Abstract
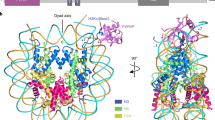
Mono-, di- and trimethylated states of particular histone lysine residues are selectively found in different regions of chromatin, thereby implying specialized biological functions for these marks ranging from heterochromatin formation to X-chromosome inactivation and transcriptional regulation1,2,3. A major challenge in chromatin biology has centred on efforts to define the connection between specific methylation states and distinct biological read-outs impacting on function4. For example, histone H3 trimethylated at lysine 4 (H3K4me3) is associated with transcription start sites of active genes5,6,7, but the molecular ‘effectors’ involved in specific recognition of H3K4me3 tails remain poorly understood. Here we demonstrate the molecular basis for specific recognition of H3(1–15)K4me3 (residues 1–15 of histone H3 trimethylated at K4) by a plant homeodomain (PHD) finger of human BPTF (bromodomain and PHD domain transcription factor), the largest subunit of the ATP-dependent chromatin-remodelling complex, NURF (nucleosome remodelling factor). We report on crystallographic and NMR structures of the bromodomain-proximal PHD finger of BPTF in free and H3(1–15)K4me3-bound states. H3(1–15)K4me3 interacts through anti-parallel β-sheet formation on the surface of the PHD finger, with the long side chains of arginine 2 (R2) and K4me3 fitting snugly in adjacent pre-formed surface pockets, and bracketing an invariant tryptophan. The observed stapling role by non-adjacent R2 and K4me3 provides a molecular explanation for H3K4me3 site specificity. Binding studies establish that the BPTF PHD finger exhibits a modest preference for K4me3- over K4me2-containing H3 peptides, and discriminates against monomethylated and unmodified counterparts. Furthermore, we identified key specificity-determining residues from binding studies of H3(1–15)K4me3 with PHD finger point mutants. Our findings call attention to the PHD finger as a previously uncharacterized chromatin-binding module found in a large number of chromatin-associated proteins.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Kouzarides, T. Histone methylation in transcriptional control. Curr. Opin. Genet. Dev. 12, 198–209 (2002)
Sims, R. J. III, Nishioka, K. & Reinberg, D. Histone lysine methylation: a signature for chromatin function. Trends Genet. 19, 629–639 (2003)
Martin, C. & Zhang, Y. The diverse functions of histone lysine methylation. Nature Rev. Mol. Cell Biol. 6, 838–849 (2005)
Fischle, W., Wang, Y. & Allis, D. Binary switches and modification cassettes in histone biology and beyond. Nature 425, 475–479 (2003)
Santos-Rosa, H. et al. Active genes are tri-methylated at K4 of histone H3. Nature 419, 407–411 (2002)
Santos-Rosa, H. et al. Methylation of histone H3 K4 mediates association of the Isw1p ATPase with chromatin. Mol. Cell 12, 1325–1332 (2003)
Schneider, R. et al. Histone H3 lysine 4 methylation patterns in higher eukaryotic genes. Nature Cell Biol. 6, 73–77 (2004)
Strahl, B. D. & Allis, C. D. The language of covalent histone modifications. Nature 403, 41–45 (2000)
Wu, J. & Grunstein, M. 25 years after the nucleosome model: chromatin modifications. Trends Biochem. Sci. 25, 619–623 (2000)
Jenuwein, T. & Allis, C. D. Translating the histone code. Science 293, 1074–1080 (2001)
Felsenfeld, G. & Groudine, M. Controlling the double helix. Nature 421, 448–453 (2003)
Pray-Grant, M. G. et al. Chd1 chromodomain links histone H3 methylation with SAGA-SLIK-dependent acetylation. Nature 433, 434–438 (2005)
Sims, R. J. III et al. Human but not yeast CHD1 binds directly and selectively to histone H3 methylated at lysine 4 via its tandem chromodomains. J. Biol. Chem. 280, 41789–41792 (2005)
Flanagan, J. F. et al. Double chromodomains cooperate to recognize the methylated histone H3 tail. Nature 438, 1181–1185 (2005)
Wysocka, J. et al. WDR5 associates with histone H3 methylated at K4 and is essential for H3 K4 methylation and vertebrate development. Cell 121, 859–872 (2005)
Wysocka, J. et al. A PHD finger of NURF couples histone H3 lysine 4 trimethylation with chromatin remodelling. Nature advance online publication, doi:10.1038/nature04815 (21 May 2006).
