Abstract
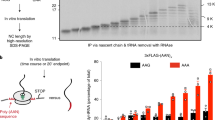
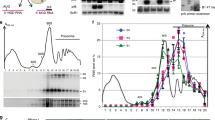
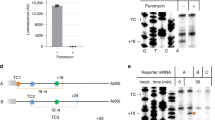
Nonsense-mediated messenger RNA decay (NMD) is triggered by premature translation termination1,2,3, but the features distinguishing premature from normal termination are unknown. One model for NMD suggests that decay-inducing factors bound to mRNAs during early processing events are routinely removed by elongating ribosomes but remain associated with mRNAs when termination is premature, triggering rapid turnover4. Recent experiments5,6,7 challenge this notion and suggest a model that posits that mRNA decay is activated by the intrinsically aberrant nature of premature termination8,9. Here we use a primer extension inhibition (toeprinting) assay10 to delineate ribosome positioning and find that premature translation termination in yeast extracts is indeed aberrant. Ribosomes encountering premature UAA or UGA codons in the CAN1 mRNA fail to release and, instead, migrate to upstream AUGs. This anomaly depends on prior nonsense codon recognition and is eliminated in extracts derived from cells lacking the principal NMD factor, Upf1p, or by flanking the nonsense codon with a normal 3′-untranslated region (UTR). Tethered poly(A)-binding protein (Pab1p), used as a mimic of a normal 3′-UTR, recruits the termination factor Sup35p (eRF3) and stabilizes nonsense-containing mRNAs. These findings indicate that efficient termination and mRNA stability are dependent on a properly configured 3′-UTR.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Jacobson, A. & Peltz, S. W. Interrelationships of the pathways of mRNA decay and translation in eukaryotic cells. Annu. Rev. Biochem. 65, 693–739 (1996)
Maquat, L. E. When cells stop making sense: effects of nonsense codons on RNA metabolism in vertebrate cells. RNA 1, 453–465 (1995)
Pulak, R. & Anderson, P. mRNA surveillance by the Caenorhabditis elegans smg genes. Genes Dev. 7, 1885–1897 (1993)
Gonzalez, C. I., Bhattacharya, A., Wang, W. & Peltz, S. W. Nonsense-mediated mRNA decay in Saccharomyces cerevisiae. Gene 274, 15–25 (2001)
Maderazo, A. B., Belk, J. P., He, F. & Jacobson, A. Nonsense-containing mRNAs that accumulate in the absence of a functional nonsense-mediated mRNA decay pathway are destabilized rapidly upon its restitution. Mol. Cell. Biol. 23, 842–851 (2003)
Gatfield, D., Unterholzner, L., Ciccarelli, F. D., Bork, P. & Izaurralde, E. Nonsense-mediated mRNA decay in Drosophila: at the intersection of the yeast and mammalian pathways. EMBO J. 22, 3960–3970 (2003)
LeBlanc, J. J. & Beemon, K. L. Unspliced Rous sarcoma virus genomic RNAs are translated and subjected to nonsense-mediated mRNA decay before packaging. J. Virol. 78, 5139–5146 (2004)
Jacobson, A. & Peltz, S. W. in Translational Control (eds Sonenberg, N., Hershey, J. W. B. & Mathews, M. B.) 827–847 (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, 2000)
Hilleren, P. & Parker, R. mRNA surveillance in eukaryotes: kinetic proofreading of proper translation termination as assessed by mRNP domain organization? RNA 5, 711–719 (1999)
Sachs, M. S. et al. Toeprint analysis of the positioning of translation apparatus components at initiation and termination codons of fungal mRNAs. Methods 26, 105–114 (2002)
Maderazo, A. B., He, F., Mangus, D. A. & Jacobson, A. Upf1p control of nonsense mRNA translation is regulated by Nmd2p and Upf3p. Mol. Cell. Biol. 20, 4591–4603 (2000)
Bonetti, B., Fu, L., Moon, J. & Bedwell, D. M. The efficiency of translation termination is determined by a synergistic interplay between upstream and downstream sequences in Saccharomyces cerevisiae. J. Mol. Biol. 251, 334–345 (1995)
Dmitriev, S. E., Pisarev, A. V., Rubtsova, M. P., Dunaevsky, Y. E. & Shatsky, I. N. Conversion of 48S translation preinitiation complexes into 80S initiation complexes as revealed by toeprinting. FEBS Lett. 533, 99–104 (2003)
Kozak, M. Primer extension analysis of eukaryotic ribosome–mRNA complexes. Nucleic Acids Res. 26, 4853–4859 (1998)
Stansfield, I., Kushnirov, V. V., Jones, K. M. & Tuite, M. F. A conditional-lethal translation termination defect in a sup45 mutant of the yeast Saccharomyces cerevisiae. Eur. J. Biochem. 245, 557–563 (1997)
Thomas, K. R. & Capecchi, M. R. Introduction of homologous DNA sequences into mammalian cells induces mutations in the cognate gene. Nature 324, 34–38 (1986)
Peabody, D. S. & Berg, P. Termination–reinitiation occurs in the translation of mammalian cell mRNAs. Mol. Cell. Biol. 6, 2695–2703 (1986)
