Abstract
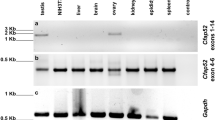
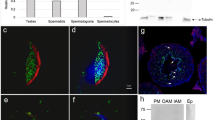
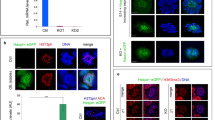
During spermatogenesis in the mammalian testis, stem cells (spermatogonia) differentiate into spermatocytes, which subsequently undergo two consecutive meiotic divisions to give rise to haploid spermatids. These cells are initially round but progressively elongate, condense their nuclei, acquire flagellar and acrosomal structures, and shed a significant amount of their cytoplasm to form spermatozoa (the sperm cells) in a developmental cascade termed spermiogenesis1,2. Defects in these processes will lead to a lack of mature sperm cells (azoospermia), which is a major cause of male infertility in the human population3. Here we report that a cell-surface protein of the immunoglobulin superfamily, junctional adhesion molecule-C (JAM-C), is critically required for the differentiation of round spermatids into spermatozoa in mice. We found that Jam-C is essential for the polarization of round spermatids, a function that we attribute to its role in the assembly of a cell polarity complex.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Cheng, C. Y. & Mruk, D. D. Cell junction dynamics in the testis: Sertoli-germ cell interactions and male contraceptive development. Physiol. Rev. 82, 825–874 (2002)
Griswold, M. D. Interactions between germ cells and Sertoli cells in the testis. Biol. Reprod. 52, 211–216 (1995)
Ezeh, U. I. Beyond the clinical classification of azoospermia: opinion. Hum. Reprod. 15, 2356–2359 (2000)
Muller, W. A. Leukocyte–endothelial-cell interactions in leukocyte transmigration and the inflammatory response. Trends Immunol. 24, 327–334 (2003)
Aurrand-Lions, M., Johnson-Leger, C. & Imhof, B. A. Role of interendothelial adhesion molecules in the control of vascular functions. Vascul. Pharmacol. 39, 239–246 (2002)
Chavakis, T., Preissner, K. T. & Santoso, S. Leukocyte trans-endothelial migration: JAMs add new pieces to the puzzle. Thromb. Haemost. 89, 13–17 (2003)
Bazzoni, G. The JAM family of junctional adhesion molecules. Curr. Opin. Cell Biol. 15, 525–530 (2003)
Ebnet, K., Suzuki, A., Ohno, S. & Vestweber, D. Junctional adhesion molecules (JAMs): more molecules with dual functions? J. Cell Sci. 117, 19–29 (2004)
Liang, T. W. et al. Vascular endothelial-junctional adhesion molecule (VE-JAM)/JAM 2 interacts with T, NK, and dendritic cells through JAM 3. J. Immunol. 168, 1618–1626 (2002)
Arrate, M. P., Rodriguez, J. M., Tran, T. M., Brock, T. A. & Cunningham, S. A. Cloning of human junctional adhesion molecule 3 (JAM3) and its identification as the JAM2 counter-receptor. J. Biol. Chem. 276, 45826–45832 (2001)
Lui, W. Y., Mruk, D., Lee, W. M. & Cheng, C. Y. Sertoli cell tight junction dynamics: their regulation during spermatogenesis. Biol. Reprod. 68, 1087–1097 (2003)
Kierszenbaum, A. L., Rivkin, E. & Tres, L. L. Acroplaxome, an F-actin-keratin-containing plate, anchors the acrosome to the nucleus during shaping of the spermatid head. Mol. Biol. Cell 14, 4628–4640 (2003)
Ozaki-Kuroda, K. et al. Nectin couples cell–cell adhesion and the actin scaffold at heterotypic testicular junctions. Curr. Biol. 12, 1145–1150 (2002)
Bouchard, M. J. et al. Defects in nuclear and cytoskeletal morphology and mitochondrial localization in spermatozoa of mice lacking nectin-2, a component of cell–cell adherens junctions. Mol. Cell. Biol. 20, 2865–2873 (2000)
Mueller, S., Rosenquist, T. A., Takai, Y., Bronson, R. A. & Wimmer, E. Loss of nectin-2 at Sertoli-spermatid junctions leads to male infertility and correlates with severe spermatozoan head and midpiece malformation, impaired binding to the zona pellucida, and oocyte penetration. Biol. Reprod. 69, 1330–1340 (2003)
Kang-Decker, N., Mantchev, G. T., Juneja, S. C., McNiven, M. A. & van Deursen, J. M. Lack of acrosome formation in Hrb-deficient mice. Science 294, 1531–1533 (2001)
Etienne-Manneville, S. & Hall, A. Cell polarity: Par6, aPKC and cytoskeletal crosstalk. Curr. Opin. Cell Biol. 15, 67–72 (2003)
