Abstract
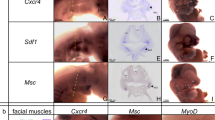
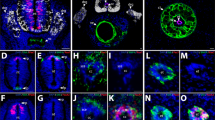
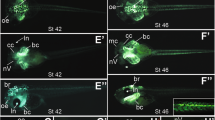
The neck and shoulder region of vertebrates has undergone a complex evolutionary history. To identify its underlying mechanisms we map the destinations of embryonic neural crest and mesodermal stem cells using Cre-recombinase-mediated transgenesis. The single-cell resolution of this genetic labelling reveals cryptic cell boundaries traversing the seemingly homogeneous skeleton of the neck and shoulders. Within this assembly of bones and muscles we discern a precise code of connectivity that mesenchymal stem cells of both neural crest and mesodermal origin obey as they form muscle scaffolds. The neural crest anchors the head onto the anterior lining of the shoulder girdle, while a Hox-gene-controlled mesoderm links trunk muscles to the posterior neck and shoulder skeleton. The skeleton that we identify as neural crest-derived is specifically affected in human Klippel–Feil syndrome, Sprengel's deformity and Arnold–Chiari I/II malformation, providing insights into their likely aetiology. We identify genes involved in the cellular modularity of the neck and shoulder skeleton and propose a new method for determining skeletal homologies that is based on muscle attachments. This has allowed us to trace the whereabouts of the cleithrum, the major shoulder bone of extinct land vertebrate ancestors, which seems to survive as the scapular spine in living mammals.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Janvier, P. Early Vertebrates (Oxford Science Publications, Oxford, 1996)
Johanson, Z. Placoderm branchial and hypobranchial muscles and origins in jawed vertebrates. J. Vert. Paleontol. 23, 735–749 (2003)
Motta, P. J. & Wilga, C. D. Environmental Biology of Fishes Vol. 60, 131–156 (Kluwer Academic, Dordrecht, 2001)
LeDouarin, N. & Kalcheim, C. The Neural Crest 2nd edn (Cambridge Univ. Press, Cambridge, 1999)
Edgeworth, F. H. The Cranial Muscles of Vertebrates (Cambridge Univ. Press, Cambridge, 1935)
Shubin, N., Tabin, C. & Carroll, S. Fossils, genes and the evolution of animal limbs. Nature 388, 639–648 (1997)
Lumsden, A., Sprawson, N. & Graham, A. Segmental origin and migration of neural crest cells in the hindbrain region of the chick embryo. Development 113, 1281–1291 (1991)
Saunders, J. W. J. The proximo-distal sequence of origin of the parts of the chick wing and the role of the ectoderm. J. Exp. Zool. 108, 363–403 (1948)
Smith, M. M. & Hall, B. K. Development and evolutionary origins of vertebrate skeletogenic and odontogenic tissues. Biol. Rev. 65, 277–373 (1990)
Smith, M. M. & Hall, B. K. in Evolutionary Biology Vol. 27 (eds Hecht, M. K., MacIntyre, R. J. & Clegg, M. T.) 387–448 (Plenum, New York, 1993)
Couly, G. F., Coltey, P. M. & LeDouarin, N. M. The triple origin of skull in higher vertebrates: a study in quail–chick chimeras. Development 114, 1–15 (1993)
Jiang, X., Iseki, S., Maxson, R. E., Sucov, H. M. & Morriss-Kay, G. M. Tissue origins and interactions in the mammalian skull vault. Dev. Biol. 241, 106–116 (2002)
Koentges, G. & Lumsden, A. G. S. Rhombencephalic neural crest segmentation is preserved throughout craniofacial ontogeny. Development 122, 3229–3242 (1996)
Huang, R. et al. Contribution of single somites to the skeleton and muscles of the occipital and cervical regions in avian embryos. Anat. Embryol. 202, 375–383 (2000)
