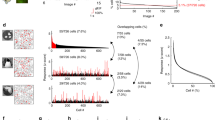
Abstract
Neurons in the cerebral cortex are organized into anatomical columns, with ensembles of cells arranged from the surface to the white matter. Within a column, neurons often share functional properties, such as selectivity for stimulus orientation; columns with distinct properties, such as different preferred orientations, tile the cortical surface in orderly patterns. This functional architecture was discovered with the relatively sparse sampling of microelectrode recordings. Optical imaging of membrane voltage or metabolic activity elucidated the overall geometry of functional maps, but is averaged over many cells (resolution >100 µm). Consequently, the purity of functional domains and the precision of the borders between them could not be resolved. Here, we labelled thousands of neurons of the visual cortex with a calcium-sensitive indicator in vivo. We then imaged the activity of neuronal populations at single-cell resolution with two-photon microscopy up to a depth of 400 µm. In rat primary visual cortex, neurons had robust orientation selectivity but there was no discernible local structure; neighbouring neurons often responded to different orientations. In area 18 of cat visual cortex, functional maps were organized at a fine scale. Neurons with opposite preferences for stimulus direction were segregated with extraordinary spatial precision in three dimensions, with columnar borders one to two cells wide. These results indicate that cortical maps can be built with single-cell precision.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Mountcastle, V. B. Modality and topographic properties of single neurons of cat's somatic sensory cortex. J. Neurophysiol. 20, 408–434 (1957)
Hubel, D. H. & Wiesel, T. N. Receptive fields, binocular interaction and functional architecture in the cat's visual cortex. J. Physiol. (Lond.) 160, 106–154 (1962)
Grinvald, A., Anglister, L., Freeman, J. A., Hildesheim, R. & Manker, A. Real-time optical imaging of naturally evoked electrical activity in intact frog brain. Nature 308, 848–850 (1984)
Blasdel, G. G. & Salama, G. Voltage-sensitive dyes reveal a modular organization in monkey striate cortex. Nature 321, 579–585 (1986)
Grinvald, A., Lieke, E., Frostig, R. D., Gilbert, C. D. & Wiesel, T. N. Functional architecture of cortex revealed by optical imaging of intrinsic signals. Nature 324, 361–364 (1986)
Bonhoeffer, T. & Grinvald, A. Iso-orientation domains in cat visual cortex are arranged in pinwheel-like patterns. Nature 353, 429–431 (1991)
Shmuel, A. & Grinvald, A. Functional organization for direction of motion and its relationship to orientation maps in cat area 18. J. Neurosci. 16, 6945–6964 (1996)
Weliky, M., Bosking, W. H. & Fitzpatrick, D. A systematic map of direction preference in primary visual cortex. Nature 379, 725–728 (1996)
Maldonado, P. E., Godecke, I., Gray, C. M. & Bonhoeffer, T. Orientation selectivity in pinwheel centers in cat striate cortex. Science 276, 1551–1555 (1997)
Parnavelas, J. G., Burne, R. A. & Lin, C. S. Receptive field properties of neurons in the visual cortex of the rat. Neurosci. Lett. 27, 291–296 (1981)
Girman, S. V., Sauve, Y. & Lund, R. D. Receptive field properties of single neurons in rat primary visual cortex. J. Neurophysiol. 82, 301–311 (1999)
Tsien, R. Y. Fluorescence measurement and photochemical manipulation of cytosolic free calcium. Trends Neurosci. 11, 419–424 (1988)
Denk, W., Strickler, J. H. & Webb, W. W. Two-photon laser scanning fluorescence microscopy. Science 248, 73–76 (1990)
Svoboda, K., Denk, W., Kleinfeld, D. & Tank, D. W. In vivo dendritic calcium dynamics in neocortical pyramidal neurons. Nature 385, 161–165 (1997)
Waters, J., Larkum, M., Sakmann, B. & Helmchen, F. Supralinear Ca2+ influx into dendritic tufts of layer 2/3 neocortical pyramidal neurons in vitro and in vivo . J. Neurosci. 23, 8558–8567 (2003)
Yuste, R. & Katz, L. C. Control of postsynaptic Ca2+ influx in developing neocortex by excitatory and inhibitory neurotransmitters. Neuron 6, 333–344 (1991)
Yuste, R., Peinado, A. & Katz, L. C. Neuronal domains in developing neocortex. Science 257, 665–669 (1992)
Stosiek, C., Garaschuk, O., Holthoff, K. & Konnerth, A. In vivo two-photon calcium imaging of neuronal networks. Proc. Natl Acad. Sci. USA 100, 7319–7324 (2003)
Wiesenfeld, Z. & Kornel, E. E. Receptive fields of single cells in the visual cortex of the hooded rat. Brain Res. 94, 401–412 (1975)
Mao, B. Q., Hamzei-Sichani, F., Aronov, D., Froemke, R. C. & Yuste, R. Dynamics of spontaneous activity in neocortical slices. Neuron 32, 883–898 (2001)