Song, H. et al. The crystal structure of human eukaryotic release factor eRF1—mechanism of stop codon recognition and peptidyl-tRNA hydrolysis. Cell 100, 311–321 (2000)
Peltz, S. W., Brown, A. H. & Jacobson, A. mRNA destabilization triggered by premature translational termination depends on at least three cis-acting sequence elements and one trans-acting factor. Genes Dev. 7, 1737–1754 (1993)
Muhlrad, D. & Parker, R. Recognition of yeast mRNAs as ‘nonsense containing’ leads to both inhibition of mRNA translation and mRNA degradation: implications for the control of mRNA decapping. Mol. Biol. Cell 10, 3971–3978 (1999)
Mangus, D. A., Evans, M. C. & Jacobson, A. Poly(A)-binding proteins: multifunctional scaffolds for the post-transcriptional control of gene expression. Genome Biol. 4, 223.1–223.14 (2003)
Hosoda, N. et al. Translation termination factor eRF3 mediates mRNA decay through the regulation of deadenylation. J. Biol. Chem. 278, 38287–38291 (2003)
Uchida, N., Hoshino, S., Imataka, H., Sonenberg, N. & Katada, T. A novel role of the mammalian GSPT/eRF3 associating with poly(A)-binding protein in Cap/Poly(A)-dependent translation. J. Biol. Chem. 277, 50286–50292 (2002)
Kim, V. N., Kataoka, N. & Dreyfuss, G. Role of the nonsense-mediated decay factor hUpf3 in the splicing-dependent exon–exon junction complex. Science 293, 1832–1836 (2001)
Gehring, N. H., Neu-Yilik, G., Schell, T., Hentze, M. W. & Kulozik, A. E. Y14 and hUpf3b form an NMD-activating complex. Mol. Cell 11, 939–949 (2003)
Le Hir, H., Izaurralde, E., Maquat, L. E. & Moore, M. J. The spliceosome deposits multiple proteins 20–24 nucleotides upstream of mRNA exon–exon junctions. EMBO J. 19, 6860–6869 (2000)
Iizuka, N. & Sarnow, P. Translation-competent extracts from Saccharomyces cerevisiae: effects of L-A RNA, 5′ cap, and 3′ poly(A) tail on translational efficiency of mRNAs. Methods 11, 353–360 (1997)
Tarun, S. Z. Jr & Sachs, A. B. A common function for mRNA 5′ and 3′ ends in translation initiation in yeast. Genes Dev. 9, 2997–3007 (1995)
Coller, J. M., Gray, N. K. & Wickens, M. P. mRNA stabilization by poly(A) binding protein is independent of poly(A) and requires translation. Genes Dev. 12, 3226–3235 (1998)
Cosson, B. et al. Poly(A)-binding protein acts in translation termination via eukaryotic release factor 3 interaction and does not influence [PSI(+ )] propagation. Mol. Cell. Biol. 22, 3301–3315 (2002)
Acknowledgements
We thank P. Stockley, University of Leeds, for MS2 coat protein antibodies; T. Serio (Brown University) and D. C. Masison (NIH) for Sup35p antibodies; J. McCarthy (UMIST) for eIF4G and eIF4E antibodies; and M. P. Wickens (University of Wisconsin–Madison) for plasmids carrying MS2 constructs. This work was supported by grants to A.J. from the National Institutes of Health and a postdoctoral fellowship to N.A. from the Human Frontier Science Program.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Supplementary Data
Supplementary results and figure legends (DOC 49 kb)
Supplementary Figure 1
Toeprint analyses of normal and premature termination. (JPG 106 kb)
Supplementary Figure 2
Data demonstrating that ribosomes can reinitiate translation at AUG codons upstream or downstream of the stop codon. (JPG 42 kb)
Supplementary Figure 3
Toeprint analyses in sup45-2 extracts. (JPG 43 kb)
Supplementary Figure 4
Data demonstrating that tethered Pab1p stabilizes nonsense-containing mRNAs. (JPG 49 kb)
Rights and permissions
About this article
Cite this article
Amrani, N., Ganesan, R., Kervestin, S. et al. A faux 3′-UTR promotes aberrant termination and triggers nonsense- mediated mRNA decay. Nature 432, 112–118 (2004). https://doi.org/10.1038/nature03060
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature03060
This article is cited by
-
tRNA therapeutics for genetic diseases
Nature Reviews Drug Discovery (2024)
-
Nonsense-Mediated mRNA Decay: Mechanistic Insights and Physiological Significance
Molecular Biotechnology (2023)
-
An improved imaging system that corrects MS2-induced RNA destabilization
Nature Methods (2022)
-
A nascent peptide code for translational control of mRNA stability in human cells
Nature Communications (2022)
-
Deciphering the nonsense-mediated mRNA decay pathway to identify cancer cell vulnerabilities for effective cancer therapy
Journal of Experimental & Clinical Cancer Research (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.