Huang, L. F. et al. Mouse clavicular development: analysis of wild-type and cleidocranial dysplasia mutant mice. Dev. Dyn. 210, 33–40 (1997)
Hall, B. K. Development of the clavicles in birds and mammals. J. Exp. Zool. 289, 153–161 (2001)
Danielian, P. S., Muccino, D., Rowitch, D. H., Michael, S. K. & McMahon, A. P. Modification of gene activity in mouse embryos in utero by a tamoxifen-inducible form of Cre recombinase. Curr. Biol. 8, 1323–1326 (1998)
Burke, A. C., Nelson, C. E., Morgan, B. A. & Tabin, C. Hox genes and the evolution of vertebrate axial morphology. Development 121, 333–346 (1995)
Clarke, R. A., Catalan, G., Diwan, A. D. & Kearsley, J. H. Heterogeneity in Klippel-Feil syndrome: a new classification. Pediatr Radiol. 28, 967–974 (1998)
Horwitz, A. E. Congenital elevation of the scapula—Sprengel's deformity. Am. J. Orthop. Surg. 6, 260–311 (1908)
Otto, F. et al. Cbfa1, a candidate gene for cleidocranial dysplasia syndrome, is essential for osteoblast differentiation and bone development. Cell 89, 765–771 (1997)
Graham, D. I. & Lantos, P. L. Greenfield's Neuropathology 7th edn (Oxford Univ. Press, London, 2002)
Kjaer, I. & Niebuhr, E. Studies of the cranial base in 23 patients with cri-du-chat syndrome suggest a cranial developmental field involved in the condition. Am. J. Med. Genet. 82, 6–14 (1999)
Huang, R., Zhi, Q., Patel, K., Wilting, J. & Christ, B. Dual origin and segmental organisation of the avian scapula. Development 127, 3789–3794 (2000)
Alvares, L. E. et al. Intrinsic, Hox-dependent cues determine the fate of skeletal muscle precursors. Dev. Cell 5, 379–390 (2003)
Schweitzer, R. et al. Analysis of the tendon cell fate using Scleraxis, a specific marker for tendons and ligaments. Development 128, 3855–3866 (2001)
Baylies, M. K. et al. Myogenesis: a view from Drosophila. Cell 93, 921–927 (1998)
Takio, Y. et al. Lamprey Hox genes and the evolution of jaws. Nature 429, 262–263 (2004)
Barrow, J. R. & Capecchi, M. R. Compensatory defects associated with mutations in Hoxa1 restore normal palatogenesis to Hoxa2 mutants. Development 126, 5011–5026 (1999)
Smith, A. et al. The EphA4 and EphB1 receptor tyrosine kinases and ephrin-B2 ligand regulate targeted migration of branchial neural crest cells. Curr. Biol. 7, 561–570 (1997)
Selleri, L. et al. Requirement for Pbx1 in skeletal patterning and programming chondrocyte proliferation and differentiation. Development 128, 3543–3557 (2001)
Dietrich, S. & Gruss, P. Undulated phenotypes suggest a role of Pax-1 for the development of vertebral and extravertebral structures. Dev. Biol. 167, 529–548 (1995)
Peters, H. et al. Pax1 and Pax9 synergistically regulate vertebral column development. Development 126, 5399–5408 (1999)
Prols, F. et al. The role of Emx2 during scapula formation. Dev. Biol. 275, 315–324 (2004)
Jarvik, E. Basic Structure and Evolution of Vertebrates Vol. 1 (Academic, London, 1980)
Shearman, R. M. Growth of the pectoral girdle of the Leopard Frog Rana pipiens (Anura: Ranidae). J. Morphol. 264, 94–104 (2005)
Schoch, R. R. Comparative osteology of Mastodonsaurus giganteus (Jaeger, 1828) from the Middle Triassic (Lettenkeuper: Longobardian) of Germany (Baden-Württemberg, Bayern, Thüringen). Stuttg. Beitr. Naturk. B 278, 1–175 (1999)
Sumida, S. S. in Amniote Origins (eds Sumida, S. S. & Martin, K. L. M.) 353–398 (Academic, San Diego, 1997)
Lebedev, O. A. in The Second Gross Symposium ‘Advances in Palaeoichthyology’ (ed. Luksevics, E.) 79–98 (Acta Universitatis Latviensis 679, 2005).
