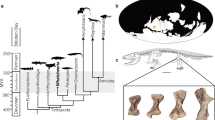
Abstract
Cyclical growth marks in cortical bone, deposited before attainment of adult body size, reflect osteogenetic changes caused by annual rhythms and are a general phenomenon in non-avian ectothermic and endothermic tetrapods1. However, the growth periods of ornithurines (the theropod group including all modern birds) are usually apomorphically shortened to less than a year2,3, so annual growth marks are almost unknown in this group4,5,6. Here we show that cortical growth marks are frequent in long bones of New Zealand's moa (Aves: Dinornithiformes), a recently extinct ratite order. Moa showed the exaggerated K-selected life-history strategy formerly common in the New Zealand avifauna, and in some instances took almost a decade to attain skeletal maturity. This indicates that reproductive maturity in moa was extremely delayed relative to all extant birds. The two presently recognized moa families (Dinornithidae and Emeidae) also showed different postnatal growth rates, which were associated with their relative differences in body size. Both species of giant Dinornis moa attained their massive stature (up to 240 kg live mass) by accelerating their juvenile growth rate compared to the smaller emeid moa species, rather than by extending the skeletal growth period.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Hall, B. K. (ed.) Bone Vol. 7 (CRC Press, Boca Raton, 1993)
Padian, K., de Ricqlès, A. & Horner, J. R. Dinosaurian growth rates and bird origins. Nature 412, 405–408 (2001)
Davies, S. J. J. F. Ratites and Tinamous (Oxford Univ. Press, Oxford, 2002)
Chinsamy, A. in Mesozoic Birds. Above the Heads of Dinosaurs (eds Chiappe, L. M. & Witmer, L. M.) 421–431 (Univ. of California Press, Berkeley, 2002)
de Ricqlès, A., Padian, K. & Horner, J. R. in New Perspectives on the Origin and Early Evolution of Birds. Proceedings of the International Symposium in Honor of John H. Ostrom (eds Gauthier, J. & Gall, L. F.) 411–426 (Peabody Museum of Natural History, Yale Univ., New Haven, 2001)
Starck, J. M. & Chinsamy, A. Bone microstructure and developmental plasticity in birds and other dinosaurs. J. Morphol. 254, 232–246 (2002)
de Margerie, E. et al. Assessing a relationship between bone microstructure and growth rate: a fluorescent labelling study in the king penguin chick (Aptenodytes patagonicus). J. Exp. Biol. 207, 869–879 (2004)
Enlow, D. H. & Brown, S. O. A comparative histological study of fossil and recent bone tissues. Part II. Tex. J. Sci. 9, 186–214 (1957)
Houde, P. Histological evidence for the systematic position of Hesperornis (Odontornithes: Hesperornithiformes). Auk 104, 125–129 (1987)
Klomp, N. I. & Furness, R. W. A technique which may allow accurate determination of the age of adult birds. Ibis 134, 245–249 (1992)
Broughton, J. M., Rampton, D. & Holanda, K. A test of an osteologically based age determination technique in the double-crested cormorant Phalacrocorax auritus. Ibis 144, 143–146 (2002)
Holdaway, R. N. & Jacomb, C. Rapid extinction of the moas (Aves: Dinornithiformes): model, test and implications. Science 287, 2250–2254 (2000)
Lydekker, R. Catalogue of the Fossil Birds in the British Museum (Natural History) (British Museum (Nat. Hist.), London, 1891)
Bunce, M. et al. Extreme reversed sexual size dimorphism in the extinct New Zealand moa Dinornis. Nature 425, 172–175 (2003)
Huynen, L., Millar, C. D., Scofield, R. P. & Lambert, D. M. Nuclear DNA sequences detect species limits in ancient moa. Nature 425, 175–178 (2003)
Worthy, T. H. & Holdaway, R. N. The Lost World of the Moa (Indiana Univ. Press, Bloomington, 2002)
