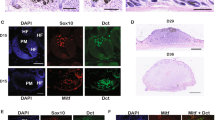
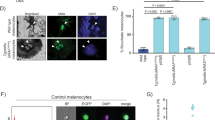
Abstract
The controls that enable melanoblasts and melanoma cells to proliferate are likely to be related, but so far no key regulator of cell cycle progression specific to the melanocyte lineage has been identified. The microphthalmia-associated transcription factor Mitf has a crucial but poorly defined role in melanoblast and melanocyte survival and in differentiation1. Here we show that Mitf can act as a novel anti-proliferative transcription factor able to induce a G1 cell-cycle arrest that is dependent on Mitf-mediated activation of the p21Cip1 (CDKN1A) cyclin-dependent kinase inhibitor gene. Moreover, cooperation between Mitf and the retinoblastoma protein Rb1 potentiates the ability of Mitf to activate transcription. The results indicate that Mitf-mediated activation of p21Cip1 expression and consequent hypophosphorylation of Rb1 will contribute to cell cycle exit and activation of the differentiation programme. The mutation of genes associated with melanoma, such as INK4a or BRAF that would affect either Mitf cooperation with Rb1 or Mitf stability respectively, would impair Mitf-mediated cell cycle control.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Vance, K. W. & Goding, C. R. The transcription network regulating melanocyte development and melanoma. Pigment Cell Res. 17, 318–325 (2004)
Bertolotto, C. et al. Microphthalmia gene product as a signal transducer in cAMP-induced differentiation of melanocytes. J. Cell Biol. 142, 827–835 (1998)
Littlewood, T. D., Hancock, D. C., Danielian, P. S., Parker, M. G. & Evan, G. I. A modified oestrogen receptor ligand-binding domain as an improved switch for the regulation of heterologous proteins. Nucleic Acids Res. 23, 1686–1690 (1995)
Quelle, D. E. et al. Cloning and characterization of murine p16INK4a and p15INK4b genes. Oncogene 11, 635–645 (1995)
Quelle, D. E., Zindy, F., Ashmun, R. A. & Sherr, C. J. Alternative reading frames of the INK4a tumor suppressor gene encode two unrelated proteins capable of inducing cell cycle arrest. Cell 83, 993–1000 (1995)
Halaban, R., Cheng, E., Zhang, Y., Mandigo, C. E. & Miglarese, M. R. Release of cell cycle constraints in mouse melanocytes by overexpressed mutant E2F1E132, but not by deletion of p16INK4a or p21WAF1/CIP1. Oncogene 16, 2489–2501 (1998)
McGill, G. G. et al. Bcl2 regulation by the melanocyte master regulator Mitf modulates lineage survival and melanoma cell viability. Cell 109, 707–718 (2002)
Aksan, I. & Goding, C. R. Targeting the microphthalmia basic helix-loop-helix-leucine zipper transcription factor to a subset of E-box elements in vitro and in vivo. Mol. Cell. Biol. 18, 6930–6938 (1998)
Bentley, N. J., Eisen, T. & Goding, C. R. Melanocyte-specific expression of the human tyrosinase promoter: activation by the microphthalmia gene product and role of the initiator. Mol. Cell. Biol. 14, 7996–8006 (1994)
Allan, L. A., Duhig, T., Read, M. & Fried, M. The p21(WAF1/CIP1) promoter is methylated in Rat-1 cells: stable restoration of p53-dependent p21(WAF1/CIP1) expression after transfection of a genomic clone containing the p21(WAF1/CIP1) gene. Mol. Cell. Biol. 20, 1291–1298 (2000)
Deng, C., Zhang, P., Harper, J. W., Elledge, S. J. & Leder, P. Mice lacking p21CIP1/WAF1 undergo normal development, but are defective in G1 checkpoint control. Cell 82, 675–684 (1995)
Yu, B. D. et al. Distinct and nonoverlapping roles for pRB and cyclin D:cyclin-dependent kinases 4/6 activity in melanocyte survival. Proc. Natl Acad. Sci. USA 100, 14881–14886 (2003)
Lerner, A. B. et al. A mouse model for vitiligo. J. Invest. Dermatol. 87, 299–304 (1986)
Yavuzer, U. et al. The microphthalmia gene product interacts with the retinoblastoma protein in vitro and is a target for deregulation of melanocyte-specific transcription. Oncogene 10, 123–134 (1995)
Chew, Y. P., Ellis, M., Wilkie, S. & Mittnacht, S. pRB phosphorylation mutants reveal role of pRB in regulating S phase completion by a mechanism independent of E2F. Oncogene 17, 2177–2186 (1998)
Takahashi, Y., Rayman, J. B. & Dynlacht, B. D. Analysis of promoter binding by the E2F and pRB families in vivo: distinct E2F proteins mediate activation and repression. Genes Dev. 14, 804–816 (2000)
