Abstract
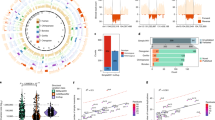
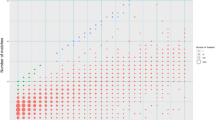
Human subtelomeres are polymorphic patchworks of interchromosomal segmental duplications at the ends of chromosomes. Here we provide evidence that these patchworks arose recently through repeated translocations between chromosome ends. We assess the relative contribution of the principal mechanisms of ectopic DNA repair to the formation of subtelomeric duplications and find that non-homologous end-joining predominates. Once subtelomeric duplications arise, they are prone to homology-based sequence transfers as shown by the incongruent phylogenetic relationships of neighbouring sections. Interchromosomal recombination of subtelomeres is a potent force for recent change. Cytogenetic and sequence analyses reveal that pieces of the subtelomeric patchwork have changed location and copy number with unprecedented frequency during primate evolution. Half of the known subtelomeric sequence has formed recently, through human-specific sequence transfers and duplications. Subtelomeric dynamics result in a gene duplication rate significantly higher than the genome average and could have both advantageous and pathological consequences in human biology. More generally, our analyses suggest an evolutionary cycle between segmental polymorphisms and genome rearrangements.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Samonte, R. V. & Eichler, E. E. Segmental duplications and the evolution of the primate genome. Nature Rev. Genet. 3, 65–72 (2002)
Bailey, J. A. et al. Recent segmental duplications in the human genome. Science 297, 1003–1007 (2002)
Bailey, J. A., Yavor, A. M., Massa, H. F., Trask, B. J. & Eichler, E. E. Segmental duplications: organization and impact within the current human genome project assembly. Genome Res. 11, 1005–1017 (2001)
Shaw, C. J. & Lupski, J. R. Implications of human genome architecture for rearrangement-based disorders: the genomic basis of disease. Hum. Mol. Genet. 13 (review issue 1), R57–R64 (2004)
Mefford, H. & Trask, B. J. The complex structure and dynamic evolution of human subtelomeres. Nature Rev. Genet. 3, 91–102 (2002)
Trask, B. J. et al. Members of the olfactory receptor gene family are contained in large blocks of DNA duplicated polymorphically near the ends of human chromosomes. Hum. Mol. Genet. 7, 13–26 (1998)
Monfouilloux, S. et al. Recent human-specific spreading of a subtelomeric domain. Genomics 51, 165–176 (1998)
Martin, C. L. et al. The evolutionary origin of human subtelomeric homologies—or where the ends begin. Am. J. Hum. Genet. 70, 972–984 (2002)
Fan, Y., Linardopoulou, E., Friedman, C., Williams, E. M. & Trask, B. J. Genomic structure and evolution of the ancestral chromosome fusion site in 2q13–2q14.1. Genome Res. 12, 1651–1662 (2002)
Riethman, H. C. et al. Integration of telomere sequences with the draft human genome sequence. Nature 409, 948–951 (2001)
Linardopoulou, E. et al. Transcriptional activity of multiple copies of a subtelomerically located olfactory receptor gene that is polymorphic in number and location. Hum. Mol. Genet. 10, 2373–2383 (2001)
Knight, S. J. & Flint, J. The use of subtelomeric probes to study mental retardation. Methods Cell Biol. 75, 799–831 (2004)
Brown, W. R. et al. Structure and polymorphism of human telomere-associated DNA. Cell 63, 119–132 (1990)
de Lange, T. et al. Structure and variability of human chromosome ends. Mol. Cell. Biol. 10, 518–527 (1990)
Flint, J. et al. Sequence comparison of human and yeast telomeres identifies structurally distinct subtelomeric domains. Hum. Mol. Genet. 6, 1305–1313 (1997)
Riethman, H. et al. Mapping and initial analysis of human subtelomeric sequence assemblies. Genome Res. 14, 18–28 (2004)
Smit, A. F. & Riggs, A. D. Tiggers and DNA transposon fossils in the human genome. Proc. Natl Acad. Sci. USA 93, 1443–1448 (1996)
Eichler, E. E., Archidiacono, N. & Rocchi, M. CAGGG repeats and the pericentromeric duplication of the hominoid genome. Genome Res. 9, 1048–1058 (1999)
