Abstract
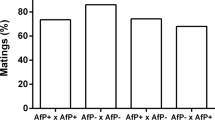
Wolbachia is a common maternally inherited bacterial symbiont able to induce crossing sterilities known as cytoplasmic incompatibility (CI) in insects1,2. Wolbachia-modified sperm are unable to complete fertilization of uninfected ova, but a rescue function allows infected eggs to develop normally. By providing a reproductive advantage to infected females, Wolbachia can rapidly invade uninfected populations3, and this could provide a mechanism for driving transgenes through pest populations4,5. CI can also occur between Wolbachia-infected populations and is usually associated with the presence of different Wolbachia strains1. In the Culex pipiens mosquito group (including the filariasis vector C. quinquefasciatus) a very unusual degree of complexity of Wolbachia-induced crossing-types has been reported, with partial or complete CI that can be unidirectional or bidirectional6,7,8,9,10,11, yet no Wolbachia strain variation was found11. Here we show variation between incompatible Culex strains in two Wolbachia ankyrin repeat-encoding genes associated with a prophage region, one of which is sex-specifically expressed in some strains, and also a direct effect of the host nuclear genome on CI rescue.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
O'Neill, S. L., Hoffmann, A. A. & Werren, J. H. (eds) Influential Passengers: Inherited Microorganisms and Arthropod Reproduction (Oxford Univ. Press, Oxford, 1997)
Werren, J. H. Biology of Wolbachia. Annu. Rev. Entomol. 42, 587–609 (1997)
Turelli, M. & Hoffmann, A. A. Rapid spread of an inherited incompatibility factor in California Drosophila. Nature 353, 440–442 (1991)
Sinkins, S. P. & O'Neill, S. L. in Insect Transgenesis: Methods and Applications (eds Handler, A. M. & James, A. A.) 271–288 (CRC, Florida, 2000)
Dobson, S. L. Reversing Wolbachia-based population replacement. Trends Parasitol. 19, 128–133 (2003)
Laven, H. in Genetics of Insect Vectors of Disease (eds Wright, R. & Pal, R.) 251–275 (Elsevier, Amsterdam, 1967)
Barr, A. R. Cytoplasmic incompatibility in natural populations of a mosquito, Culex pipiens L. Nature 283, 71–72 (1980)
Irving-Bell, R. J. Cytoplasmic incompatibility within and between Culex molestus and Cx. quinquefasciatus (Diptera: Culicidae). J. Med. Entomol. 20, 44–48 (1983)
O'Neill, S. L. & Paterson, H. E. Crossing type variability associated with cytoplasmic incompatibility in Australian populations of the mosquito Culex quinquefasciatus Say. Med. Vet. Entomol. 6, 209–216 (1992)
Magnin, M., Pasteur, N. & Raymond, M. Multiple incompatibilities within populations of Culex pipiens L. in southern France. Genetica 74, 125–130 (1987)
Guillemaud, T., Pasteur, N. & Rousset, F. Contrasting levels of variability between cytoplasmic genomes and incompatibility types in the mosquito Culex pipiens. Proc. R. Soc. Lond. B 264, 245–251 (1997)
Zhou, W., Rousset, F. & O'Neill, S. L. Phylogeny and PCR-based classification of Wolbachia strains using WSP gene sequences. Proc. R. Soc. Lond. B 265, 509–515 (1998)
Wu, M. et al. Phylogenomics of the reproductive parasite Wolbachia pipientis wMel: a streamlined genome overrun by mobile genetic elements. PLOS Biol. 2, E69–E83 (2004)
Elfring, L. K. et al. Drosophila PLUTONIUM protein is a specialized cell cycle regulator required at the onset of embryogenesis. Mol. Biol. Cell 8, 583–593 (1997)
Caturegli, P. et al. ankA: an Ehrlichia phagocytophila group gene encoding a cytoplasmic protein antigen with ankyrin repeats. Infect. Immun. 68, 5277–5283 (2000)
Callaini, G., Dallai, R. & Riparbelli, M. G. Wolbachia-induced delay of paternal chromatin condensation does not prevent maternal chromosomes from entering anaphase in incompatible crosses of Drosophila simulans. J. Cell Sci. 110, 271–280 (1997)
