Abstract
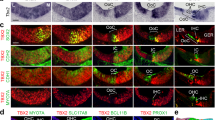
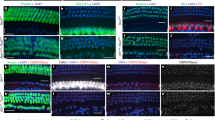
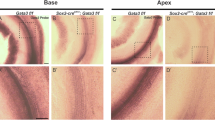
Sensory hair cells and their associated non-sensory supporting cells in the inner ear are fundamental for hearing and balance. They arise from a common progenitor1, but little is known about the molecular events specifying this cell lineage. We recently identified two allelic mouse mutants, light coat and circling (Lcc) and yellow submarine (Ysb), that show hearing and balance impairment2. Lcc/Lcc mice are completely deaf, whereas Ysb/Ysb mice are severely hearing impaired2. We report here that inner ears of Lcc/Lcc mice fail to establish a prosensory domain and neither hair cells nor supporting cells differentiate, resulting in a severe inner ear malformation, whereas the sensory epithelium of Ysb/Ysb mice shows abnormal development with disorganized and fewer hair cells. These phenotypes are due to the absence (in Lcc mutants) or reduced expression (in Ysb mutants) of the transcription factor SOX2, specifically within the developing inner ear. SOX2 continues to be expressed in the inner ears of mice lacking Math1 (also known as Atoh1 and HATH1), a gene essential for hair cell differentiation, whereas Math1 expression is absent in Lcc mutants, suggesting that Sox2 acts upstream of Math1.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Fekete, D. M., Muthukumar, S. & Karagogeos, D. Hair cells and supporting cells share a common progenitor in the avian inner ear. J. Neurosci. 18, 7811–7821 (1998)
Dong, S. et al. Circling, deafness, and yellow coat displayed by yellow submarine (Ysb) and light coat and circling (Lcc) mice with mutations on chromosome 3. Genomics 79, 777–784 (2002)
Eddison, M., Le, R. I. & Lewis, J. Notch signaling in the development of the inner ear: lessons from Drosophila. Proc. Natl Acad. Sci. USA 97, 11692–11699 (2000)
Jan, Y. N. & Jan, L. Y. Neuronal cell fate specification in Drosophila. Curr. Opin. Neurobiol. 4, 8–13 (1994)
Bermingham, N. A. et al. Math1: an essential gene for the generation of inner ear hair cells. Science 284, 1837–1841 (1999)
Chen, P., Johnson, J. E., Zoghbi, H. Y. & Segil, N. The role of Math1 in inner ear development: Uncoupling the establishment of the sensory primordium from hair cell fate determination. Development 129, 2495–2505 (2002)
Lyon, M. F., Phillips, R. J. & Fisher, G. Dose-response curves for radiation-induced gene mutations in mouse oocytes and their interpretation. Mutat. Res. 63, 161–173 (1979)
Wood, H. B. & Episkopou, V. Comparative expression of the mouse Sox1, Sox2 and Sox3 genes from pre-gastrulation to early somite stages. Mech. Dev. 86, 197–201 (1999)
Uchikawa, M., Kamachi, Y. & Kondoh, H. Two distinct subgroups of Group B Sox genes for transcriptional activators and repressors: their expression during embryonic organogenesis of the chicken. Mech. Dev. 84, 103–120 (1999)
Avilion, A. A. et al. Multipotent cell lineages in early mouse development depend on SOX2 function. Genes Dev. 17, 126–140 (2003)
Knowlton, V. Y. Correlation of the development of membranous and bony labyrinths, acoustics ganglia, nerves, and brain centers of the chick embryos. J. Morphol. 121, 179–208 (1967)
Cole, L. K. et al. Sensory organ generation in the chicken inner ear: contributions of bone morphogenetic protein 4, serrate1, and lunatic fringe. J. Comp. Neurol. 424, 509–520 (2000)
Zappone, M. V. et al. Sox2 regulatory sequences direct expression of a (beta)-geo transgene to telencephalic neural stem cells and precursors of the mouse embryo, revealing regionalization of gene expression in CNS stem cells. Development 127, 2367–2382 (2000)
Uchikawa, M., Ishida, Y., Takemoto, T., Kamachi, Y. & Kondoh, H. Functional analysis of chicken Sox2 enhancers highlights an array of diverse regulatory elements that are conserved in mammals. Dev. Cell 4, 509–519 (2003)
Cheah, K. S. et al. Human COL2A1-directed SV40 T antigen expression in transgenic and chimeric mice results in abnormal skeletal development. J. Cell Biol. 128, 223–237 (1995)
Cai, J. et al. Properties of a fetal multipotent neural stem cell (NEP cell). Dev. Biol. 251, 221–240 (2002)
Buescher, M., Hing, F. S. & Chia, W. Formation of neuroblasts in the embryonic central nervous system of Drosophila melanogaster is controlled by SoxNeuro. Development 129, 4193–4203 (2002)
Overton, P. M., Meadows, L. A., Urban, J. & Russell, S. Evidence for differential and redundant function of the Sox genes Dichaete and SoxN during CNS development in Drosophila. Development 129, 4219–4228 (2002)