Ohno, S. Intercellular junctions and cellular polarity: the PAR-aPKC complex, a conserved core cassette playing fundamental roles in cell polarity. Curr. Opin. Cell Biol. 13, 641–648 (2001)
Erickson, J. W. & Cerione, R. A. Multiple roles for Cdc42 in cell regulation. Curr. Opin. Cell Biol. 13, 153–157 (2001)
Wodarz, A. Establishing cell polarity in development. Nature Cell Biol. 4, E39–E44 (2002)
Ebnet, K. et al. The junctional adhesion molecule (JAM) family members JAM-2 and JAM-3 associate with the cell polarity protein PAR-3: a possible role for JAMs in endothelial cell polarity. J. Cell Sci. 116, 3879–3891 (2003)
Ebnet, K. et al. The cell polarity protein ASIP/PAR-3 directly associates with junctional adhesion molecule (JAM). EMBO J. 20, 3738–3748 (2001)
Roh, M. H., Fan, S., Liu, C. J. & Margolis, B. The Crumbs3–Pals1 complex participates in the establishment of polarity in mammalian epithelial cells. J. Cell Sci. 116, 2895–2906 (2003)
Straight, S. W. et al. Loss of PALS1 expression leads to tight junction and polarity defects. Mol. Biol. Cell 15, 1981–1990 (2004)
Nam, S. C. & Choi, K. W. Interaction of Par-6 and Crumbs complexes is essential for photoreceptor morphogenesis in Drosophila. Development 130, 4363–4372 (2003)
Hurd, T. W., Gao, L., Roh, M. H., Macara, I. G. & Margolis, B. Direct interaction of two polarity complexes implicated in epithelial tight junction assembly. Nature Cell Biol. 5, 137–142 (2003)
Hutterer, A., Betschinger, J., Petronczki, M. & Knoblich, J. A. Sequential roles of Cdc42, Par-6, aPKC and Lgl in the establishment of epithelial polarity during Drosophila embryogenesis. Dev. Cell 6, 845–854 (2004)
Lallemand, Y., Luria, V., Haffner-Krausz, R. & Lonai, P. Maternally expressed PGK-Cre transgene as a tool for early and uniform activation of the Cre site-specific recombinase. Transgenic Res. 7, 105–112 (1998)
Izumi, Y. et al. An atypical PKC directly associates and colocalizes at the epithelial tight junction with ASIP, a mammalian homologue of Caenorhabditis elegans polarity protein PAR-3. J. Cell Bi ol. 143, 95–106 (1998)
Compagni, A., Logan, M., Klein, R. & Adams, R. H. Control of skeletal patterning by ephrinB1–EphB interactions. Dev. Cell 5, 217–230 (2003)
Acknowledgements
We thank B. Margolis, T. Pawson, E. Peles, A. Suzuki, A. Le Bivic, C. Nobes and P. Parker for reagents and N. Hogg for comments on the manuscript. This work was funded by Cancer Research UK, the EMBO Young Investigator Program (R.H.A.), the Swiss National Science Foundation (M.A.L. and B.A.I.) and Oncosuisse (B.A.I).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Supplementary Figure 1
JAM-C gene targeting strategy and verification of the null allele. (PDF 146 kb)
Supplementary Figure 2
Histology showing the lack of mature spermatozoa in the epididymis of JAM-C-deficient mice. (PDF 176 kb)
Supplementary Figure 3
Data confirming the integrity of mutant Sertoli-Sertoli tight junctions. (PDF 214 kb)
Supplementary Figure 4
Additional immunofluorescence data for cell polarity proteins. (PDF 136 kb)
Rights and permissions
About this article
Cite this article
Gliki, G., Ebnet, K., Aurrand-Lions, M. et al. Spermatid differentiation requires the assembly of a cell polarity complex downstream of junctional adhesion molecule-C. Nature 431, 320–324 (2004). https://doi.org/10.1038/nature02877
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature02877
This article is cited by
-
A novel homozygous variant in JAM3 gene causing hemorrhagic destruction of the brain, subependymal calcification, and congenital cataracts (HDBSCC) with neonatal onset
Neurological Sciences (2021)
-
The sperm of Xenacoelomorpha revisited: implications for the evolution of early bilaterians
Zoomorphology (2019)
-
Multiple aspects of male germ cell development and interactions with Sertoli cells require inositol hexakisphosphate kinase-1
Scientific Reports (2018)
-
Dynamic of VE-cadherin-mediated spermatid–Sertoli cell contacts in the mouse seminiferous epithelium
Histochemistry and Cell Biology (2018)
-
Single-cell analyses identify bioengineered niches for enhanced maintenance of hematopoietic stem cells
Nature Communications (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.