Tsukiyama, T. & Wu, C. Purification and properties of an ATP-dependent nucleosome remodeling factor. Cell 83, 1011–1020 (1995)
Xiao, H. et al. Dual functions of largest NURF subunit NURF301 in nucleosome sliding and transcription factor interactions. Mol. Cell 8, 531–543 (2001)
Narlikar, G. J., Fan, H. Y. & Kingston, R. E. Cooperation between complexes that regulate chromatin structure and transcription. Cell 108, 475–487 (2002)
Pascual, J., Martinez-Yamout, M., Dyson, H. J. & Wright, P. E. Structure of the PHD zinc finger from human Williams-Beuren syndrome transcription factor. J. Mol. Biol. 304, 723–729 (2000)
Capili, A. D., Schultz, D. C., Rauscher, F. J. III & Borden, K. L. Solution structure of the PHD domain from the KAP-1 corepressor: structural determinants for PHD, RING and LIM zinc-binding domains. EMBO J. 20, 165–177 (2001)
Bottomley, M. J. et al. NMR structure of the first PHD finger of autoimmune regulator protein (AIRE1). J. Biol. Chem. 280, 11505–11512 (2005)
Kwan, A. H. et al. Engineering a protein scaffold from a PHD finger. Structure 11, 803–813 (2003)
Nielsen, P. R. et al. Structure of the HP1 chromodomain bound to histone H3 methylated at lysine 9. Nature 416, 103–107 (2002)
Jacobs, S. A. & Khorasanizadeh, S. Structure of HP1 chromodomain bound to a lysine 9-methylated histone H3 tail. Science 295, 2080–2083 (2002)
Min, J., Zhang, Y. & Xu, R.-M. Structural basis for specific binding of polycomb chromodomain to histone H3 methylated at Lys 27. Genes Dev. 17, 1823–1828 (2003)
Zeng, L. & Zhou, M. M. Bromodomain: an acetyl-lysine binding domain. FEBS Lett. 513, 124–128 (2002)
Bottomley, M. J. Structures of protein domains that create or recognize histone modifications. EMBO Rep. 5, 464–469 (2004)
Dhalluin, C. et al. Structure and ligand of a histone acetyltransferase bromodomain. Nature 399, 491–496 (1999)
Owen, D. J. et al. The structural basis for the recognition of acetylated histone H4 by the bromodomain of histone acetyltransferase Gcn5p. EMBO J. 19, 6141–6149 (2000)
Bienz, M. The PHD finger, a nuclear protein-interaction domain. Trends Biochem. Sci. 31, 35–40 (2006)
Acknowledgements
D.J.P. is supported by funds from the Abby Rockefeller Mauze Trust, and the Dewitt Wallace and Maloris Foundations. C.D.A. is supported by an NIH MERIT award and funds from Rockefeller University. S.I. holds a Ruth Kirschstein NIH postdoctoral fellowship and J.W. holds a Damon-Runyon CRF Fellowship. We thank the Peptide Core Facilities at Sloan-Kettering (S. S. Yi at Microchemistry and Proteomics) and Rockefeller University for the synthesis and purification of K4-methylated H3 peptides. We would like to thank the staff at beam line X25 at the Brookhaven National Laboratory and beam lines 23ID-D and 24ID-C of the Advanced Photon Source at the Argonne National Laboratory, supported by the US Department of Energy, for assistance with data collection. D.J.P. is a member of the New York Structural Biology Center, supported in part by funds from the NIH. Author Contributions H.L. is responsible for the X-ray and surface plasmon resonance studies, S.I. for the NMR studies, W.W. for the calorimetric studies, B.D. for fluorescence polarization studies and J.W. for identification and functional characterization of the BPTF PHD finger as a H3K4me3 reader. D.J.P. and C.D.A. supervised the structural and functional (see companion paper) aspects of the project, respectively, and take overall responsibility for their joint research. All authors discussed the results and commented on the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
Coordinates of the X-ray structures of the BPTF PHD finger-linker-bromodomain in the free state and when bound to H3(1–15)K4me3 and H3(1–15)K4me2 peptides have been deposited in the RCSB Protein Data Bank under accession codes 2F6N, 2F6J and 2FSA, respectively. Coordinates of the NMR structures of the BPTF PHD finger in the free state and H3(1–15)K4me3 peptide-bound state have been deposited in the RCSB Protein Data Bank under accession codes 2FUI and 2FUU, respectively. Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Supplementary Notes
This file contains Supplementary Methods, Supplementary Notes, Supplementary Figures 1–13 and Supplementary Tables 1–3. (PDF 2841 kb)
Rights and permissions
About this article
Cite this article
Li, H., Ilin, S., Wang, W. et al. Molecular basis for site-specific read-out of histone H3K4me3 by the BPTF PHD finger of NURF. Nature 442, 91–95 (2006). https://doi.org/10.1038/nature04802
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature04802
This article is cited by
-
Targeting regulation of VEGF by BPTF in non-small cell lung cancer and its potential clinical significance
European Journal of Medical Research (2022)
-
The Arabidopsis APOLO and human UPAT sequence-unrelated long noncoding RNAs can modulate DNA and histone methylation machineries in plants
Genome Biology (2022)
-
Reading and erasing of the phosphonium analogue of trimethyllysine by epigenetic proteins
Communications Chemistry (2022)
-
Histone Methylation Regulates Gene Expression in the Round Spermatids to Set the RNA Payloads of Sperm
Reproductive Sciences (2022)
-
Structure, function and inhibition of critical protein–protein interactions involving mixed lineage leukemia 1 and its fusion oncoproteins
Journal of Hematology & Oncology (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.