Pfeiffer, P., Goedecke, W. & Obe, G. Mechanisms of DNA double-strand break repair and their potential to induce chromosomal aberrations. Mutagenesis 15, 289–302 (2000)
Rothkamm, K., Kruger, I., Thompson, L. H. & Lobrich, M. Pathways of DNA double-strand break repair during the mammalian cell cycle. Mol. Cell. Biol. 23, 5706–5715 (2003)
Bailey, J. A., Liu, G. & Eichler, E. E. An Alu transposition model for the origin and expansion of human segmental duplications. Am. J. Hum. Genet. 73, 823–834 (2003)
Zhou, Y. & Mishra, B. Quantifying the mechanisms for segmental duplications in mammalian genomes by statistical analysis and modeling. Proc. Natl Acad. Sci. USA 102, 4151–4156 (2005)
Nergadze, S. G., Rocchi, M., Azzalin, C. M., Mondello, C. & Giulotto, E. Insertion of telomeric repeats at intrachromosomal break sites during primate evolution. Genome Res. 14, 1704–1710 (2004)
Sawyer, S. Statistical tests for detecting gene conversion. Mol. Biol. Evol. 6, 526–538 (1989)
Mefford, H. C., Linardopoulou, E., Coil, D., van den Engh, G. & Trask, B. J. Comparative sequencing of a multicopy subtelomeric region containing olfactory receptor genes reveals multiple interactions between non-homologous chromosomes. Hum. Mol. Genet. 10, 2363–2372 (2001)
Der-Sarkissian, H., Vergnaud, G., Borde, Y. M., Thomas, G. & Londono-Vallejo, J. A. Segmental polymorphisms in the proterminal regions of a subset of human chromosomes. Genome Res. 12, 1673–1678 (2002)
Lemmers, R. J. et al. Facioscapulohumeral muscular dystrophy is uniquely associated with one of the two variants of the 4q subtelomere. Nature Genet. 32, 235–236 (2002)
Chen, F. C. & Li, W. H. Genomic divergences between humans and other hominoids and the effective population size of the common ancestor of humans and chimpanzees. Am. J. Hum. Genet. 68, 444–456 (2001)
Liu, G. et al. Analysis of primate genomic variation reveals a repeat-driven expansion of the human genome. Genome Res. 13, 358–368 (2003)
Lynch, M. & Conery, J. S. The evolutionary fate and consequences of duplicate genes. Science 290, 1151–1155 (2000)
She, X. et al. The structure and evolution of centromeric transition regions within the human genome. Nature 430, 857–864 (2004)
Ricchetti, M., Dujon, B. & Fairhead, C. Distance from the chromosome end determines the efficiency of double strand break repair in subtelomeres of haploid yeast. J. Mol. Biol. 328, 847–862 (2003)
Bass, H. W. Telomere dynamics unique to meiotic prophase: formation and significance of the bouquet. Cell. Mol. Life Sci. 60, 2319–2324 (2003)
d'Adda di Fagagna, F. et al. A DNA damage checkpoint response in telomere-initiated senescence. Nature 426, 194–198 (2003)
Zou, Y., Sfeir, A., Gryaznov, S. M., Shay, J. W. & Wright, W. E. Does a sentinel or a subset of short telomeres determine replicative senescence? Mol. Biol. Cell 15, 3709–3718 (2004)
Takai, H., Smogorzewska, A. & de Lange, T. DNA damage foci at dysfunctional telomeres. Curr. Biol. 13, 1549–1556 (2003)
Cornforth, M. N. & Eberle, R. L. Termini of human chromosomes display elevated rates of mitotic recombination. Mutagenesis 16, 85–89 (2001)
Hansen, T. F., Carter, A. J. & Chiu, C. H. Gene conversion may aid adaptive peak shifts. J. Theor. Biol. 207, 495–511 (2000)
Halme, A., Bumgarner, S., Styles, C. & Fink, G. R. Genetic and epigenetic regulation of the FLO gene family generates cell-surface variation in yeast. Cell 116, 405–415 (2004)
Fabre, E. et al. Comparative genomics in hemiascomycete yeasts: evolution of sex, silencing, and subtelomeres. Mol. Biol. Evol. 22, 856–873 (2005)
De Las Penas, A. et al. Virulence-related surface glycoproteins in the yeast pathogen Candida glabrata are encoded in subtelomeric clusters and subject to RAP1- and SIR-dependent transcriptional silencing. Genes Dev. 17, 2245–2258 (2003)
Warburton, D. De novo balanced chromosome rearrangements and extra marker chromosomes identified at prenatal diagnosis: clinical significance and distribution of breakpoints. Am. J. Hum. Genet. 49, 995–1013 (1991)
Genetics and Public Policy Center. Genetics Information: Translocations. http://www.dnapolicy.org/genetics/translocations.jhtml (2004).
