigenesis in the prostate, a tissue that is exquisitely sensitive to pAkt levels, and demonstrate that Pml specifically recruits the Akt phosphatase PP2a as well as pAkt into Pml nuclear bodies. Notably, we find that Pml-null cells are impaired in PP2a phosphatase activity towards Akt, and thus accumulate nuclear pAkt. As a consequence, the progressive reduction in Pml dose leads to inactivation of Foxo3a-mediated transcription of proapoptotic Bim and the cell cycle inhibitor p27kip1. Our results demonstrate that Pml orchestrates a nuclear tumour suppressor network for inactivation of nuclear pAkt, and thus highlight the importance of AKT compartmentalization in human cancer pathogenesis and treatment.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Luo, J., Manning, B. D. & Cantley, L. C. Targeting the PI3K–Akt pathway in human cancer: rationale and promise. Cancer Cell 4, 257–262 (2003)
Brenkman, A. B. & Burgering, B. M. FoxO3a eggs on fertility and aging. Trends Mol. Med. 9, 464–467 (2003)
Salomoni, P. & Pandolfi, P. P. The role of PML in tumor suppression. Cell 108, 165–170 (2002)
Gurrieri, C. et al. Loss of the tumour suppressor PML in human cancers of multiple histologic origins. J. Natl Cancer Inst. 96, 269–279 (2004)
Goel, A. et al. Frequent inactivation of PTEN by promoter hypermethylation in microsatellite instability-high sporadic colorectal cancers. Cancer Res. 64, 3014–3021 (2004)
Porkka, K. P. & Visakorpi, T. Molecular mechanisms of prostate cancer. Eur. Urol. 45, 683–691 (2004)
Di Cristofano, A., Pesce, B., Cordon-Cardo, C. & Pandolfi, P. P. Pten is essential for embryonic development and tumour suppression. Nature Genet. 19, 348–355 (1998)
Podsypanina, K. et al. Mutation of Pten/Mmac1 in mice causes neoplasia in multiple organ systems. Proc. Natl Acad. Sci. USA 96, 1563–1568 (1999)
Suzuki, A. et al. High cancer susceptibility and embryonic lethality associated with mutation of the PTEN tumor suppressor gene in mice. Curr. Biol. 8, 1169–1178 (1998)
Di Cristofano, A. et al. Impaired Fas response and autoimmunity in Pten+/- mice. Science 285, 2122–2125 (1999)
Trotman, L. C. et al. Pten dose dictates cancer progression in the prostate. PLoS Biol. 1, E59 (2003)
Wang, S. et al. Prostate-specific deletion of the murine Pten tumor suppressor gene leads to metastatic prostate cancer. Cancer Cell 4, 209–221 (2003)
Di Cristofano, A., De Acetis, M., Koff, A., Cordon-Cardo, C. & Pandolfi, P. P. Pten and p27KIP1 cooperate in prostate cancer tumor suppression in the mouse. Nature Genet. 27, 222–224 (2001)
Maehama, T., Taylor, G. S. & Dixon, J. E. PTEN and myotubularin: novel phosphoinositide phosphatases. Annu. Rev. Biochem. 70, 247–279 (2001)
Stambolic, V. et al. Negative regulation of PKB/Akt-dependent cell survival by the tumor suppressor PTEN. Cell 95, 29–39 (1998)
Sarbassov, D. D., Guertin, D. A., Ali, S. M. & Sabatini, D. M. Phosphorylation and regulation of Akt/PKB by the rictor–mTOR complex. Science 307, 1098–1101 (2005)
Brazil, D. P., Yang, Z. Z. & Hemmings, B. A. Advances in protein kinase B signalling: AKTion on multiple fronts. Trends Biochem. Sci. 29, 233–242 (2004)
Tran, H., Brunet, A., Griffith, E. C. & Greenberg, M. E. The many forks in FOXO's road. Sci. STKE 2003, RE5 (2003)
Brunet, A. et al. Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell 96, 857–868 (1999)
Castrillon, D. H., Miao, L., Kollipara, R., Horner, J. W. & DePinho, R. A. Suppression of ovarian follicle activation in mice by the transcription factor Foxo3a. Science 301, 215–218 (2003)
