Abstract
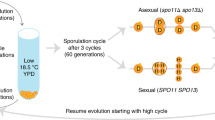
Why sex evolved and persists is a problem for evolutionary biology, because sex disrupts favourable gene combinations and requires an expenditure of time and energy1. Further, in organisms with unequal-sized gametes, the female transmits her genes at only half the rate of an asexual equivalent (the twofold cost of sex)2. Many modern theories that provide an explanation for the advantage of sex incorporate an idea originally proposed by Weismann more than 100 years ago: sex allows natural selection to proceed more effectively because it increases genetic variation3,4,5. Here we test this hypothesis, which still lacks robust empirical support, with the use of experiments on yeast populations. Capitalizing on recent advances in the molecular biology of recombination in yeast, we produced by genetic manipulation strains that differed only in their capacity for sexual reproduction. We show that, as predicted by the theory, sex increases the rate of adaptation to a new harsh environment but has no measurable effect on fitness in a new benign environment where there is little selection.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Bell, G. The Masterpiece of Nature (Univ. California Press, Berkeley, 1982)
Michod, R. E. & Levin, B. R. The Evolution of Sex: An Examination of Current Ideas (Sinauer, Sunderland, Massachusetts, 1988)
Weismann, A. The Evolution Theory (Edward Arnold, London, 1904)
Barton, N. H. & Charlesworth, B. Why sex and recombination? Science 281, 1986–1990 (1998)
Burt, A. Sex, recombination, and the efficacy of selection—was Weismann right? Evolution 54, 337–351 (2000)
Bruggeman, J., Debets, A. J. M., Wijngaarden, P. J., deVisser, J. A. G. M. & Hoekstra, R. F. Sex slows down the accumulation of deleterious mutations in the homothallic fungus Aspergillus nidulans . Genetics 164, 479–485 (2003)
Otto, S. P. & Lenormand, T. Resolving the paradox of sex and recombination. Nature Rev. Genet. 3, 252–261 (2002)
Birdsell, J. & Wills, C. Significant competitive advantage conferred by meiosis and syngamy in the yeast Saccharomyces cerevisiae . Proc. Natl Acad. Sci. USA 93, 908–912 (1996)
Greig, D., Borts, R. H. & Louis, E. J. The effect of sex on adaptation to high temperature in heterozygous and homozygous yeast. Proc. R. Soc. Lond. B 265, 1017–1023 (1998)
Colegrave, N. Sex releases the speed limit on evolution. Nature 420, 664–666 (2002)
Kaltz, O. & Bell, G. The ecology and genetics of fitness in Chlamydomonas. XII. Repeated sexual episodes increase the rates of adaptation to novel environments. Evolution 56, 1743–1753 (2002)
Rice, W. R. Experimental effects of the adaptive significance of sexual recombination. Nature Rev. Genet. 3, 241–251 (2002)
Rice, W. R. & Chippindale, A. K. Sexual recombination and the power of natural selection. Science 294, 555–559 (2001)
Goho, S. & Bell, G. Mild environmental stress elicits mutations affecting fitness in Chlamydomonas . Proc. R. Soc. Lond. B 267, 123–129 (2000)
Marini, A., Matmati, N. & Morpurgo, G. Starvation in yeast increases non-adaptive mutation. Curr. Genet. 35, 77–81 (1999)
Berry, D. R. The Biology of Yeasts (Edward Arnold, London, 1982)
Grewal, N. S. & Miller, J. J. Formation of asci with two diploid spores by diploid cells of Saccharomyces . Can. J. Microbiol. 18, 1897–1905 (1972)
Klapholz, S., Waddell, C. S. & Esposito, R. E. The role of the SPO11 gene in meiotic recombination in yeast. Genetics 110, 187–216 (1985)
Shonn, M. A., McCarroll, R. & Murray, A. W. Spo13 protects meiotic cohesin at centromeres in meiosis I. Genes Dev. 16, 1659–1671 (2002)
Wang, H., Frackman, S., Kowalisyn, J., Esposito, R. E. & Elder, R. Developmental regulation of SPO13, a gene required for separation of homologous chromosomes at meiosis I. Mol. Cell. Biol. 7, 1425–1435 (1987)
Steele, D. F., Morris, M. E. & Jinks-Robertson, S. Allelic and ectopic interactions in recombination-defective yeast strains. Genetics 127, 53–60 (1991)
Wach, A. et al. PCR-based gene targeting in S. cerevisiae . Methods Microbiol. 26, 67–81 (1998)
Baganz, F., Hayes, A., Marren, D., Gardner, D. C. J. & Oliver, S. G. Suitability of replacement markers for functional analysis studies in Saccharomyces cerevisiae . Yeast 13, 1536–1573 (1997)
Burke, D., Dawson, D. & Stearns, T. Methods in Yeast Genetics. A Cold Spring Harbor Laboratory Course Manual (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, 2000)
Pinheiro, J. C. & Bates, D. M. Mixed-effects Models in S and S-PLUS (Springer, New York, 2000)
Zeyl, C. & DeVisser, J. A. G. M. Estimates of the rate and distribution of fitness effects of spontaneous mutation in Saccharomyces cerevisiae . Genetics 157, 53–61 (2001)
Burt, A. The evolution of fitness. Evolution 49, 1–8 (1995)
Muller, H. J. Some genetic aspects of sex. Am. Nat. 66, 118–138 (1932)
Barton, N. A general model for the evolution of recombination. Genet. Res. 65, 123–144 (1995)
Acknowledgements
We thank M. Rees for comments on statistical analyses. This work was supported by the UK Natural Environment Research Council (NERC).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Supplementary Methods
This file contains more detailed descriptions of the methods used in this study. (DOC 37 kb)
Rights and permissions
About this article
Cite this article
Goddard, M., Godfray, H. & Burt, A. Sex increases the efficacy of natural selection in experimental yeast populations. Nature 434, 636–640 (2005). https://doi.org/10.1038/nature03405
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature03405
This article is cited by
-
Sexual recombination and temporal gene flow maintain host resistance and genetic diversity
Evolutionary Ecology (2023)
-
The transcriptomic signature of obligate parthenogenesis
Heredity (2022)
-
Sexual morph specialisation in a trioecious nematode balances opposing selective forces
Scientific Reports (2022)
-
Understanding the drivers of dispersal evolution in range expansions and their ecological consequences
Evolutionary Ecology (2022)
-
Genetic diversity of rhizobia isolated from nodules of Trigonella foenum-graecum L. (fenugreek) cultivated in Northwestern Morocco
Archives of Microbiology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.