Abstract
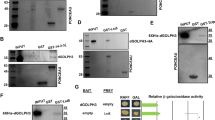
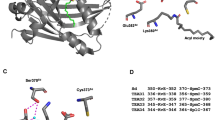
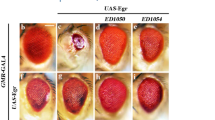
Precise regulation of the NFAT (nuclear factor of activated T cells) family of transcription factors (NFAT1–4) is essential for vertebrate development and function1. In resting cells, NFAT proteins are heavily phosphorylated and reside in the cytoplasm; in cells exposed to stimuli that raise intracellular free Ca2+ levels, they are dephosphorylated by the calmodulin-dependent phosphatase calcineurin and translocate to the nucleus1. NFAT dephosphorylation by calcineurin is countered by distinct NFAT kinases, among them casein kinase 1 (CK1) and glycogen synthase kinase 3 (GSK3)1,2,3,4,5. Here we have used a genome-wide RNA interference (RNAi) screen in Drosophila6,7 to identify additional regulators of the signalling pathway leading from Ca2+–calcineurin to NFAT. This screen was successful because the pathways regulating NFAT subcellular localization (Ca2+ influx, Ca2+–calmodulin–calcineurin signalling and NFAT kinases) are conserved across species8,9, even though Ca2+-regulated NFAT proteins are not themselves represented in invertebrates. Using the screen, we have identified DYRKs (dual-specificity tyrosine-phosphorylation regulated kinases) as novel regulators of NFAT. DYRK1A and DYRK2 counter calcineurin-mediated dephosphorylation of NFAT1 by directly phosphorylating the conserved serine-proline repeat 3 (SP-3) motif of the NFAT regulatory domain, thus priming further phosphorylation of the SP-2 and serine-rich region 1 (SRR-1) motifs by GSK3 and CK1, respectively. Thus, genetic screening in Drosophila can be successfully applied to cross evolutionary boundaries and identify new regulators of a transcription factor that is expressed only in vertebrates.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Hogan, P. G., Chen, L., Nardone, J. & Rao, A. Transcriptional regulation by calcium, calcineurin, and NFAT. Genes Dev. 17, 2205–2232 (2003)
Beals, C. R., Sheridan, C. M., Turck, C. W., Gardner, P. & Crabtree, G. R. Nuclear export of NF-ATc enhanced by glycogen synthase kinase-3. Science 275, 1930–1934 (1997)
Zhu, J. et al. Intramolecular masking of nuclear import signal on NF-AT4 by casein kinase I and MEKK1. Cell 93, 851–861 (1998)
Okamura, H. et al. Concerted dephosphorylation of the transcription factor NFAT1 induces a conformational switch that regulates transcriptional activity. Mol. Cell 6, 539–550 (2000)
Okamura, H. et al. A conserved docking motif for CK1 binding controls the nuclear localization of NFAT1. Mol. Cell. Biol. 24, 4184–4195 (2004)
Boutros, M. et al. Genome-wide RNAi analysis of growth and viability in Drosophila cells. Science 303, 832–835 (2004)
Armknecht, S. et al. High-throughput RNA interference screens in Drosophila tissue culture cells. Methods Enzymol. 392, 55–73 (2005)
Myers, E. W. et al. A whole-genome assembly of Drosophila. Science 287, 2196–2204 (2000)
Yeromin, A. V., Roos, J., Stauderman, K. A. & Cahalan, M. D. A store-operated calcium channel in Drosophila S2 cells. J. Gen. Physiol. 123, 167–182 (2004)
Aramburu, J. et al. Affinity-driven peptide selection of an NFAT inhibitor more selective than cyclosporin A. Science 285, 2129–2133 (1999)
Neal, J. W. & Clipstone, N. A. Glycogen synthase kinase-3 inhibits the DNA binding activity of NFATc. J. Biol. Chem. 276, 3666–3673 (2001)
Carafoli, E. The calcium signalling saga: tap water and protein crystals. Nature Rev. Mol. Cell Biol. 4, 326–332 (2003)
Roderick, H. L. & Bootman, M. D. Calcium influx: is Homer the missing link? Curr. Biol. 13, R976–R978 (2003)
Roos, J. et al. STIM1, an essential and conserved component of store-operated Ca2+ channel function. J. Cell Biol. 169, 435–445 (2005)
Liou, J. et al. STIM is a Ca2+ sensor essential for Ca2+-store-depletion-triggered Ca2+ influx. Curr. Biol. 15, 1235–1241 (2005)
Zhang, S. L. et al. STIM1 is a Ca2+ sensor that activates CRAC channels and migrates from the Ca2+ store to the plasma membrane. Nature 437, 902–905 (2005)
Suzuki, N. et al. Severe impairment of IL-1 and TLR signalling in mice lacking IRAK-4. Nature 416, 750–756 (2002)
Kannan, N. & Neuwald, A. F. Evolutionary constraints associated with functional specificity of the CMGC protein kinases MAPK, CDK, GSK, SRPK, DYRK, and CK2α. Protein Sci. 13, 2059–2077 (2004)
Lochhead, P. A., Sibbet, G., Morrice, N. & Cleghon, V. Activation-loop autophosphorylation is mediated by a novel transitional intermediate form of DYRKs. Cell 121, 925–936 (2005)