Clack, J. A. & Finney, S. M. Pederpes finneyae, an articulated tetrapod from the Tournaisian of Western Scotland. J. Syst. Palaeont. 2, 311–346 (2005)
Reisz, R. R., Berman, D. S. & Scott, D. The anatomy and relationships of the Lower Permian reptile Araeoscelis. J. Vertebr. Paleontol. 4, 57–67 (1984)
Jaekel, O. Die Wirbeltierfunde aus dem Keuper von Halberstadt. Paläont. Z. 2, 88–214 (1915–16)
Joyce, W. The presence of cleithra in the primitive turtle Kayentachelys aprix. J. Vert. Paleontol.. 23 (suppl.), 66A (2003)
Ferguson, C. A. & Graham, A. Redefining the head–trunk interface for the neural crest. Dev. Biol. 269, 70–80 (2004)
Soriano, P. Generalized lacZ expression with the ROSA26 Cre reporter strain. Nature Genet. 21, 70–71 (1999)
Mao, X. et al. Activation of EGFP expression by Cre-mediated excision in a new ROSA26 reporter mouse strain. Blood 97, 324–326 (2001)
Condie, B. G. & Capecchi, M. R. Mice with targeted disruptions in the paralogous genes Hoxa-3 and Hoxd-3 reveal synergistic interactions. Nature 370, 304–307 (1994)
Zhang, F. et al. Elements both 5′ and 3′ to the murine Hoxd4 gene establish anterior borders of expression in mesoderm and neuroectoderm. Mech. Dev. 67, 49–58 (1997)
Loonstra, A. et al. Growth inhibition and DNA damage induced by Cre recombinase in mammalian cells. Proc. Natl Acad. Sci. USA 98, 9209–9214 (2001)
Winterbottom, R. A descriptive synonymy of the striated muscles of the teleostei. Proc. Acad. Nat. Sci. Phila. 125, 225–317 (1974)
Acknowledgements
We thank A. Lumsden for help with a complex manuscript; P. Soriano and S. Orkin for providing Cre reporters; and A. West, G. Felsenfeld and J. Green for advice on insulators and plasmids. This work was funded by the BBSRC (G.K., P.E.A.), the Wellcome Trust (G.K., W.D.R.), the MRC UK (W.D.R.), the Swedish Research Council (P.E.A.), the NIH (A.P.M.) and WIBR-UCL (G.K.). G.K. and T.M. were long-term postdoctoral fellows of HFSPO. G.K. thanks S. Moncada for support in establishing a new laboratory.Author Contributions T.M. and P.E.A. contributed equally to this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Supplementary Figure S1
This figure details mouse mutant neck phenotypes. (PDF 147 kb)
Supplementary Methods S1
This file contains Supplementary Methods and analysis. (PDF 343 kb)
Supplemental Methods S2
New methodology for determining neck/shoulder homologies. Also includes additional references. (PDF 46 kb)
Rights and permissions
About this article
Cite this article
Matsuoka, T., Ahlberg, P., Kessaris, N. et al. Neural crest origins of the neck and shoulder. Nature 436, 347–355 (2005). https://doi.org/10.1038/nature03837
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature03837
This article is cited by
-
Associations across 22 dental and craniovertebral anomalies or variations, sagittal skeletal relationships, and vertical growth patterns: a comprehensive epidemiological study of 43 dentoskeletal traits
BMC Oral Health (2023)
-
Erythropoietin regulates developmental myelination in the brain stimulating postnatal oligodendrocyte maturation
Scientific Reports (2023)
-
Fossil evidence for a pharyngeal origin of the vertebrate pectoral girdle
Nature (2023)
-
Targeted deletion of the RNA-binding protein Caprin1 leads to progressive hearing loss and impairs recovery from noise exposure in mice
Scientific Reports (2022)
-
Molecular and functional heterogeneity in dorsal and ventral oligodendrocyte progenitor cells of the mouse forebrain in response to DNA damage
Nature Communications (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.