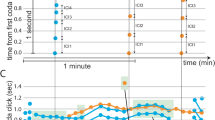
Abstract
Conventional models1,2,3,4 explaining extreme sexual ornaments propose that these reflect male genetic quality2,3,4 or are arbitrary results of genetic linkage between female preference and the ornament1. The chase-away model5 emphasizes sexual conflict: male signals attract females because they exploit receiver biases6,7,8,9. As males gain control of mating decisions, females may experience fitness costs through suboptimal mating rates or post-copulatory exploitation. Elaboration of male signals is expected if females increase their response threshold to resist such exploitation. If ornaments target otherwise adaptive biases such as feeding responses8,9,10, selection on females might eventually separate sexual and non-sexual responses to the signal. Here we show that the terminal yellow band (TYB) of several Goodeinae species evokes both feeding and sexual responses; sexual responsiveness phylogenetically pre-dates the expression of the TYB in males and is comparable across taxa, yet feeding responsiveness decreases in species with more elaborated TYBs. Displaying a TYB is costly, and thus provides an example where a trait arose as a sensory trap but has evolved into an honest signal.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Fisher, R. A. The Genetical Theory of Natural Selection 146–156 (Dover Publications, New York, 1958)
Andersson, M. Evolution of condition-dependent sex ornaments and mating preferences: sexual selection based on viability differences. Evolution 40, 804–816 (1986)
Hamilton, W. D. & Zuk, M. Heritable true fitness and bright birds: a role for parasites? Science 218, 384–387 (1982)
Milinski, M. & Bakker, T. C. M. Female sticklebacks use male coloration in mate choice and hence avoid parasitized males. Nature 344, 330–333 (1990)
Holland, B. & Rice, W. R. Chase-away sexual selection: antagonistic seduction versus resistance. Evolution 52, 1–7 (1998)
Ryan, M. J. Sexual selection, sensory systems and sensory exploitation. Oxf. Surv. Evol. Biol. 7, 157–195 (1990)
Ryan, M. J. & Keddy-Hector, A. Directional patterns of female mate choice and the role of sensory biases. Am. Nat. 139, S4–S35 (1992)
Dawkins, M. S. & Guilford, T. Sensory bias and the adaptiveness of female choice. Am. Nat. 148, 937–942 (1996)
Endler, J. A. & Basolo, A. L. Sensory ecology, receiver biases and sexual selection. Trends Ecol. Evol. 13, 415–420 (1998)
Rodd, F. H., Hughes, K. A., Grether, G. F. & Baril, C. T. A possible non-sexual origin of mate preference: are male guppies mimicking fruit? Proc. R. Soc. Lond. B 269, 475–481 (2002)
Kokko, H., Brooks, R., McNamara, J. M. & Houston, A. I. The sexual selection continuum. Proc. R. Soc. Lond. B 269, 1331–1340 (2002)
Kokko, H., Brooks, R., Jennions, M. D. & Morley, J. The evolution of mate choice and mating biases. Proc. R. Soc. Lond. B 270, 653–664 (2003)
Cordero, C. & Eberhard, W. G. Female choice of sexually antagonistic male adaptations: a critical review of some current research. J. Evol. Biol. 16, 1–6 (2003)
Webb, S. A. et al. Molecular phylogeny of the live-bearing Goodeidae (Cyprinodontiformes). Mol. Phylogenet. Evol. 30, 527–544 (2004)
LeBas, N., Hockham, L. R. & Ritchie, M. G. Nonlinear and correlational sexual selection on ‘honest’ female ornamentation. Proc. R. Soc. Lond. B 270, 2159–2165 (2003)
Kodric-Brown, A. Dietary carotenoids and male mating success in the guppy: an environmental component to female choice. Behav. Ecol. Sociobiol. 25, 393–401 (1989)
Olson, V. A. & Owens, I. P. F. Costly sexual signals: are carotenoids rare, risky or required? Trends Ecol. Evol. 13, 510–514 (1998)
Partridge, L. & Parker, G. A. in Evolution of Biological Diversity (eds Magurran, A. E. & May, R. M.) 130–159 (Oxford Univ. Press, Oxford, 1999)
Basolo, A. L. Congruence between the sexes in preexisting receiver responses. Behav. Ecol. 13(6), 832–837 (2002)
Jayasooriya, A. P., Weisinger, R. S., Weisinger, H. S., Mathai, M. L. & Sinclair, A. J. Attraction to orange: sexiness, not gluttony. Science 296, 847–848 (2002)
Grether, G. F., Hughes, K. A. & Rodd, F. H. Response to Jayasooriya et al. Science 296, 847–848 (2002)
