Abstract
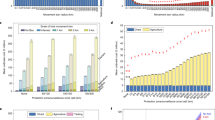
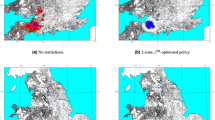
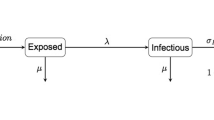
Foot-and-mouth disease (FMD) in the UK provides an ideal opportunity to explore optimal control measures for an infectious disease. The presence of fine-scale spatio-temporal data for the 2001 epidemic has allowed the development of epidemiological models that are more accurate than those generally created for other epidemics1,2,3,4,5 and provide the opportunity to explore a variety of alternative control measures. Vaccination was not used during the 2001 epidemic; however, the recent DEFRA (Department for Environment Food and Rural Affairs) contingency plan6 details how reactive vaccination would be considered in future. Here, using the data from the 2001 epidemic, we consider the optimal deployment of limited vaccination capacity in a complex heterogeneous environment. We use a model of FMD spread to investigate the optimal deployment of reactive ring vaccination of cattle constrained by logistical resources. The predicted optimal ring size is highly dependent upon logistical constraints but is more robust to epidemiological parameters. Other ways of targeting reactive vaccination can significantly reduce the epidemic size; in particular, ignoring the order in which infections are reported and vaccinating those farms closest to any previously reported case can substantially reduce the epidemic. This strategy has the advantage that it rapidly targets new foci of infection and that determining an optimal ring size is unnecessary.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Keeling, M. J. et al. Dynamics of the 2001 UK foot and mouth epidemic: stochastic dispersal in a heterogeneous landscape. Science 294, 813–817 (2001)
Keeling, M. J., Woolhouse, M. E. J., May, R. M., Davies, G. & Grenfell, B. T. Modelling vaccination strategies against foot-and-mouth disease. Nature 421, 136–142 (2003)
Ferguson, N. M., Donnelly, C. S. & Anderson, R. M. The foot-and-mouth epidemic in Great Britain: pattern of spread and impact of interventions. Science 292, 1155–1160 (2001)
Ferguson, N. M., Donnelly, C. S. & Anderson, R. M. Transmission intensity and impact of control policies on the foot and mouth epidemic in Great Britain. Nature 413, 542–548 (2001)
Morris, R. S., Wilesmith, J. W., Stern, M. W., Sanson, R. L. & Stevenson, M. A. Predictive spatial modelling of alternative control strategies for the foot-and-mouth disease epidemic in Great Britain, 2001. Vet Recórd 149, 137–144 (2001)
DEFRA Foot and Mouth Disease Contingency Planhttp://www.defra.gov.uk/footandmouth/contingency/index.htm (2004).
Anderson, I. Foot and Mouth Disease 2001: Lessons to be Learned Inquiry (The Stationery Office, London, 2002)
Council Directive 2003/85/EC. Official J. Eur. Union L306, 46, 1–87 (2003).
Muller, J., Schonfisch, B. & Kirkilionis, M. Ring vaccination. Math. Biol. 41, 143–171 (2000)
Barnett, P. V. & Carabin, H. A review of emergency foot-and-mouth disease (FMD) vaccines. Vaccine 20, 1505–1514 (2002)
Woolhouse, M. E. J., Haydon, D. T., Pearson, A. & Kitching, R. P. Failure of vaccination to prevent outbreaks of foot-and-mouth disease. Epidem. Infect. 116, 363–371 (1996)
Ferguson, N. M. et al. Planning for smallpox outbreaks. Nature 425, 681–685 (2003)
McLean, A. R. & Blower, S. M. Modelling HIV vaccination. Trends Microbiol. 3, 458–463 (1995)
Williams, J. R., Nokes, D. J. & Anderson, R. M. Targeted hepatitis B vaccination—a cost effective immunisation strategy for the UK? J. Epidemiol. Comm. Health 50, 667–673 (1996)
Woolhouse, M. E. J., Haydon, D. T. & Bundy, D. A. P. The design of veterinary vaccination programmes. Vet. J. 153, 41–47 (1997)
Woolhouse, M. E. J. et al. Heterogeneities in the transmission of infectious agents: implications for the design of control programs. Proc. Natl Acad. Sci. USA 94, 338–342 (1997)
Matthews, L. et al. Neighbourhood control policies and the spread of infectious disease. Proc. R. Soc. Lond. Ser. B 270, 1659–1666 (2003)
Acknowledgements
This research was supported by the Wellcome Trust. Author Contributions M.J.T. and M.J.K. were responsible for the model formulation and analysis of results; N.J.S. provided helpful discussions throughout; D.J.S. generated cleaned demographic and epidemic data; R.D. and S.P.B. provided vital statistical input; M.E.J.W. and B.T.G. were instrumental in the initial development of the project. All authors contributed to the writing of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Supplementary Notes
This file provides extra details of model formalism and parameterisation, the robustness of optimal outside-in ring vaccination strategies, and a comparison of shortest-distance and outside-in prioritisation strategies. (PDF 605 kb)
Rights and permissions
About this article
Cite this article
Tildesley, M., Savill, N., Shaw, D. et al. Optimal reactive vaccination strategies for a foot-and-mouth outbreak in the UK. Nature 440, 83–86 (2006). https://doi.org/10.1038/nature04324
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature04324
This article is cited by
-
Cost-effectiveness analysis of typhoid conjugate vaccines in an outbreak setting: a modeling study
BMC Infectious Diseases (2023)
-
Supervised classification of spatial epidemics incorporating infection time uncertainty
Statistical Methods & Applications (2023)
-
What can urban mobility data reveal about the spatial distribution of infection in a single city?
BMC Public Health (2019)
-
The role of movement restrictions in limiting the economic impact of livestock infections
Nature Sustainability (2019)
-
Bayesian inference of epidemiological parameters from transmission experiments
Scientific Reports (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.