Abstract
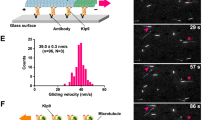
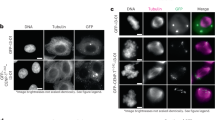
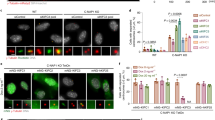
For high-fidelity chromosome segregation, kinetochores must be properly captured by spindle microtubules, but the mechanisms underlying initial kinetochore capture have remained elusive. Here we visualized individual kinetochore–microtubule interactions in Saccharomyces cerevisiae by regulating the activity of a centromere. Kinetochores are captured by the side of microtubules extending from spindle poles, and are subsequently transported poleward along them. The microtubule extension from spindle poles requires microtubule plus-end-tracking proteins and the Ran GDP/GTP exchange factor. Distinct kinetochore components are used for kinetochore capture by microtubules and for ensuring subsequent sister kinetochore bi-orientation on the spindle. Kar3, a kinesin-14 family member, is one of the regulators that promote transport of captured kinetochores along microtubules. During such transport, kinetochores ensure that they do not slide off their associated microtubules by facilitating the conversion of microtubule dynamics from shrinkage to growth at the plus ends. This conversion is promoted by the transport of Stu2 from the captured kinetochores to the plus ends of microtubules.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
McIntosh, J. R., Grishchuk, E. L. & West, R. R. Chromosome-microtubule interactions during mitosis. Annu. Rev. Cell Dev. Biol. 18, 193–219 (2002)
Rieder, C. L. & Alexander, S. P. Kinetochores are transported poleward along a single astral microtubule during chromosome attachment to the spindle in newt lung cells. J. Cell Biol. 110, 81–95 (1990)
Hayden, J. H., Bowser, S. S. & Rieder, C. L. Kinetochores capture astral microtubules during chromosome attachment to the mitotic spindle: direct visualization in live newt lung cells. J. Cell Biol. 111, 1039–1045 (1990)
Merdes, A. & De Mey, J. The mechanism of kinetochore-spindle attachment and polewards movement analyzed in PtK2 cells at the prophase-prometaphase transition. Eur. J. Cell Biol. 53, 313–325 (1990)
Goshima, G. & Yanagida, M. Establishing biorientation occurs with precocious separation of the sister kinetochores, but not the arms, in the early spindle of budding yeast. Cell 100, 619–633 (2000)
He, X., Asthana, S. & Sorger, P. K. Transient sister chromatid separation and elastic deformation of chromosomes during mitosis in budding yeast. Cell 101, 763–775 (2000)
Tanaka, T., Fuchs, J., Loidl, J. & Nasmyth, K. Cohesin ensures bipolar attachment of microtubules to sister centromeres and resists their precocious separation. Nature Cell Biol. 2, 492–499 (2000)
Pearson, C. G., Maddox, P. S., Salmon, E. D. & Bloom, K. Budding yeast chromosome structure and dynamics during mitosis. J. Cell Biol. 152, 1255–1266 (2001)
Winey, M. & O'Toole, E. T. The spindle cycle in budding yeast. Nature Cell Biol. 3, E23–E27 (2001)
Tanaka, T. U. et al. Evidence that the Ipl1-Sli15 (Aurora kinase-INCENP) complex promotes chromosome bi-orientation by altering kinetochore-spindle pole connections. Cell 108, 317–329 (2002)
Tanaka, T. U. Chromosome bi-orientation on the mitotic spindle. Phil. Trans. R. Soc. Lond. B (in the press); doi:10.1098/rstb.2004.1612
Hill, A. & Bloom, K. Genetic manipulation of centromere function. Mol. Cell. Biol. 7, 2397–2405 (1987)