Becker, W. & Joost, H. G. Structural and functional characteristics of Dyrk, a novel subfamily of protein kinases with dual specificity. Prog. Nucleic Acid Res. Mol. Biol. 62, 1–17 (1999)
Kentrup, H. et al. Dyrk, a dual specificity protein kinase with unique structural features whose activity is dependent on tyrosine residues between subdomains VII and VIII. J. Biol. Chem. 271, 3488–3495 (1996)
Wiechmann, S. et al. Unusual function of the activation loop in the protein kinase DYRK1A. Biochem. Biophys. Res. Commun. 302, 403–408 (2003)
Himpel, S. et al. Specificity determinants of substrate recognition by the protein kinase DYRK1A. J. Biol. Chem. 275, 2431–2438 (2000)
Campbell, L. E. & Proud, C. G. Differing substrate specificities of members of the DYRK family of arginine-directed protein kinases. FEBS Lett. 510, 31–36 (2002)
Woods, Y. L. et al. The kinase DYRK phosphorylates protein-synthesis initiation factor eIF2Bɛ at Ser539 and the microtubule-associated protein tau at Thr212: potential role for DYRK as a glycogen synthase kinase 3-priming kinase. Biochem. J. 355, 609–615 (2001)
Nishi, Y. & Lin, R. DYRK2 and GSK-3 phosphorylate and promote the timely degradation of OMA-1, a key regulator of the oocyte-to-embryo transition in C. elegans. Dev. Biol. 288, 139–149 (2005)
Shirayama, M. et al. The conserved kinases CDK-1, GSK-3, KIN-19, and MBK-2 promote OMA-1 destruction to regulate the oocyte-to-embryo transition in C. elegans. Curr. Biol. 16, 47–55 (2006)
Harwood, A. J. Regulation of GSK-3: a cellular multiprocessor. Cell 105, 821–824 (2001)
Flotow, H. et al. Phosphate groups as substrate determinants for casein kinase I action. J. Biol. Chem. 265, 14264–14269 (1990)
Bhattacharyya, A. & Svendsen, C. N. Human neural stem cells: a new tool for studying cortical development in Down's syndrome. Genes Brain Behav. 2, 179–186 (2003)
Acknowledgements
We thank B. Mathey-Prevot, N. Perrimon, S. Armknecht, N. Ramadan, J. Murphy, K. Richardson, M. Booker and staff at the Drosophila RNAi Screening Centre at Harvard Medical School for valuable assistance with the screens. We also thank M. Dziadek for the gift of antibody against STIM1, and W. C. Yeh and T. Mak for sending spleen and lymph nodes isolated from Irak4+/- and Irak4-/-mice. This work was supported by NIH grants to A.R. and S.F. S. Sharma is supported by a postdoctoral fellowship from the Canadian Institutes of Health Research. Author Contributions The Drosophila RNAi screen was designed, validated and performed by Y.G., and positive candidates were identified through visual screening performed by Y.G., J.N. and H.O. S. Sharma identified DYRK as a functional NFAT kinase through biochemical and functional analyses of the human homologues of candidate Drosophila kinases, drawing in part on unpublished data and reagents provided by H.O. Selected other candidates were investigated in detail by Y.G., S. Sharma, D.B. and S. Srikanth. A.I. and S.F. performed measurements of intracellular calcium concentration in mammalian cells. J.N. and B.T. were responsible for bioinformatic analysis. P.G.H. and A.R. provided overall direction and supervised project planning and execution.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Supplementary Figures
This file contains Supplementary Figures 1–6 and their associated legends. (PDF 1773 kb)
Supplementary Tables
This file contains Supplementary Tables 1–3. (PDF 297 kb)
Supplementary Methods
This file contains additional details on the methods used in this study. (PDF 87 kb)
Supplementary Notes
This file contains additional references. (PDF 49 kb)
Rights and permissions
About this article
Cite this article
Gwack, Y., Sharma, S., Nardone, J. et al. A genome-wide Drosophila RNAi screen identifies DYRK-family kinases as regulators of NFAT. Nature 441, 646–650 (2006). https://doi.org/10.1038/nature04631
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature04631
This article is cited by
-
CRTAC1 enhances the chemosensitivity of non-small cell lung cancer to cisplatin by eliciting RyR-mediated calcium release and inhibiting Akt1 expression
Cell Death & Disease (2023)
-
A novel CDC25A/DYRK2 regulatory switch modulates cell cycle and survival
Cell Death & Differentiation (2022)
-
Regenerative approaches to preserve pancreatic β-cell mass and function in diabetes pathogenesis
Endocrine (2022)
-
Inhibition of Dyrk1A Attenuates LPS-Induced Neuroinflammation via the TLR4/NF-κB P65 Signaling Pathway
Inflammation (2022)
-
Cas9-mediated genome editing reveals a significant contribution of calcium signaling pathways to anhydrobiosis in Pv11 cells
Scientific Reports (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.