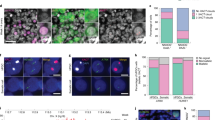
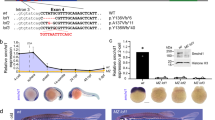
Abstract
In mammals, one of the two X chromosomes is inactivated in females to enable dosage compensation for X-linked gene products1. In rodents and marsupials, only the X chromosome of paternal origin (Xp) is silenced during early embryogenesis. This could be due to a carry-over effect of the X chromosome's passage through the male germ line, where it becomes transiently silenced together with the Y chromosome, during meiotic sex chromosome inactivation (MSCI)2. Here we show that XIST (X inactive specific transcript) transgenes, located on autosomes, do not undergo MSCI in the male germ line of mice and yet can induce imprinted cis-inactivation when paternally inherited, with identical kinetics to the Xp chromosome. This suggests that MSCI is not necessary for imprinted X-chromosome inactivation in mice. We also show that the Xp is transcribed, like autosomes, at zygotic gene activation rather than being ‘pre-inactivated’3. We propose that expression of the paternal Xist gene at zygotic gene activation is sufficient to trigger cis-inactivation of the X chromosome, or of an autosome carrying a Xist transgene.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Lyon, M. F. Gene action in the X chromosome of the mouse (Mus musculus L.). Nature 190, 372–373 (1961)
Lifschytz, E. & Lindsley, D. L. The role of X-chromosome inactivation during spermatogenesis (Drosophila-allocycly-chromosome evolution-male sterility-dosage compensation). Proc. Natl Acad. Sci. USA 69, 182–186 (1972)
Huynh, K. D. & Lee, J. T. Inheritance of a pre-inactivated paternal X chromosome in early mouse embryos. Nature 426, 857–862 (2003)
Takagi, N. & Sasaki, M. Preferential inactivation of the paternally derived X-chromosome in the extraembryonic membranes of the mouse. Nature 256, 640–642 (1975)
Mak, W. et al. Reactivation of the paternal X chromosome in early mouse embryos. Science 303, 666–669 (2004)
Okamoto, I., Otto, A. P., Allis, C. D., Reinberg, D. & Heard, E. Epigenetic dynamics of imprinted X inactivation during early mouse development. Science 303, 644–649 (2004)
Cooper, D. et al. X-inactivation in marsupials and monotremes. Semin. Dev. Biol. 4, 117–128 (1993)
Vandeberg, J. L., Robinson, E. S., Samollow, P. B. & Johnston, P. G. Isozymes: Current Topics in Biological and Medical Research 225–253 (Liss, New York, 1987)
Lyon, M. F. & Rastan, S. Parental source of chromosome imprinting and its relevance to X-chromosome inactivation. Differentiation 26, 63–67 (1984)
Shao, C. & Takagi, N. An extra maternally derived X chromosome is deleterious to early mouse development. Development 110, 969–975 (1990)
Tada, T. & Takagi, N. Parental imprinting on the mouse X chromosome: effects on the early development of X0, XXY and XXX embryos. Genet. Res. 62, 139–148 (1993)
Tada, T. et al. Imprint switching for non-random X-chromosome inactivation during mouse oocyte growth. Development 127, 3101–3105 (2000)
Heard, E. Recent advances in X-chromosome inactivation. Curr. Opin. Cell Biol. 16, 247–255 (2004)
Monk, M. & McLaren, A. X-chromosome activity in foetal germ cells of the mouse. J. Embryol. Exp. Morphol. 63, 75–84 (1981)
Grant, S. G. & Chapman, V. M. Mechanisms of X-chromosome regulation. Annu. Rev. Genet. 22, 199–233 (1988)
Heard, E. et al. Transgenic mice carrying an Xist-containing YAC. Hum. Mol. Genet. 5, 441–450 (1996)
Heard, E., Mongelard, F., Arnaud, D. & Avner, P. Xist yeast artificial chromosome transgenes function as X-inactivation centers only in multicopy arrays and not as single copies. Mol. Cell. Biol. 19, 3156–3166 (1999)
Penny, G. D., Kay, G. F., Sheardown, S. A., Rastan, S. & Brockdorff, N. Requirement for Xist in X chromosome inactivation. Nature 379, 131–137 (1996)
Marahrens, Y., Panning, B., Dausman, J., Strauss, W. & Jaenisch, R. Xist-deficient mice are defective in dosage compensation but not spermatogenesis. Genes Dev. 11, 156–166 (1997)
Simmler, M. C. et al. Localization and expression analysis of a novel conserved brain expressed transcript, Brx/BRX, lying within the Xic/XIC candidate region. Mamm. Genome 8, 760–766 (1997)
Carrel, L., Hunt, P. A. & Willard, H. F. Tissue and lineage-specific variation in inactive X chromosome expression of the murine Smcx gene. Hum. Mol. Genet. 5, 1361–1366 (1996)
Carrel, L. et al. X inactivation analysis and DNA methylation studies of the ubiquitin activating enzyme E1 and PCTAIRE-1 genes in human and mouse. Hum. Mol. Genet. 5, 391–401 (1996)