Michaelis, C., Ciosk, R. & Nasmyth, K. Cohesins: chromosomal proteins that prevent premature separation of sister chromatids. Cell 91, 35–45 (1997)
Nasmyth, K., Peters, J. M. & Uhlmann, F. Splitting the chromosome: cutting the ties that bind sister chromatids. Science 288, 1379–1385 (2000)
O'Toole, E. T., Winey, M. & McIntosh, J. R. High-voltage electron tomography of spindle pole bodies and early mitotic spindles in the yeast Saccharomyces cerevisiae. Mol. Biol. Cell 10, 2017–2031 (1999)
Gupta, M. L. Jr et al. β-Tubulin C354 mutations that severely decrease microtubule dynamics do not prevent nuclear migration in yeast. Mol. Biol. Cell 13, 2919–2932 (2002)
McAinsh, A. D., Tytell, J. D. & Sorger, P. K. Structure, function, and regulation of budding yeast kinetochores. Annu. Rev. Cell Dev. Biol. 19, 519–539 (2003)
Dewar, H., Tanaka, K., Nasmyth, K. & Tanaka, T. U. Tension between two kinetochores suffices for their bi-orientation on the mitotic spindle. Nature 428, 93–97 (2004)
Akhmanova, A. & Hoogenraad, C. C. Microtubule plus-end-tracking proteins: mechanisms and functions. Curr. Opin. Cell Biol. 17, 47–54 (2005)
Corbett, A. H. & Silver, P. A. Nucleocytoplasmic transport of macromolecules. Microbiol. Mol. Biol. Rev. 61, 193–211 (1997)
Hetzer, M., Gruss, O. J. & Mattaj, I. W. The Ran GTPase as a marker of chromosome position in spindle formation and nuclear envelope assembly. Nature Cell Biol. 4, E177–E184 (2002)
Quimby, B. B. & Dasso, M. The small GTPase Ran: interpreting the signs. Curr. Opin. Cell Biol. 15, 338–344 (2003)
Wilde, A. et al. Ran stimulates spindle assembly by altering microtubule dynamics and the balance of motor activities. Nature Cell Biol. 3, 221–227 (2001)
Carazo-Salas, R. E., Gruss, O. J., Mattaj, I. W. & Karsenti, E. Ran-GTP coordinates regulation of microtubule nucleation and dynamics during mitotic-spindle assembly. Nature Cell Biol. 3, 228–234 (2001)
Biggins, S. et al. The conserved protein kinase Ipl1 regulates microtubule binding to kinetochores in budding yeast. Genes Dev. 13, 532–544 (1999)
Cheeseman, I. M., Enquist-Newman, M., Muller-Reichert, T., Drubin, D. G. & Barnes, G. Mitotic spindle integrity and kinetochore function linked by the Duo1p/Dam1p complex. J. Cell Biol. 152, 197–212 (2001)
He, X., Rines, D. R., Espelin, C. W. & Sorger, P. K. Molecular analysis of kinetochore-microtubule attachment in budding yeast. Cell 106, 195–206 (2001)
Janke, C., Ortiz, J., Tanaka, T. U., Lechner, J. & Schiebel, E. Four new subunits of the Dam1–Duo1 complex reveal novel functions in sister kinetochore biorientation. EMBO J. 21, 181–193 (2002)
Li, Y. et al. The mitotic spindle is required for loading of the DASH complex onto the kinetochore. Genes Dev. 16, 183–197 (2002)
Maddox, P. S., Bloom, K. S. & Salmon, E. D. The polarity and dynamics of microtubule assembly in the budding yeast Saccharomyces cerevisiae. Nature Cell Biol. 2, 36–41 (2000)
Tirnauer, J. S., O'Toole, E., Berrueta, L., Bierer, B. E. & Pellman, D. Yeast Bim1p promotes the G1-specific dynamics of microtubules. J. Cell Biol. 145, 993–1007 (1999)
van Breugel, M., Drechsel, D. & Hyman, A. Stu2p, the budding yeast member of the conserved Dis1/XMAP215 family of microtubule-associated proteins is a plus end-binding microtubule destabilizer. J. Cell Biol. 161, 359–369 (2003)
Lin, H. et al. Polyploids require Bik1 for kinetochore–microtubule attachment. J. Cell Biol. 155, 1173–1184 (2001)
