Abstract
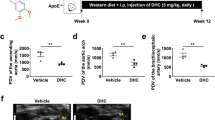
Atherosclerosis is a chronic inflammatory disease, and is the primary cause of heart disease and stroke in Western countries1. Derivatives of cannabinoids such as delta-9-tetrahydrocannabinol (THC) modulate immune functions2 and therefore have potential for the treatment of inflammatory diseases. We investigated the effects of THC in a murine model of established atherosclerosis. Oral administration of THC (1 mg kg-1 per day) resulted in significant inhibition of disease progression. This effective dose is lower than the dose usually associated with psychotropic effects of THC. Furthermore, we detected the CB2 receptor (the main cannabinoid receptor expressed on immune cells2,3) in both human and mouse atherosclerotic plaques. Lymphoid cells isolated from THC-treated mice showed diminished proliferation capacity and decreased interferon-γ secretion. Macrophage chemotaxis, which is a crucial step for the development of atherosclerosis1, was also inhibited in vitro by THC. All these effects were completely blocked by a specific CB2 receptor antagonist4. Our data demonstrate that oral treatment with a low dose of THC inhibits atherosclerosis progression in the apolipoprotein E knockout mouse model, through pleiotropic immunomodulatory effects on lymphoid and myeloid cells. Thus, THC or cannabinoids with activity at the CB2 receptor may be valuable targets for treating atherosclerosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Libby, P. Inflammation in atherosclerosis. Nature 420, 868–874 (2002)
Klein, T. W. et al. The cannabinoid system and immune modulation. J. Leukoc. Biol. 74, 486–496 (2003)
Buckley, N. E. et al. Immunomodulation by cannabinoids is absent in mice deficient for the cannabinoid CB(2) receptor. Eur. J. Pharmacol. 396, 141–149 (2000)
Rinaldi-Carmona, M. et al. SR144528, the first potent and selective antagonist of the CB2 cannabinoid receptor. J. Pharmacol. Exp. Ther. 284, 644–650 (1998)
Libby, P., Ridker, P. M. & Maseri, A. Inflammation and atherosclerosis. Circulation 105, 1135–1143 (2002)
Mach, F. Statins as immunomodulatory agents. Circulation 109 (suppl.), II15–II17 (2004)
Srivastava, M. D., Srivastava, B. I. & Brouhard, B. Δ9 tetrahydrocannabinol and cannabidiol alter cytokine production by human immune cells. Immunopharmacology 40, 179–185 (1998)
Zhu, L. X. et al. Δ-9-tetrahydrocannabinol inhibits antitumor immunity by a CB2 receptor-mediated, cytokine-dependent pathway. J. Immunol. 165, 373–380 (2000)
Yuan, M. et al. Δ9-Tetrahydrocannabinol regulates Th1/Th2 cytokine balance in activated human T cells. J. Neuroimmunol. 133, 124–131 (2002)
Lyman, W. D., Sonett, J. R., Brosnan, C. F., Elkin, R. & Bornstein, M. B. Δ9-Tetrahydrocannabinol: a novel treatment for experimental autoimmune encephalomyelitis. J. Neuroimmunol. 23, 73–81 (1989)
Malfait, A. M. et al. The nonpsychoactive cannabis constituent cannabidiol is an oral anti-arthritic therapeutic in murine collagen-induced arthritis. Proc. Natl Acad. Sci. USA 97, 9561–9566 (2000)
Sulcova, E., Mechoulam, R. & Fride, E. Biphasic effects of anandamide. Pharmacol. Biochem. Behav. 59, 347–352 (1998)
Brenneisen, R., Egli, A., Elsohly, M. A., Henn, V. & Spiess, Y. The effect of orally and rectally administered Δ9-tetrahydrocannabinol on spasticity: a pilot study with 2 patients. Int. J. Clin. Pharmacol. Ther. 34, 446–452 (1996)
Chesher, G. B., Bird, K. D., Jackson, D. M., Perrignon, A. & Starmer, G. A. The effects of orally administered Δ9-tetrahydrocannabinol in man on mood and performance measures: a dose-response study. Pharmacol. Biochem. Behav. 35, 861–864 (1990)
Lichtman, A. H., Poklis, J. L., Poklis, A., Wilson, D. M. & Martin, B. R. The pharmacological activity of inhalation exposure to marijuana smoke in mice. Drug Alcohol Depend. 63, 107–116 (2001)
Varvel, S. A., Hamm, R. J., Martin, B. R. & Lichtman, A. H. Differential effects of Δ9-THC on spatial reference and working memory in mice. Psychopharmacology (Berl.) 157, 142–150 (2001)
Nakashima, Y., Plump, A. S., Raines, E. W., Breslow, J. L. & Ross, R. ApoE-deficient mice develop lesions of all phases of atherosclerosis throughout the arterial tree. Arterioscler. Thromb. 14, 133–140 (1994)