Kishi, M. et al. Requirement of Sox2-mediated signaling for differentiation of early Xenopus neuroectoderm. Development 127, 791–800 (2000)
Chen, P. & Segil, N. p27(Kip1) links cell proliferation to morphogenesis in the developing organ of Corti. Development 126, 1581–1590 (1999)
Zheng, J. L. & Gao, W. Q. Overexpression of Math1 induces robust production of extra hair cells in postnatal rat inner ears. Nature Neurosci. 3, 580–586 (2000)
Woods, C., Montcouquiol, M. & Kelley, M. W. Math1 regulates development of the sensory epithelium in the mammalian cochlea. Nature Neurosci. 7, 1310–1318 (2004)
Chang, W., Brigande, J. V., Fekete, D. M. & Wu, D. K. The development of semicircular canals in the inner ear: role of FGFs in sensory cristae. Development 131, 4201–4211 (2004)
Pirvola, U. et al. FGFR1 is required for the development of the auditory sensory epithelium. Neuron 35, 671–680 (2002)
Pauley, S. et al. Expression and function of FGF10 in mammalian inner ear development. Dev. Dyn. 227, 203–215 (2003)
Kiernan, A. E. et al. The Notch ligand Jagged1 is required for inner ear sensory development. Proc. Natl Acad. Sci. USA 98, 3873–3878 (2001)
Tsai, H. et al. The mouse slalom mutant demonstrates a role for Jagged1 in neuroepithelial patterning in the organ of Corti. Hum. Mol. Genet. 10, 507–512 (2001)
Morsli, H., Choo, D., Ryan, A., Johnson, R. & Wu, D. K. Development of the mouse inner ear and origin of its sensory organs. J. Neurosci. 18, 3327–3335 (1998)
Li, H., Liu, H. & Heller, S. Pluripotent stem cells from the adult mouse inner ear. Nature Med. 9, 1293–1299 (2003)
Li, H., Roblin, G., Liu, H. & Heller, S. Generation of hair cells by stepwise differentiation of embryonic stem cells. Proc. Natl Acad. Sci. USA 100, 13495–13500 (2003)
Acknowledgements
This work was supported by grants from the Research Grants Council (Hong Kong) to K.S.E.C., by the UK Medical Research Council, Defeating Deafness, the European Commission and the Louis Jeantet Foundation. We thank C. L. So and M. Chan for mouse care and research assistance, and H. Zoghbi for the gift of Math1 null mice. We are also grateful to P. Tam, D. Loebel, V. Episkopou, J. Brigande and the RLB laboratory for critical comments and discussion.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Supplementary Figure S1
This figure shows the sensory defects in both the cochlea and vestibule of Ysb/Ysb inner ears at P0-P1 identified by scanning electron microscopy. (PDF 603 kb)
Supplementary Figure S2
This figure shows that loss/downregulation of SOX2 expression correlates with absence/irregular hair cell differentiation in Lcc/Lcc and Ysb/Ysb vestibules, respectively as determined by immunohistochemistry at E16.5 using antibodies to SOX2 and MYO7A. (PDF 215 kb)
Supplementary Figure S3
This figure shows that hair cell markers Pou4f3 and GFI1 are expressed in Ysb/Ysb cochlea hair cells at E16.5. The gene expression also shows the abnormal hair cell differentiation in Ysb/Ysb. (PDF 66 kb)
Supplementary Figure S4
This figure shows the genomic DNA sequence that is amplified during the PCR genotyping of Lcc mutants. The naturally occurring polymorphism (SINE) in Lcc wild type DNA which does not segregate with the Lcc allele is highlighted as are the primer sequences. (PDF 167 kb)
Supplementary Table S1
This table shows the raw data from the endocochlear potential measurements of each mouse mutant. The results show that most mutants mutants have an endocochlear potential within the normal range. (DOC 24 kb)
Supplementary Figure Legends
Legends to accompany the above Supplementary Figures and Supplementary Table. (DOC 25 kb)
Rights and permissions
About this article
Cite this article
Kiernan, A., Pelling, A., Leung, K. et al. Sox2 is required for sensory organ development in the mammalian inner ear. Nature 434, 1031–1035 (2005). https://doi.org/10.1038/nature03487
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature03487
This article is cited by
-
Stem Cell-Based Hair Cell Regeneration and Therapy in the Inner Ear
Neuroscience Bulletin (2024)
-
Integrated scRNAseq analyses of mouse cochlear supporting cells reveal the involvement of Ezh2 in hair cell regeneration
Molecular Biology Reports (2024)
-
Early radial positional information in the cochlea is optimized by a precise linear BMP gradient and enhanced by SOX2
Scientific Reports (2023)
-
Gata3 is required in late proneurosensory development for proper sensory cell formation and organization
Scientific Reports (2023)
-
Early development of the cochlea of the common marmoset, a non-human primate model
Neural Development (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.