Carvalho, P., Gupta, M. L. Jr, Hoyt, M. A. & Pellman, D. Cell cycle control of kinesin-mediated transport of Bik1 (CLIP-170) regulates microtubule stability and dynein activation. Dev. Cell 6, 815–829 (2004)
Hildebrandt, E. R. & Hoyt, M. A. Mitotic motors in Saccharomyces cerevisiae. Biochim. Biophys. Acta 1496, 99–116 (2000)
Saunders, W. S., Koshland, D., Eshel, D., Gibbons, I. R. & Hoyt, M. A. Saccharomyces cerevisiae kinesin- and dynein-related proteins required for anaphase chromosome segregation. J. Cell Biol. 128, 617–624 (1995)
Meluh, P. B. & Rose, M. D. KAR3, a kinesin-related gene required for yeast nuclear fusion. Cell 60, 1029–1041 (1990)
Maddox, P. S., Stemple, J. K., Satterwhite, L., Salmon, E. D. & Bloom, K. The minus end-directed motor Kar3 is required for coupling dynamic microtubule plus ends to the cortical shmoo tip in budding yeast. Curr. Biol. 13, 1423–1428 (2003)
Muller-Reichert, T. et al. Analysis of the distribution of the kinetochore protein Ndc10p in Saccharomyces cerevisiae using 3-D modeling of mitotic spindles. Chromosoma 111, 417–428 (2003)
Adams, I. R. & Kilmartin, J. V. Spindle pole body duplication: a model for centrosome duplication? Trends Cell Biol. 10, 329–335 (2000)
Cheeseman, I. M. et al. Phospho-regulation of kinetochore-microtubule attachments by the Aurora kinase Ipl1p. Cell 111, 163–172 (2002)
Westermann, S. et al. Architecture of the budding yeast kinetochore reveals a conserved molecular core. J. Cell Biol. 163, 215–222 (2003)
Nekrasov, V. S., Smith, M. A., Peak-Chew, S. & Kilmartin, J. V. Interactions between centromere complexes in Saccharomyces cerevisiae. Mol. Biol. Cell 14, 4931–4946 (2003)
De Wulf, P., McAinsh, A. D. & Sorger, P. K. Hierarchical assembly of the budding yeast kinetochore from multiple subcomplexes. Genes Dev. 17, 2902–2921 (2003)
Miranda, J. L., De Wulf, P., Sorger, P. & Harrison, S. C. The yeast DASH complex forms closed rings on microtubules. Nature Struct. Mol. Biol. 12, 138–143 (2005)
Westermann, S. et al. Formation of a dynamic kinetochore-microtubule interface through assembly of the Dam1 ring complex. Mol. Cell 17, 277–290 (2005)
Pickett-Heaps, J. D. Cell division in diatoms. Int. Rev. Cytol. 128, 63–107 (1991)
Carazo-Salas, R. E. & Karsenti, E. Long-range communication between chromatin and microtubules in Xenopus egg extracts. Curr. Biol. 13, 1728–1733 (2003)
Alexander, S. P. & Rieder, C. L. Chromosome motion during attachment to the vertebrate spindle: initial saltatory-like behavior of chromosomes and quantitative analysis of force production by nascent kinetochore fibers. J. Cell Biol. 113, 805–815 (1991)
King, J. M., Hays, T. S. & Nicklas, R. B. Dynein is a transient kinetochore component whose binding is regulated by microtubule attachment, not tension. J. Cell Biol. 151, 739–748 (2000)
Acknowledgements
We thank J. R. Swedlow, M. J. R. Stark, A. Gartner, M. A. Hoyt, A. Desai, P. R. Clarke, P. D. Andrews and members of the Tanaka laboratory for discussions and for reading the manuscript; T. Hyman, K. Nasmyth, I. W. Mattaj, E. Karsenti, F. Uhlmann and J. Ellenberg for discussions; J.-F. Maure, N. Rachidi and M. J. R. Stark for sharing their unpublished data; Y. Kitamura, S. Swift, M. Romao and G. Keir for technical help; F. Wheatley and the media kitchen for media preparation; M. A. Hoyt, D. Pellman, R. Ciosk, F. Uhlmann, K. Nasmyth, T. C. Huffaker, J. V. Kilmartin, E. Schiebel, S. Biggins, C. S. M. Chan, I. M. Cheeseman, G. Barnes, R. Tsien, S. J. Elledge, J. Lechner, A. H. Corbett, P. A. Silver, P. K. Sorger, X. He, A. F. Straight, M. D. Rose, V. Doye, F. Severin, I. Ouspenski, K. Bloom, T. Nishimoto, J. E. Haber, T. N. Davis, EUROSCARF and the Yeast Resource Center for reagents. This work was supported by The Wellcome Trust, Cancer Research UK and the EMBO Young Investigator Program.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Supplementary Notes