Hosaka, T. et al. Disruption of forkhead transcription factor (FOXO) family members in mice reveals their functional diversification. Proc. Natl Acad. Sci. USA 101, 2975–2980 (2004)
Millward, T. A., Zolnierowicz, S. & Hemmings, B. A. Regulation of protein kinase cascades by protein phosphatase 2A. Trends Biochem. Sci. 24, 186–191 (1999)
Gao, T., Furnari, F. & Newton, A. C. PHLPP: a phosphatase that directly dephosphorylates Akt, promotes apoptosis, and suppresses tumor growth. Mol. Cell 18, 13–24 (2005)
Janssens, V., Goris, J. & Van Hoof, C. PP2A: the expected tumor suppressor. Curr. Opin. Genet. Dev. 15, 34–41 (2005)
Li, L., Ren, C. H., Tahir, S. A., Ren, C. & Thompson, T. C. Caveolin-1 maintains activated Akt in prostate cancer cells through scaffolding domain binding site interactions with and inhibition of serine/threonine protein phosphatases PP1 and PP2A. Mol. Cell. Biol. 23, 9389–9404 (2003)
Hanahan, D. & Weinberg, R. A. The hallmarks of cancer. Cell 100, 57–70 (2000)
Chen, Z. et al. Crucial role of p53-dependent cellular senescence in suppression of Pten-deficient tumorigenesis. Nature 436, 725–730 (2005)
Wang, Z. G. et al. PML is essential for multiple apoptotic pathways. Nature Genet. 20, 266–272 (1998)
Guo, A. et al. The function of PML in p53-dependent apoptosis. Nature Cell Biol. 2, 730–736 (2000)
Brunet, A. et al. Stress-dependent regulation of FOXO transcription factors by the SIRT1 deacetylase. Science 303, 2011–2015 (2004)
Acknowledgements
We thank R. Bernardi, B. Carver, Z. Chen, S. Clohessy, T. Maeda and T. Yung for discussion, help with reagents and data analysis; W. Golden and M. S. Jiao for help with pathology analysis; and C. Le, M. Lupu and C. Matei for help with MR imaging. This work was supported by NIH grants to P.P.P. and P.P.S. as well as grants to J.A.K. and the Memorial Sloan-Kettering Cancer Center. Author Contributions The experiments were conceived and designed by L.C.T., A.A., P.P.S., J.A.K., C.C.-C. and P.P.P. Experiments were performed by L.C.T., A.A., P.P.S. and J.A.K. Data were analysed by L.C.T., A.A., P.P.S., J.A.K., C.C.-C. and P.P.P. The paper was written by L.C.T. and P.P.P.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Supplementary Notes
This file contains the Supplementary Figures and Supplementary Methods. (PDF 9021 kb)
Rights and permissions
About this article
Cite this article
Trotman, L., Alimonti, A., Scaglioni, P. et al. Identification of a tumour suppressor network opposing nuclear Akt function. Nature 441, 523–527 (2006). https://doi.org/10.1038/nature04809
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature04809
This article is cited by
-
Filamentous nuclear actin regulation of PML NBs during the DNA damage response is deregulated by prelamin A
Cell Death & Disease (2022)
-
PML-II regulates ERK and AKT signal activation and IFNα-induced cell death
Cell Communication and Signaling (2021)
-
Regulation of PKCβ levels and autophagy by PML is essential for high-glucose-dependent mesenchymal stem cell adipogenesis
International Journal of Obesity (2019)
-
PML: Regulation and multifaceted function beyond tumor suppression
Cell & Bioscience (2018)
-
Transient PP2A inhibition alleviates normal tissue stem cell susceptibility to cell death during radiotherapy
Cell Death & Disease (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.