Reddick, R. L., Zhang, S. H. & Maeda, N. Atherosclerosis in mice lacking apo E. Evaluation of lesional development and progression. Arterioscler. Thromb. 14, 141–147 (1994)
Tangirala, R. K., Rubin, E. M. & Palinski, W. Quantitation of atherosclerosis in murine models: correlation between lesions in the aortic origin and in the entire aorta, and differences in the extent of lesions between sexes in LDL receptor-deficient and apolipoprotein E-deficient mice. J. Lipid Res. 36, 2320–2328 (1995)
Johnson, Z. et al. Interference with heparin binding and oligomerization creates a novel anti-inflammatory strategy targeting the chemokine system. J. Immunol. 173, 5776–5785 (2004)
Daugherty, A. & Rateri, D. L. T lymphocytes in atherosclerosis: the yin-yang of Th1 and Th2 influence on lesion formation. Circ. Res. 90, 1039–1040 (2002)
Moeller, F. & Nielsen, L. B. Aortic recruitment of blood lymphocytes is most pronounced in early stages of lesion formation in apolipoprotein-E-deficient mice. Atherosclerosis 168, 49–56 (2003)
Song, L., Leung, C. & Schindler, C. Lymphocytes are important in early atherosclerosis. J. Clin. Invest. 108, 251–259 (2001)
Benagiano, M. et al. T helper type 1 lymphocytes drive inflammation in human atherosclerotic lesions. Proc. Natl Acad. Sci. USA 100, 6658–6663 (2003)
Laurat, E. et al. In vivo downregulation of T helper cell 1 immune responses reduces atherogenesis in apolipoprotein E-knockout mice. Circulation 104, 197–202 (2001)
Mulhaupt, F. et al. Statins (HMG-CoA reductase inhibitors) reduce CD40 expression in human vascular cells. Cardiovasc. Res. 59, 755–766 (2003)
Kwak, B. R. et al. Reduced connexin43 expression inhibits atherosclerotic lesion formation in low-density lipoprotein receptor-deficient mice. Circulation 107, 1033–1039 (2003)
Giroud, C. et al. Δ9-THC, 11-OH-Δ9-THC and Δ9-THCCOOH plasma or serum to whole blood concentrations distribution ratios in blood samples taken from living and dead people. Forensic Sci. Int. 123, 159–164 (2001)
Hickey, M. J., Bullard, D. C., Issekutz, A. & James, W. G. Leukocyte-endothelial cell interactions are enhanced in dermal postcapillary venules of MRL/fas(lpr) (lupus-prone) mice: roles of P- and E-selectin. J. Immunol. 168, 4728–4736 (2002)
Veillard, N. R., Steffens, S., Burger, F., Pelli, G. & Mach, F. Differential expression patterns of proinflammatory and antiinflammatory mediators during atherogenesis in mice. Arterioscler. Thromb. Vasc. Biol. 24, 2339–2344 (2004)
Acknowledgements
This work was supported by grants from the Swiss National Science Foundation to F.M. and J.-L.F., from the Ernst and Lucie Schmidheiny Foundation (Geneva) to F.M., and from the Foundation for Medical Research (France) to C.A. The authors of this manuscript belong to the European Vascular Genomics Network (http://www.evgn.org), a Network of Excellence supported by the European Community's sixth Framework Programme for Research, Priority 1. We would like to thank M. Kosco-Vilbois for helpful discussion in preparing the manuscript, and M.-L. Bochaton-Piallat (Pathology Department) for technical advice.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Rights and permissions
About this article
Cite this article
Steffens, S., Veillard, N., Arnaud, C. et al. Low dose oral cannabinoid therapy reduces progression of atherosclerosis in mice. Nature 434, 782–786 (2005). https://doi.org/10.1038/nature03389
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature03389
This article is cited by
-
Aquaporin 3 promotes human extravillous trophoblast migration and invasion
Reproductive Biology and Endocrinology (2021)
-
Cannabis and Athletic Performance
Sports Medicine (2021)
-
Cannabis sativa extracts protect LDL from Cu2+-mediated oxidation
Journal of Cannabis Research (2020)
-
A high-quality reference genome of wild Cannabis sativa
Horticulture Research (2020)
-
The endocannabinoid anandamide has an anti-inflammatory effect on CCL2 expression in vascular smooth muscle cells
Basic Research in Cardiology (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.