Lingenfelter, P. A. et al. Escape from X inactivation of Smcx is preceded by silencing during mouse development. Nature Genet. 18, 212–223 (1998)
Turner, J. et al. Silencing of unsynapsed meiotic chromosomes in the mouse. Nature Genet. 37, 41–47 (2005)
Fernandez-Capetillo, O. et al. H2AX is required for chromatin remodeling and inactivation of sex chromosomes in male mouse meiosis. Dev. Cell 4, 497–513 (2003)
Hoyer-Fender, S., Costanzi, C. & Pehrson, J. R. Histone macroH2A1.2 is concentrated in the XY-body by the early pachytene stage of spermatogenesis. Exp. Cell Res. 258, 254–260 (2000)
Baart, E. B., de Rooij, D. G., Keegan, K. S. & de Boer, P. Distribution of Atr protein in primary spermatocytes of a mouse chromosomal mutant: a comparison of preparation techniques. Chromosoma 109, 139–147 (2000)
Baarends, W. M. et al. Histone ubiquitination and chromatin remodeling in mouse spermatogenesis. Dev. Biol. 235, 322–333 (1999)
Kay, G. F., Barton, S. C., Surani, M. A. & Rastan, S. Imprinting and X chromosome counting mechanisms determine Xist expression in early mouse development. Cell 77, 639–650 (1994)
Heard, E., Chaumeil, J., Masui, O. & Okamoto, I. Mammalian X-chromosome inactivation: An epigenetics paradigm. Cold Spring Harb. Symp. Quant. Biol. 69, 89–102 (2004)
Acknowledgements
We would like to thank G. Almouzni, P. A. Defossez and N. Takagi for critical reading of the manuscript; members of our group and V. Colot for discussions; D. Reinberg and D. Papadopoulou for gifts of antibodies; and M. Blanche for help with mice. I.O. was supported by the CNRS; E.H. and I.O. were financed by the CNRS (ATIP programme), the French Ministry of Research (Action Concertée Incitative), the Curie Institute (Program Incitatif et Collaboratif) and the EU Network of Excellence (Epigenome). P.A. was supported by contracts from ARC by the Ministry of Research under the ACI programme and the EU Network of Excellence (Epigenome).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Supplementary Data
Analysis of Xist expression patterns in blastocysts derived from transgenic males or females. (DOC 21 kb)
Supplementary Discussion
A possible explanation for the evolution of imprinted X inactivation in rodents is provided. (DOC 20 kb)
Supplementary Methods
Additional details of methods used in this study, which could not be mentioned in the main text. (DOC 25 kb)
Supplementary Figure Legends
Text to accompany the below Supplementary Figures (DOC 26 kb)
Supplementary Figure 1
Paternally and maternally transmitted autosomal Xist YAC transgenes. (PDF 6056 kb)
Supplementary Figure 2
Additional marks of imprinted X inactivation on females embryos carrying paternally transmitted Tg53 Xist transgenes. (PDF 17739 kb)
Supplementary Figure 3
Transcriptional activity of the paternal X chromosome assayed by gene specific RNA FISH. (PDF 15342 kb)
Supplementary Figure 4
Transcriptional activity of the paternal X chromosome assayed by Cot-1 RNA FISH. (PDF 13679 kb)
Supplementary Figure 5
Chromatin marks associated with the XY body in pachytene stage spermatocytes do not affect the chromosome 13 pair in Tg53 testis. (PDF 9902 kb)
Supplementary Figure 6
Analysis of transgene induced cis-inactivation in embryos derived from homozygous males. (PDF 6222 kb)
Supplementary Tables
Supplementary Table 1 details Xist expression patterns in pre-implantation embryos derived from WT females and YAC Tg53 males. Supplementary Table 2 details antibodies and dilutions used. (DOC 88 kb)
Rights and permissions
About this article
Cite this article
Okamoto, I., Arnaud, D., Le Baccon, P. et al. Evidence for de novo imprinted X-chromosome inactivation independent of meiotic inactivation in mice. Nature 438, 369–373 (2005). https://doi.org/10.1038/nature04155
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature04155
This article is cited by
-
GATA transcription factors drive initial Xist upregulation after fertilization through direct activation of long-range enhancers
Nature Cell Biology (2023)
-
From genotype to phenotype: genetics of mammalian long non-coding RNAs in vivo
Nature Reviews Genetics (2022)
-
Gene regulation in time and space during X-chromosome inactivation
Nature Reviews Molecular Cell Biology (2022)
-
Parental-to-embryo switch of chromosome organization in early embryogenesis
Nature (2020)
-
Maternal H3K27me3-dependent autosomal and X chromosome imprinting
Nature Reviews Genetics (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.