Tram, U. & Sullivan, W. Role of delayed nuclear envelope breakdown and mitosis in Wolbachia-induced cytoplasmic incompatibility. Science 296, 1124–1126 (2002)
Song, S. W., Zhao, T. Y., Dong, Y. D. & Lu, B. L. Hybridization of Culex pipiens complex and their relationship with the infections of Wolbachia in China. Acta Entomol. Sin. 45, 705–710 (2002)
Masui, S., Kamoda, S., Sasaki, T. & Ishikawa, H. Distribution and evolution of bacteriophage WO in Wolbachia, the endosymbiont causing sexual alterations in arthropods. J. Mol. Evol. 51, 491–497 (2000)
Masui, S. et al. Bacteriophage WO and virus-like particles in Wolbachia, an endosymbiont of arthropods. Biochem. Biophys. Res. Commun. 283, 1099–1104 (2001)
Wright, J. D., Sjostrand, F. S., Portaro, J. K. & Barr, A. R. The ultrastructure of the Rickettsia-like microorganism Wolbachia pipientis and associated virus-like bodies in the mosquito Culex pipiens. J. Ultrastruct. Res. 63, 79–85 (1978)
Rousset, F., Raymond, M. & Kjelberg, F. Cytoplasmic incompatibilities in the mosquito Culex pipiens: how to explain a cytotype polymorphism? J. Evol. Biol. 4, 69–81 (1991)
Turelli, M. Evolution of incompatibility-inducing microbes and their hosts. Evolution 48, 1500–1513 (1994)
Raymond, M., Callaghan, A., Fort, P. & Pasteur, N. Worldwide migration of amplified insecticide resistance genes in mosquitoes. Nature 350, 151–153 (1991)
Olson, K. E. et al. Genetically engineered resistance to dengue-2 virus transmission in mosquitoes. Science 272, 884–886 (1996)
Ito, J., Ghosh, A., Moreira, L. A., Wimmer, E. A. & Jacobs-Lorena, M. Transgenic anopheline mosquitoes impaired in transmission of a malaria parasite. Nature 417, 452–455 (2002)
Sinkins, S. P. & Godfray, H. C. J. Use of Wolbachia to drive nuclear transgenes through insect populations. Proc. R. Soc. Lond. B 271, 1421–1426 (2004)
Sanogo, Y. O. & Dobson, S. L. Molecular discrimination of Wolbachia in the Culex pipiens complex: Evidence for variable bacteriophage hyperparasitism. Insect Mol. Biol. 13, 365–369 (2004)
Williams, E. H., Fields, S. & Saul, G. B. Transfer of incompatibility factors between stocks of Nasonia vitripennis. J. Invert. Pathol. 61, 206–210 (1993)
Collins, F. H. et al. A ribosomal RNA gene probe differentiates member species of the Anopheles gambiae complex. Am. J. Trop. Med. Hyg. 37, 37–41 (1987)
Acknowledgements
We thank S. O'Neill for Wolbachia genomics input, and S. Song for providing Culex material. This research was supported by grants from the Wellcome Trust, and the UNDP/World Bank/WHO Special Programme for Research and Training in Tropical Diseases (TDR).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Sequences have been deposited with NCBI GenBank under accession numbers DQ000469– DQ000472. Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Sinkins, S., Walker, T., Lynd, A. et al. Wolbachia variability and host effects on crossing type in Culex mosquitoes. Nature 436, 257–260 (2005). https://doi.org/10.1038/nature03629
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature03629
This article is cited by
-
Wolbachia infection dynamics in a natural population of the pear psyllid Cacopsylla pyri (Hemiptera: Psylloidea) across its seasonal generations
Scientific Reports (2022)
-
Wolbachia prevalence in the vector species Culex pipiens and Culex torrentium in a Sindbis virus-endemic region of Sweden
Parasites & Vectors (2021)
-
Wolbachia in mosquitoes from the Central Valley of California, USA
Parasites & Vectors (2020)
-
Diverse novel resident Wolbachia strains in Culicine mosquitoes from Madagascar
Scientific Reports (2018)
-
Culex pipiens and Culex restuans mosquitoes harbor distinct microbiota dominated by few bacterial taxa
Parasites & Vectors (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.