Burton, B. G. & Laughlin, S. B. Neural images of pursuit targets in the photoreceptor arrays of male and female houseflies Musca domestica . J. Exp. Biol. 206, 3963–3977 (2003)
Córdoba-Aguilar, A. Sensory trap as the mechanism of sexual selection in a damselfly genitalic trait (insecta: calopterygidae). Am. Nat. 160, 594–601 (2002)
Holland, B. & Rice, W. R. Experimental removal of sexual selection reverses intersexual antagonistic coevolution and removes a reproductive load. Proc. Natl Acad. Sci. USA 96, 5083–5088 (1999)
Ryan, M. J. & Wagner, E. Asymmetries in mating preferences between species: female swordtails prefer heterospecific males. Science 236, 595–597 (1987)
Basolo, A. L. Evolutionary change in a receiver bias: a comparison of female preference functions. Proc. R. Soc. Lond. B 265, 2223–2228 (1998)
Acknowledgements
We Thank the Lunáticos behaviour discussion group for constructive discussion throughout, and M. Ritchie, J. Graves and H. Kokko for useful comments on previous versions of the manuscript. A. Gaona and M. Molina helped with some of the behavioural records, and E. Ávila Luna provided logistical aid with fish husbandry and data collection. This project was partly supported with a grant from CONACyT to C.M.G. and is based on E.R.'s BSc thesis (Biology), which was supervised by C.M.G.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Supplementary Figure S1
The left column shows the spectral reflectance of the sub-terminal tail band of males and females of each species. In the central column are the spectra of the terminal band and the last column shows the difference between the spectra in the first two columns. (PDF 241 kb)
Supplementary Figure S2
This file is an expanded version of Figure 2 from the main document. This includes one variable (frequency of visits to males with contrasting TYB conspicuousness) not included in Figure 2. (PDF 14 kb)
Supplementary Figure S3
This file is an expanded version of Figure 3 from the main document. This includes one variable (frequency of visits to tails with [ ] or without [ ] a TYB) that is not included in Figure 3. (PDF 52 kb)
Supplementary Figure S4
This shows the regression of rate of mass change on time in the experiment. This demonstrates that regenerating lost tail tissue is costly. The statistics of the regression analysis and its significance are provided in the main text. (PDF 14 kb)
Supplementary Figure S5
Supplementary Figure S5 shows four panels from a video sequence of male C. audax biting the tail of a fish (X. variata) showing a TYB while ignoring a tail lacking a TYB. Individual X. variata are hold in cages as described in Methods. (PDF 27 kb)
Supplementary Figure legends
Legends to accompany the above Supplementary Figures. (DOC 23 kb)
Supplementary Tables S1 and S2
This file contains Supplementary Table S1, which presents the ANOVA tables of all the tests (18) conducted to assess female sexual responsiveness for males with contrasting TYB. Supplementary Table S2 presents a full account of the results and statistics of the exposure of X. variata to elaborated TYB from heterospecific fish to test for feeding responses. The results are mentioned in the main text but expanded on here. (DOC 224 kb)
Rights and permissions
About this article
Cite this article
Garcia, C., Ramirez, E. Evidence that sensory traps can evolve into honest signals. Nature 434, 501–505 (2005). https://doi.org/10.1038/nature03363
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature03363
This article is cited by
-
Leg coloration is associated with habitat type and social environment in swallows and martins
Journal of Ornithology (2023)
-
Star finches Neochmia ruficauda have a visual preference for white dot patterns: a possible case of trypophilia
Animal Cognition (2022)
-
Ineffective nuptial gifts suggest female emancipation from sensory exploitation
Behavioral Ecology and Sociobiology (2021)
-
Spider lures exploit insect preferences for floral colour and symmetry
Evolutionary Ecology (2020)
-
Mimicry, Camouflage and Perceptual Exploitation: the Evolution of Deception in Nature
Biosemiotics (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.