This file contains Supplementary Notes and Supplementary Figures S1-S13. (PDF 462 kb)
Supplementary Video Legends
Legends to accompany the below Supplementary Videos. (RTF 4 kb)
Supplementary Video S1
Video of the cell shown in Fig 1b. Speed in the video is 100 times faster than the actual motion. (MOV 6892 kb)
Supplementary Video S2
Video of the cell shown in Fig 1c. Speed in the video is 100 times faster than the actual motion. (MOV 6034 kb)
Supplementary Video S3
Video of the cell shown in Supplementary Fig S2. Speed in the video is 100 times faster than the actual motion. (MOV 3798 kb)
Supplementary Video S4
Video of the cell shown in Supplementary Fig S4a. Speed in the video is 100 times faster than the actual motion. (MOV 1839 kb)
Supplementary Video S5
Video of the cell shown in Fig 3a, top. Speed in the video is 100 times faster than the actual motion. (MOV 2663 kb)
Supplementary Video S6
Video of the cell shown in Fig 3a, bottom. Speed in the video is 100 times faster than the actual motion. (MOV 5923 kb)
Supplementary Video S7
Video of the cells shown in Fig 4a. Speed in the video is 50 times faster than the actual motion. (MOV 5193 kb)
Supplementary Video S8
Video of a wild type control for Supplementary Video S9 and S10. This cell is not shown in any panels of figures. KAR3+ cells (T3531) were treated as in Fig 5. GFP and YFP signals were collected together as in Supplementary Video S9 and S10). Speed in the video is 300 times faster than the actual motion. (MOV 2532 kb)
Supplementary Video S9
Video of the kar3-1 cell shown in Fig 5a. Speed in the video is 300 times faster than the actual motion. (MOV 2601 kb)
Supplementary Video S10
Video of the KAR3-overexpressed cell shown in Fig 5a. Speed in the video is 300 times faster than the actual motion. (MOV 2460 kb)
Supplementary Video S11
Video of the cell shown in Supplementary Fig S12. Speed in the video is 150 times faster than the actual motion. (MOV 1603 kb)
Video S12
Video of the cell shown in Fig 6a. Speed in the video is 50 times faster than the actual motion. (MOV 4278 kb)
Rights and permissions
About this article
Cite this article
Tanaka, K., Mukae, N., Dewar, H. et al. Molecular mechanisms of kinetochore capture by spindle microtubules. Nature 434, 987–994 (2005). https://doi.org/10.1038/nature03483
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature03483
This article is cited by
-
Defining a core configuration for human centromeres during mitosis
Nature Communications (2023)
-
The dark side of centromeres: types, causes and consequences of structural abnormalities implicating centromeric DNA
Nature Communications (2018)
-
Unattached kinetochores drive their own capturing by sequestering a CLASP
Nature Communications (2018)
-
Mitotic spindle assembly in animal cells: a fine balancing act
Nature Reviews Molecular Cell Biology (2017)
-
Aurora-B kinase pathway controls the lateral to end-on conversion of kinetochore-microtubule attachments in human cells
Nature Communications (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.