Chinsamy, A. Histological perspectives on growth in the birds Struthio camelus and Sagittarius serpentarius. Courier Forsch.-Inst. Senckenberg 181, 317–323 (1995)
Chinsamy, A., Rich, T. & Vickers-Rich, P. Polar dinosaur bone histology. J. Vert. Paleontol. 18, 385–390 (1998)
Castanet, J., Curry Rogers, K., Cubo, J. & Boisard, J.-J. Periosteal bone growth rates in extant ratites (ostriche and emu). Implications for assessing growth in dinosaurs. Comptes Rendus Acad. Sci. Paris, Sciences de la vie 323, 543–550 (2000)
Castanet, J., Grandin, A., Abourachid, A. & de Ricqlès, A. Expression de la dynamique de croissance dans la structure de l'os périostique chez Anas platyrhynchos. Comptes Rendus Acad. Sci. Paris, Sciences de la vie 319, 301–308 (1996)
Amprino, R. La structure du tissue osseux envisage comme expression de differences dans la vitesse de l'accroissement. Arch. Biol. (Paris) 58, 315–330 (1947)
Padian, K., Horner, J. R. & de Ricqlès, A. Growth in small dinosaurs and pterosaurs: the evolution of archosaurian growth strategies. J. Vert. Paleontol. 24, 555–571 (2004)
Erickson, G. M. et al. Gigantism and comparative life-history parameters of tyrannosaurid dinosaurs. Nature 430, 772–775 (2004)
Sander, P. M. et al. Adaptive radiation in sauropod dinosaurs: bone histology indicates rapid evolution of giant body size through acceleration. Org. Div. Evol. 4, 165–173 (2004)
Cracraft, J. Phylogeny and evolution of the ratite birds. Ibis 116, 494–521 (1974)
Cooper, A. et al. Complete mitochondrial genome sequences of two extinct moas clarify ratite evolution. Nature 409, 704–707 (2001)
Bennett, P. M. & Owens, I. P. F. Evolutionary Ecology of Birds (Oxford Univ. Press, Oxford, 2002)
Owens, I. P. F. & Bennett, P. M. Ecological basis of extinction risk in birds: habitat loss versus human persecution and introduced predators. Proc. Natl Acad. Sci. USA 97, 12144–12148 (2000)
Johnson, C. N. Determinants of loss of mammal species during the Late Quaternary ‘megafauna’ extinctions: life history and ecology, but not body size. Proc. R. Soc. Lond. B 269, 2221–2227 (2002)
Horner, J. R., de Ricqlès, A. & Padian, K. Variation in dinosaur skeletochronology indicators: implications for age assessment and physiology. Paleobiology 25, 295–304 (1999)
Acknowledgements
We are grateful to R. P. Scofield, H. A. Schlumpf, N. Carson, A. J. D. Tennyson, J. A. Bartle, M. Bunce, T. H. Worthy, L. J. Shorey, T. Robinson, R. Spiers, M. Giesen, S. Veldhuizen, A. Chinsamy-Turan, J. Horner, P. Houde and K. Padian. Funding was provided by a Banks Alecto postdoctoral fellowship from the Royal Society of London (S.T.T.) and the New Zealand Foundation for Research, Science and Technology (R.N.H.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Turvey, S., Green, O. & Holdaway, R. Cortical growth marks reveal extended juvenile development in New Zealand moa. Nature 435, 940–943 (2005). https://doi.org/10.1038/nature03635
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature03635
This article is cited by
-
Osteohistology of the Antarctic penguin Pygoscelis adeliae (Aves, Sphenisciformes): definitive evidence of medullary bone
Polar Biology (2023)
-
Variability of growth pattern observed in Metoposaurus krasiejowensis humeri and its biological meaning
Journal of Iberian Geology (2018)
-
Bone histology sheds new light on the ecology of the dodo (Raphus cucullatus, Aves, Columbiformes)
Scientific Reports (2017)
-
An extremely low-density human population exterminated New Zealand moa
Nature Communications (2014)
-
Insight into the growth dynamics and systematic affinities of the Late Cretaceous Gargantuavis from bone microstructure
Naturwissenschaften (2014)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.