Abstract
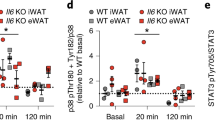
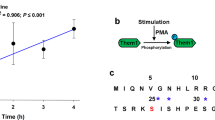
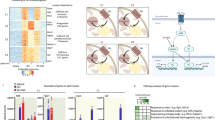
Hormones mobilize intracellular second messengers and initiate signalling cascades involving protein kinases and phosphatases, which are often spatially compartmentalized by anchoring proteins to increase signalling specificity1. These scaffold proteins may themselves be modulated by hormones2,3,4. In adipocytes, stimulation of β-adrenergic receptors increases cyclic AMP levels and activates protein kinase A (PKA)5, which stimulates lipolysis by phosphorylating hormone-sensitive lipase and perilipin6,7,8. Acute insulin treatment activates phosphodiesterase 3B, reduces cAMP levels and quenches β-adrenergic receptor signalling9. In contrast, chronic hyperinsulinaemic conditions (typical of type 2 diabetes) enhance β-adrenergic receptor-mediated cAMP production10. This amplification of cAMP signalling is paradoxical because it should enhance lipolysis, the opposite of the known short-term effect of hyperinsulinaemia. Here we show that in adipocytes, chronically high insulin levels inhibit β-adrenergic receptors (but not other cAMP-elevating stimuli) from activating PKA. We measured this using an improved fluorescent reporter and by phosphorylation of endogenous cAMP-response-element binding protein (CREB). Disruption of PKA scaffolding mimics the interference of insulin with β-adrenergic receptor signalling. Chronically high insulin levels may disrupt the close apposition of β-adrenergic receptors and PKA, identifying a new mechanism for crosstalk between heterologous signal transduction pathways.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Wong, W. & Scott, J. D. AKAP signalling complexes: focal points in space and time. Nature Rev. Mol. Cell Biol. 5, 959–970 (2004)
Salvador, L. M. et al. Neuronal microtubule-associated protein 2D is a dual A-kinase anchoring protein expressed in rat ovarian granulosa cells. J. Biol. Chem. 279, 27621–27632 (2004)
Carr, D. W., DeManno, D. A., Atwood, A., Hunzicker-Dunn, M. & Scott, J. D. Follicle-stimulating hormone regulation of A-kinase anchoring proteins in granulosa cells. J. Biol. Chem. 268, 20729–20732 (1993)
Feliciello, A., Rubin, C. S., Avvedimento, E. V. & Gottesman, M. E. Expression of a kinase anchor protein 121 is regulated by hormones in thyroid and testicular germ cells. J. Biol. Chem. 273, 23361–23366 (1998)
Taylor, S. S. et al. PKA: a portrait of protein kinase dynamics. Biochim. Biophys. Acta 1697, 259–269 (2004)
Londos, C., Honnor, R. C. & Dhillon, G. S. cAMP-dependent protein kinase and lipolysis in rat adipocytes. III. Multiple modes of insulin regulation of lipolysis and regulation of insulin responses by adenylate cyclase regulators. J. Biol. Chem. 260, 15139–15145 (1985)
Egan, J. J., Greenberg, A. S., Chang, M. K. & Londos, C. Control of endogenous phosphorylation of the major cAMP-dependent protein kinase substrate in adipocytes by insulin and beta-adrenergic stimulation. J. Biol. Chem. 265, 18769–18775 (1990)
Greenberg, A. S. et al. Perilipin, a major hormonally regulated adipocyte-specific phosphoprotein associated with the periphery of lipid storage droplets. J. Biol. Chem. 266, 11341–11346 (1991)
Elks, M. L. & Manganiello, V. C. Antilipolytic action of insulin: role of cAMP phosphodiesterase activation. Endocrinology 116, 2119–2121 (1985)
Hupfeld, C. J., Dalle, S. & Olefsky, J. M. β-Arrestin 1 down-regulation after insulin treatment is associated with supersensitization of β2 adrenergic receptor Gαs signalling in 3T3–L1 adipocytes. Proc. Natl Acad. Sci. USA 100, 161–166 (2003)
Zhang, J., Ma, Y., Taylor, S. S. & Tsien, R. Y. Genetically encoded reporters of protein kinase A activity reveal impact of substrate tethering. Proc. Natl Acad. Sci. USA 98, 14997–15002 (2001)
Muslin, A. J., Tanner, J. W., Allen, P. M. & Shaw, A. S. Interaction of 14-3-3 with signalling proteins is mediated by the recognition of phosphoserine. Cell 84, 889–897 (1996)
Durocher, D. et al. The molecular basis of FHA domain:phosphopeptide binding specificity and implications for phospho-dependent signalling mechanisms. Mol. Cell 6, 1169–1182 (2000)