Wong, Z., Royle, N. J. & Jeffreys, A. J. A novel human DNA polymorphism resulting from transfer of DNA from chromosome 6 to chromosome 16. Genomics 7, 222–234 (1990)
Sebat, J. et al. Large-scale copy number polymorphism in the human genome. Science 305, 525–528 (2004)
Iafrate, A. J. et al. Detection of large-scale variation in the human genome. Nature Genet. 36, 949–951 (2004)
Tatusova, T. A. & Madden, T. L. BLAST 2 Sequences, a new tool for comparing protein and nucleotide sequences. FEMS Microbiol. Lett. 174, 247–250 (1999)
Bray, N. & Pachter, L. MAVID multiple alignment server. Nucleic Acids Res. 31, 3525–3526 (2003)
Swofford, D. L. PAUP*. Phylogenetic Analysis Using Parsimony (*and Other Methods) (Sinauer Associates, Sunderland, 2000)
Acknowledgements
We are grateful to the many contributors to the Human Genome Project who generated the sequences that made this study possible, and to the Eichler and Haussler laboratory groups for making data on segmental duplications readily accessible. Our work was supported by the NIH. We thank E. Eichler, H. Malik, D. Gottschling, J. Gogarten, K. Rudd and M. Schlador for comments on the manuscript, and J. Felsenstein for advice on phylogenetic analyses.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Supplementary Methods
Detailed description of computational and experimental methods used in study and additional observations. (Includes references cited in Supplementary Tables S1-S11). (PDF 265 kb)
Supplmentary Tables S1-S11
Eleven supplementary tables, provided together in one pdf file. (PDF 536 kb)
Supplementary Figure S1
Diagram summarizing PCR analyses of a monochromosomal hybrid panel using 55 primer pairs distributed across subtelomeric regions. The results confirm block boundaries defined by available sequence and identify additional copies not yet represented in genome assembly. (PDF 445 kb)
Supplementary Figure S2
Diagram of an NAHR-medicated translocation leading to a homology breakpoint within subtelomeres. (PDF 709 kb)
Supplementary Figure S3
Diagram of one of three breakpoints with degenerate telomere repeats at the junction and undetermined mechanism. (PDF 203 kb)
Supplementary Figure S4
Diagram of deletion formed by non-allelic homologous recombination of Alu elements on either the same or different chromosomes. (PDF 595 kb)
Supplementary Figure S5
Multiple changes in the historical relationships among subtelomeric sequences are apparent in this diagram of the best matching pairs in 2-kbp windows across the multiple sequence alignment of a region shared by seven chromosomes. (PDF 94 kb)
Supplementary Figure S6
Close-up view of the gross disparity in subtelomeric content of two sequenced alleles of 16p. (PDF 100 kb)
Supplementary Figure S7
The distribution of the percent identity of all pairwise alignments of all subtelomeric blocks shows that most subtelomeric duplications are very recent. (PDF 64 kb)
Supplementary Figure S8
Diagram of the best match for each block in each subtelomeric contig illustrates the extensive regions created by recent exchange and their spatial arrangement. (PDF 250 kb)
Supplementary Data
Sequences of representative copies of each block in fasta format. (TXT 883 kb)
Rights and permissions
About this article
Cite this article
Linardopoulou, E., Williams, E., Fan, Y. et al. Human subtelomeres are hot spots of interchromosomal recombination and segmental duplication. Nature 437, 94–100 (2005). https://doi.org/10.1038/nature04029
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature04029
This article is cited by
-
Bread wheat satellitome: a complex scenario in a huge genome
Plant Molecular Biology (2024)
-
Emergence and influence of sequence bias in evolutionarily malleable, mammalian tandem arrays
BMC Biology (2023)
-
Telomere and subtelomere high polymorphism might contribute to the specificity of homologous recognition and pairing during meiosis in barley in the context of breeding
BMC Genomics (2023)
-
Acquisition of new function through gene duplication in the metallocarboxypeptidase family
Scientific Reports (2023)
-
Modelling segmental duplications in the human genome
BMC Genomics (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.