Ts'o, D. Y., Frostig, R. D., Lieke, E. E. & Grinvald, A. Functional organization of primate visual cortex revealed by high resolution optical imaging. Science 249, 417–420 (1990)
Payne, B. R., Berman, N. & Murphy, E. H. Organization of direction preferences in cat visual cortex. Brain Res. 211, 445–450 (1981)
Swindale, N. V., Matsubara, J. A. & Cynader, M. S. Surface organization of orientation and direction selectivity in cat area 18. J. Neurosci. 7, 1414–1427 (1987)
Ohki, K., Matsuda, Y., Ajima, A., Kim, D. S. & Tanaka, S. Arrangement of orientation pinwheel centers around area 17/18 transition zone in cat visual cortex. Cereb. Cortex 10, 593–601 (2000)
Gilbert, C. D. & Wiesel, T. N. Morphology and intracortical projections of functionally characterised neurones in the cat visual cortex. Nature 280, 120–125 (1979)
Martin, K. A. & Whitteridge, D. The relationship of receptive field properties to the dendritic shape of neurones in the cat striate cortex. J. Physiol. (Lond.) 356, 291–302 (1984)
Hirsch, J. A. Synaptic physiology and receptive field structure in the early visual pathway of the cat. Cereb. Cortex 13, 63–69 (2003)
Adams, D. L. & Horton, J. C. Capricious expression of cortical columns in the primate brain. Nature Neurosci. 6, 113–114 (2003)
Rose, D. & Blakemore, C. An analysis of orientation selectivity in the cat's visual cortex. Exp. Brain Res. 20, 1–17 (1974)
Braitenberg, V. & Schuz, A. Anatomy of the Cortex: Statistics and Geometry (Springer, Berlin, 1991)
Reid, R. C. & Alonso, J. M. Specificity of monosynaptic connections from thalamus to visual cortex. Nature 378, 281–284 (1995)
Peters, A. & Yilmaz, E. Neuronal organization in area 17 of cat visual cortex. Cereb. Cortex 3, 49–68 (1993)
Mountcastle, V. B. Perceptual Neuroscience: the Cerebral Cortex (Harvard University, Cambridge, 1998)
Pologruto, T. A., Sabatini, B. L. & Svoboda, K. ScanImage: flexible software for operating laser scanning microscopes. Biomed. Eng. Online 2, 13 〈http://www.biomedical-engineering-online.com〉 (2003)
Kara, P. & Reid, R. C. Efficacy of retinal spikes in driving cortical responses. J. Neurosci. 23, 8547–8557 (2003)
Berman, N. E., Wilkes, M. E. & Payne, B. R. Organization of orientation and direction selectivity in areas 17 and 18 of cat cerebral cortex. J. Neurophysiol. 58, 676–699 (1987)
Kim, D. S., Matsuda, Y., Ohki, K., Ajima, A. & Tanaka, S. Geometrical and topological relationships between multiple functional maps in cat primary visual cortex. Neuroreport 10, 2515–2522 (1999)
Acknowledgements
We thank E. Takahashi for involvement in the first set of experiments; B. Sabatini, W. Regehr, R. Yuste and F. Engert for discussions and technical advice; S. Yurgenson for technical support and programming; A. Kerlin and J. Leong for programming; A. Vagodny for surgical assistance; and R. Yuste and J. Pezaris for comments on the manuscript. This work was supported by grants from the NEI and fellowships from the Uehara Foundation (K.O.), the Goldenson Fund (S.C.) and HHMI (Y.H.C.).Authors' contributions K.O. started this work and played the major role in the project; S.C., Y.H.C. and P.K. contributed equally to its completion.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Supplementary Figure 1
The calcium indicator OGB-1 AM preferentially stains neurons rather than glia when pressure ejected directly into layer 2/3 of the visual cortex in vivo. (JPG 57 kb)
Supplementary Figure 2
Single condition ( F/F) maps in rat and cat visual cortex. (PDF 161 kb)
Supplementary Figure 3
Columnar organization of direction discontinuity maps in cat visual cortex. (JPG 55 kb)
Supplementary Discussion
This section addresses various technical issues concerning two-photon calcium imaging that we deemed too important to omit, but were of insufficient interest to the general readership to include in the main discussion. (PDF 89 kb)
Rights and permissions
About this article
Cite this article
Ohki, K., Chung, S., Ch'ng, Y. et al. Functional imaging with cellular resolution reveals precise micro-architecture in visual cortex. Nature 433, 597–603 (2005). https://doi.org/10.1038/nature03274
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature03274
This article is cited by
-
The logic of recurrent circuits in the primary visual cortex
Nature Neuroscience (2024)
-
Stimulus edges induce orientation tuning in superior colliculus
Nature Communications (2023)
-
Distributed context-dependent choice information in mouse posterior cortex
Nature Communications (2023)
-
The cellular coding of temperature in the mammalian cortex
Nature (2023)
-
Construction and use of an adaptive optics two-photon microscope with direct wavefront sensing
Nature Protocols (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.