Umek, R. M., Friedman, A. D. & McKnight, S. L. CCAAT-enhancer binding protein: a component of a differentiation switch. Science 251, 288–292 (1991)
Okura, M., Maeda, H., Nishikawa, S. & Mizoguchi, M. Effects of monoclonal anti-c-kit antibody (ACK2) on melanocytes in newborn mice. J. Invest. Dermatol. 105, 322–328 (1995)
Hornyak, T. J., Hayes, D. J., Chiu, L. Y. & Ziff, E. B. Transcription factors in melanocyte development: distinct roles for Pax-3 and Mitf. Mech. Dev. 101, 47–59 (2001)
Konyukhov, B. V. & Sazhina, M. V. Interaction of the genes of ocular retardation and microphthalmia in mice. Folia (Praha) 12, 116–123 (1966)
Davies, H. et al. Mutations of the BRAF gene in human cancer. Nature 417, 949–954 (2002)
Jiang, H. et al. The melanoma differentiation-associated gene mda-6, which encodes the cyclin-dependent kinase inhibitor p21, is differentially expressed during growth, differentiation and progression in human melanoma cells. Oncogene 10, 1855–1864 (1995)
Hodgkinson, C. A. et al. Mutations at the mouse microphthalmia locus are associated with defects in a gene encoding a novel basic-helix–loop–helix-zipper protein. Cell 74, 395–404 (1993)
Carreira, S., Liu, B. & Goding, C. R. The gene encoding the T-box transcription factor Tbx2 is a target for the microphthalmia-associated transcription factor in melanocytes. J. Biol. Chem. 275, 21920–21927 (2000)
Prince, S., Carreira, S., Vance, K. W., Abrahams, A. & Goding, C. R. Tbx2 directly represses the expression of the p21(WAF1) cyclin-dependent kinase inhibitor. Cancer Res. 64, 1669–1674 (2004)
Jacobs, J. J. et al. Senescence bypass screen identifies TBX2, which represses cdkn2a (p19ARF) and is amplified in a subset of human breast cancers. Nature Genet. 26, 291–299 (2000)
Vigo, E. et al. CDC25A phosphatase is a target of E2F and is required for efficient E2F-induced S phase. Mol. Cell. Biol. 19, 6379–6395 (1999)
Carreira, S., Dexter, T. J., Yavuzer, U., Easty, D. J. & Goding, C. R. Brachyury-related transcription factor Tbx2 and repression of the melanocyte-specific TRP-1 promoter. Mol. Cell. Biol. 18, 5099–5108 (1998)
Hirst, K., Fisher, F., McAndrew, P. C. & Goding, C. R. The transcription factor, the Cdk, its cyclin and their regulator: directing the transcriptional response to a nutritional signal. EMBO J. 13, 5410–5420 (1994)
Galibert, M. D., Carreira, S. & Goding, C. R. The Usf-1 transcription factor is a novel target for the stress-responsive p38 kinase and mediates UV-induced tyrosinase expression. EMBO J. 20, 5022–5031 (2001)
Acknowledgements
We thank M. Serrano for the WT and p21-null MEFs; S. Mittnacht for the Rb1 expression vectors and the C33a cells; B. Vogelstein for the p21Cip1 promoter; K. Helin for the HA.ER-expression vector; D. Stillman for yeast reporter strain DY1641; C. Wellbrock, R. Marais, R. Ballotti and C. Bertolotto for communication of unpublished results; and H. Arnheiter for discussions. This work was supported by Marie Curie Cancer Care, the Association for International Cancer Research and a European Union Marie Curie fellowship to M.-D.G.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Rights and permissions
About this article
Cite this article
Carreira, S., Goodall, J., Aksan, I. et al. Mitf cooperates with Rb1 and activates p21Cip1 expression to regulate cell cycle progression. Nature 433, 764–769 (2005). https://doi.org/10.1038/nature03269
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature03269
This article is cited by
-
Identification of BRAF, CCND1, and MYC mutations in a patient with multiple primary malignant tumors: a case report and review of the literature
World Journal of Surgical Oncology (2023)
-
Acetylation reprograms MITF target selectivity and residence time
Nature Communications (2023)
-
A unique hyperdynamic dimer interface permits small molecule perturbation of the melanoma oncoprotein MITF for melanoma therapy
Cell Research (2023)
-
Signal pathways of melanoma and targeted therapy
Signal Transduction and Targeted Therapy (2021)
-
MITF is a driver oncogene and potential therapeutic target in kidney angiomyolipoma tumors through transcriptional regulation of CYR61
Oncogene (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.