Abstract
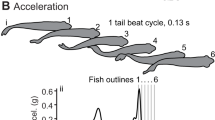
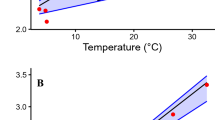
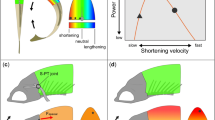
Effects of temperature on muscle contraction and powering movement are profound, outwardly obvious, and of great consequence to survival1,2. To cope with the effects of environmental temperature fluctuations, endothermic birds and mammals maintain a relatively warm and constant body temperature, whereas most fishes and other vertebrates are ectothermic and conform to their thermal niche, compromising performance at colder temperatures2,3. However, within the fishes the tunas and lamnid sharks deviate from the ectothermic strategy, maintaining elevated core body temperatures4,5 that presumably confer physiological advantages for their roles as fast and continuously swimming pelagic predators. Here we show that the salmon shark, a lamnid inhabiting cold, north Pacific waters, has become so specialized for endothermy that its red, aerobic, locomotor muscles, which power continuous swimming, seem mammal-like, functioning only within a markedly elevated temperature range (20–30 °C). These muscles are ineffectual if exposed to the cool water temperatures, and when warmed even 10 °C above ambient they still produce only 25–50% of the power produced at 26 °C. In contrast, the white muscles, powering burst swimming, do not show such a marked thermal dependence and work well across a wide range of temperatures.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Bennett, A. F. Temperature and muscle. J. Exp. Biol. 115, 333–344 (1985)
Johnston, I. A. & Temple, G. K. Thermal plasticity of skeletal muscle phenotype in ectothermic vertebrates and its significance for locomotory behaviour. J. Exp. Biol. 205, 2305–2322 (2002)
Bennett, A. F. Thermal dependence of locomotor capacity. Am. J. Physiol. 259, R253–R258 (1990)
Carey, F. G. & Teal, J. M. Mako and porbeagle: warm bodied sharks. Comp. Biochem. Physiol. 28, 199–204 (1969)
Carey, F. G. & Teal, J. M. Regulation of body temperature by the bluefin tuna. Comp. Biochem. Physiol. 28, 205–213 (1969)
Bernal, D., Dickson, K. A., Shadwick, R. E. & Graham, J. B. Analysis of the evolutionary convergence for high performance swimming in lamnid sharks and tunas. Comp. Biochem. Physiol. 129, 695–726 (2001)
Bernal, D. et al. Comparative studies of high performance swimming in sharks. II. Metabolic biochemistry of locomotor and myocardial muscle in endothermic and ectothermic sharks. J. Exp. Biol. 206, 2845–2857 (2003)
Graham, J. B., Dewar, H., Lai, N. C., Lowell, W. R. & Arce, S. M. Aspects of shark swimming performance determined using a large water tunnel. J. Exp. Biol. 151, 175–192 (1990)
Dewar, H. & Graham, J. B. Studies of tropical tuna swimming performance in a large water tunnel. I. Energetics. J. Exp. Biol. 192, 13–31 (1994)
Dickson, K. A. Unique adaptations of the metabolic biochemistry of tunas and billfishes for life in the pelagic environment. Environ. Biol. Fish. 42, 65–97 (1995)
Katz, S. L. Design of heterothermic muscle in fish. J. Exp. Biol. 205, 2251–2266 (2002)
Hulbert, L. B. & Rice, S. D. Salmon Shark, Lamna ditropis, Movements, Diet, and Abundance in the Eastern North Pacific Ocean and Prince William Sound, Alaska. Exxon Valdez Oil Spill Restoration Project Final Report (Restoration Project 02396, NOAA Fisheries Auke Bay Laboratory, Juneau, Alaska, 2002)
Rhodes, D. & Smith, R. Body temperature of the salmon shark, Lamna ditropis. J. Mar. Biol. Assoc. UK 63, 243–244 (1983)
Anderson, S. A. & Goldman, K. J. Temperature measurements from salmon sharks, Lamna ditropis, in Alaskan waters. Copeia 2001, 794–796 (2001)
Paust, B. & Smith, R. Salmon Shark Manual. The Development of a Commercial Salmon Shark, Lamna ditropis, Fishery in the North Pacific (Report No. 86–01, University of Alaska, Sea Grant College Program, Fairbanks, Alaska, 1989)
Syme, D. A. & Shadwick, R. E. Effects of longitudinal body position and swimming speed on mechanical power of deep red muscle from skipjack tuna (Katsuwonus pelamis). J. Exp. Biol. 205, 189–200 (2002)
Johnson, T. P. & Johnston, I. A. Temperature adaptation and the contractile properties of live muscle fibres from teleost fish. J. Comp. Physiol. Biochem. 161, 27–36 (1991)
Altringham, J. D. & Block, B. A. Why do tuna maintain elevated slow muscle temperatures? Power output of muscle isolated from endothermic and ectothermic fish. J. Exp. Biol. 200, 2617–2627 (1997)
Curtin, N. A. & Woledge, R. C. Power output and force-velocity relationship of live fibres from white myotomal muscle of the dogfish, Scyliorhinus canicula. J. Exp. Biol. 140, 187–197 (1988)
James, R. S., Cole, N. J., Davies, M. L. F. & Johnston, I. A. Scaling of intrinsic contractile properties and myofibrillar protein composition of fast muscle in the fish Myoxocephalus scorpius L. J. Exp. Biol. 201, 901–912 (1998)
Katz, S. L., Syme, D. A. & Shadwick, R. E. High-speed swimming: Enhanced power in yellowfin tuna. Nature 410, 770–771 (2000)
Donley, J. M., Sepulveda, C. A., Konstantinidis, P., Gemballa, S. & Shadwick, R. E. Convergent evolution in mechanical design of lamnid sharks and tunas. Nature 429, 61–65 (2004)
Acknowledgements
We thank the staff at the University of Alaska Seward Marine Center for the use of laboratory facilities, the captain and crew of the F/V Legend for their assistance in the fishing operations, and J. Valdez for logistical support. This work was supported by funding from the NSF and the University of California San Diego Academic Senate (R.E.S. and D.B.) and the NSERC (D.A.S.). Author Contributions All authors contributed equally to project planning, experimental work and writing of this Letter.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Supplementary Figure 1
In situ measures of muscle twitches. (PDF 2961 kb)
Supplementary Figure Legend
Text to accompany the above Supplementary Figure. (DOC 21 kb)
Rights and permissions
About this article
Cite this article
Bernal, D., Donley, J., Shadwick, R. et al. Mammal-like muscles power swimming in a cold-water shark. Nature 437, 1349–1352 (2005). https://doi.org/10.1038/nature04007
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature04007
This article is cited by
-
From extraocular photoreception to pigment movement regulation: a new control mechanism of the lanternshark luminescence
Scientific Reports (2020)
-
Thermal effects on red muscle contractile performance in deep-diving, large-bodied fishes
Fish Physiology and Biochemistry (2020)
-
Melanin-concentrating hormone is not involved in luminescence emission in the velvet belly lanternshark, Etmopterus spinax
Marine Biology (2019)
-
Contamination Profile of DDTs in the Shark Somniosus microcephalus from Greenland Seawaters
Bulletin of Environmental Contamination and Toxicology (2018)
-
Temperature effects on the blood oxygen affinity in sharks
Fish Physiology and Biochemistry (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.