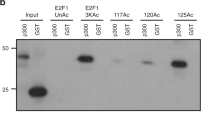
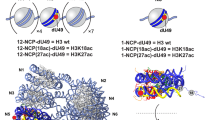
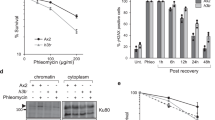
Abstract
DNA breaks are extremely harmful lesions that need to be repaired efficiently throughout the genome. However, the packaging of DNA into nucleosomes is a significant barrier to DNA repair, and the mechanisms of repair in the context of chromatin are poorly understood1. Here we show that lysine 56 (K56) acetylation is an abundant modification of newly synthesized histone H3 molecules that are incorporated into chromosomes during S phase. Defects in the acetylation of K56 in histone H3 result in sensitivity to genotoxic agents that cause DNA strand breaks during replication. In the absence of DNA damage, the acetylation of histone H3 K56 largely disappears in G2. In contrast, cells with DNA breaks maintain high levels of acetylation, and the persistence of the modification is dependent on DNA damage checkpoint proteins. We suggest that the acetylation of histone H3 K56 creates a favourable chromatin environment for DNA repair and that a key component of the DNA damage response is to preserve this acetylation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Peterson, C. L. & Côté, J. Cellular machineries for chromosomal DNA repair. Genes Dev. 18, 602–616 (2004)
Kuo, M. H. et al. Transcription-linked acetylation by Gcn5p of histones H3 and H4 at specific lysines. Nature 383, 269–272 (1996)
Sklenar, A. R. & Parthun, M. R. Characterization of yeast histone H3-specific type B histone acetyltransferases identifies an ADA2-independent Gcn5p activity. BMC Biochem. 5, 1–12 (2004)
Ye, J. et al. Histone H4 lysine 91 acetylation: A core domain modification associated with chromatin assembly. Mol. Cell 18, 123–130 (2005)
Verreault, A., Kaufman, P. D., Kobayashi, R. & Stillman, B. Nucleosome assembly by a complex of CAF-1 and acetylated histones H3/H4. Cell 87, 95–104 (1996)
Gunjan, A. & Verreault, A. A Rad53 kinase-dependent surveillance mechanism that regulates histone protein levels in S. cerevisiae. Cell 115, 537–549 (2003)
Downs, J. A., Lowndes, N. F. & Jackson, S. P. A role for Saccharomyces cerevisiae histone H2A in DNA repair. Nature 408, 1001–1004 (2000)
Redon, C. et al. Yeast histone 2A serine 129 is essential for the efficient repair of checkpoint-blind DNA damage. EMBO Rep. 4, 678–684 (2003)
Pilch, D. R. et al. Characteristics of γ-H2AX foci at DNA double-strand break sites. Biochem. Cell Biol. 81, 123–129 (2003)
Bird, A. W. et al. Acetylation of histone H4 by Esa1 is required for DNA double-strand break repair. Nature 419, 411–415 (2002)
Haber, J. E. Partners and pathways: repairing a double-strand break. Trends Genet. 16, 259–264 (2000)
Pommier, Y. et al. Repair of and checkpoint response to topoisomerase I-mediated DNA damage. Mutat. Res. 532, 173–203 (2003)
Hsiang, Y. H., Lihou, M. G. & Liu, L. F. Arrest of replication forks by drug-stabilized topoisomerase I-DNA cleavable complexes as a mechanism of cell killing by camptothecin. Cancer Res. 49, 5077–5082 (1989)
Nitiss, J. L. & Wang, J. C. Mechanisms of cell killing by drugs that trap covalent complexes between DNA topoisomerases and DNA. Mol. Pharmacol. 50, 1095–1102 (1996)
Tercero, J. A., Longhese, M. P. & Diffley, J. F. X. A central role for DNA replication forks in checkpoint activation and response. Mol. Cell 11, 1323–1326 (2003)
Davey, C. A., Sargent, D. F., Luger, K., Maeder, A. W. & Richmond, T. J. Solvent mediated interactions in the structure of the nucleosome core particle at 1.9 Å resolution. J. Mol. Biol. 319, 1097–1113 (2002)
Norton, V. G., Imai, B. S., Yau, P. & Bradbury, E. M. Histone acetylation reduces nucleosome core particle linking number change. Cell 57, 449–457 (1989)
Wechser, M. A., Kladde, M. P., Alfieri, J. A. & Peterson, C. L. Effects of Sin- versions of histone H4 on yeast chromatin structure and function. EMBO J. 16, 2086–2095 (1997)
Sogo, J. M., Stahl, H., Koller, T. & Knippers, R. Structure of replicating simian virus 40 minichromosomes. The replication fork, core histone segregation and terminal structures. J. Mol. Biol. 189, 189–204 (1986)
Downs, J. A. et al. Binding of chromatin-modifying activities to phosphorylated histone H2A at DNA damage sites. Mol. Cell 16, 979–990 (2004)
Kushnirov, V. V. Rapid and reliable protein extraction from yeast. Yeast 16, 857–860 (2000)
Acknowledgements
We thank W. Bonner, M. Christman, J. Diffley, L. Drury, P. Fitzjohn, H. Nash, A. Kristjuhan, D. Lyon, C. Redon, D. Stillman, J. Svejstrup and S. Tanaka for reagents, and A. Castillo and A. Gunjan for critical reading of the manuscript. This work was funded by Cancer Research UK and the International Association for Cancer Research. H.M. was supported by postdoctoral fellowships from the NAITO Foundation and Cancer Research UK.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Supplementary Notes
This contains Supplementary Methods, Supplementary Table 1, Supplementary References, and Legends for Supplementary Figures S1-S9. (DOC 80 kb)
Rights and permissions
About this article
Cite this article
Masumoto, H., Hawke, D., Kobayashi, R. et al. A role for cell-cycle-regulated histone H3 lysine 56 acetylation in the DNA damage response. Nature 436, 294–298 (2005). https://doi.org/10.1038/nature03714
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature03714
This article is cited by
-
Histone H3 N-terminal tail provides tolerance to tartrazine induced stress in Saccharomyces cerevisiae
Biologia (2024)
-
Oxidative replication stress induced by long-term exposure to hydroxyurea in root meristem cells of Vicia faba
Plant Cell Reports (2024)
-
Epigenetic modifications evidenced by isolation of proteins on nascent DNA and immunofluorescence in hydroxyurea-treated root meristem cells of Vicia faba
Planta (2023)
-
NAA60 (HAT4): the newly discovered bi-functional Golgi member of the acetyltransferase family
Clinical Epigenetics (2022)
-
Mechanisms of chromatin-based epigenetic inheritance
Science China Life Sciences (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.