Nerbonne, J. M., Richard, S., Nargeot, J. & Lester, H. A. New photoactivatable cyclic nucleotides produce intracellular jumps in cyclic AMP and cyclic GMP concentrations. Nature 310, 74–76 (1984)
Kosaki, A., Yamada, K. & Kuzuya, H. Reduced expression of the leptin gene (ob) by catecholamine through a Gs protein-coupled pathway in 3T3–L1 adipocytes. Diabetes 45, 1744–1749 (1996)
Fasshauer, M., Klein, J., Neumann, S., Eszlinger, M. & Paschke, R. Adiponectin gene expression is inhibited by β-adrenergic stimulation via protein kinase A in 3T3–L1 adipocytes. FEBS Lett. 507, 142–146 (2001)
Fasshauer, M., Klein, J., Neumann, S., Eszlinger, M. & Paschke, R. Isoproterenol inhibits resistin gene expression through a Gs-protein-coupled pathway in 3T3–L1 adipocytes. FEBS Lett. 500, 60–63 (2001)
Mayr, B. & Montminy, M. Transcriptional regulation by the phosphorylation-dependent factor CREB. Nature Rev. Mol. Cell Biol. 2, 599–609 (2001)
Scott, J. D. & Faux, M. C. Use of synthetic peptides in the dissection of protein-targeting interactions. Methods Mol. Biol. 88, 161–185 (1998)
Fraser, I. D. et al. Assembly of an A kinase-anchoring protein–β2-adrenergic receptor complex facilitates receptor phosphorylation and signalling. Curr. Biol. 10, 409–412 (2000)
Lin, F., Wang, H. & Malbon, C. C. Gravin-mediated formation of signalling complexes in β2-adrenergic receptor desensitization and resensitization. J. Biol. Chem. 275, 19025–19034 (2000)
Jurevicius, J. & Fischmeister, R. cAMP compartmentation is responsible for a local activation of cardiac Ca2+ channels by β-adrenergic agonists. Proc. Natl Acad. Sci. USA 93, 295–299 (1996)
Zaccolo, M. & Pozzan, T. Discrete microdomains with high concentration of cAMP in stimulated rat neonatal cardiac myocytes. Science 295, 1711–1715 (2002)
Tao, J., Wang, H. Y. & Malbon, C. C. Protein kinase A regulates AKAP250 (gravin) scaffold binding to the β2-adrenergic receptor. EMBO J. 22, 6419–6429 (2003)
Griesbeck, O., Baird, G. S., Campbell, R. E., Zacharias, D. A. & Tsien, R. Y. Reducing the environmental sensitivity of yellow fluorescent protein. Mechanism and applications. J. Biol. Chem. 276, 29188–29194 (2001)
Dalle, S. et al. Insulin induces heterologous desensitization of G-protein-coupled receptor and insulin-like growth factor I signalling by downregulating β-arrestin-1. Mol. Cell. Biol. 22, 6272–6285 (2002)
Acknowledgements
We thank Y. Ma for technical assistance and discussions and M. B. Yaffe for providing FHA cDNA. This work was supported by the Howard Hughes Medical Institute, the Alliance for Cell Signaling (R.Y.T.), the NIH (R.Y.T, C.J.H. and S.S.T) and the Johns Hopkins University School of Medicine and W. M. Keck Foundation (J.Z.). Author Contributions J.Z. and C.J.H. performed all the experiments; all authors contributed to planning the experiments, interpreting the data and writing the paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Supplementary Figures
Supplementary Figures S1-S4, showing effect of insulin treatment on βAR-associated cAMP generation, effect of insulin treatment on PKA activity in the nucleus, effect of the functionally inactive Ht31p, and a model for insulin modulated compartmentation in adipocytes. (PDF 93 kb)
Supplementary Methods
Additional information about the imaging methods used in this study. (DOC 24 kb)
Rights and permissions
About this article
Cite this article
Zhang, J., Hupfeld, C., Taylor, S. et al. Insulin disrupts β-adrenergic signalling to protein kinase A in adipocytes. Nature 437, 569–573 (2005). https://doi.org/10.1038/nature04140
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature04140
This article is cited by
-
A fifty percent leucine-restricted diet reduces fat mass and improves glucose regulation
Nutrition & Metabolism (2021)
-
An ultrasensitive biosensor for high-resolution kinase activity imaging in awake mice
Nature Chemical Biology (2021)
-
Testosterone is sequestered in dysfunctional adipose tissue, modifying androgen-responsive genes
International Journal of Obesity (2020)
-
Rational Design of a Protein Kinase A Nuclear-cytosol Translocation Reporter
Scientific Reports (2020)
-
Defective fasting-induced PKA activation impairs adipose tissue glycogen degradation in obese Zucker rats
International Journal of Obesity (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.