Abstract
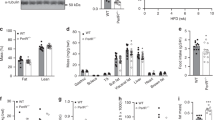
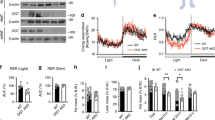
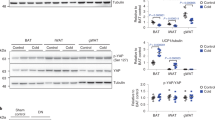
Elucidating the signalling mechanisms by which obesity leads to impaired insulin action is critical in the development of therapeutic strategies for the treatment of diabetes1. Recently, mice deficient for S6 Kinase 1 (S6K1), an effector of the mammalian target of rapamycin (mTOR) that acts to integrate nutrient and insulin signals2, were shown to be hypoinsulinaemic, glucose intolerant and have reduced β-cell mass3. However, S6K1-deficient mice maintain normal glucose levels during fasting, suggesting hypersensitivity to insulin3, raising the question of their metabolic fate as a function of age and diet. Here, we report that S6K1-deficient mice are protected against obesity owing to enhanced β-oxidation. However on a high fat diet, levels of glucose and free fatty acids still rise in S6K1-deficient mice, resulting in insulin receptor desensitization. Nevertheless, S6K1-deficient mice remain sensitive to insulin owing to the apparent loss of a negative feedback loop from S6K1 to insulin receptor substrate 1 (IRS1), which blunts S307 and S636/S639 phosphorylation; sites involved in insulin resistance4,5. Moreover, wild-type mice on a high fat diet as well as K/K Ay and ob/ob (also known as Lep/Lep) mice—two genetic models of obesity—have markedly elevated S6K1 activity and, unlike S6K1-deficient mice, increased phosphorylation of IRS1 S307 and S636/S639. Thus under conditions of nutrient satiation S6K1 negatively regulates insulin signalling.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Saltiel, A. R. & Kahn, C. R. Insulin signalling and the regulation of glucose and lipid metabolism. Nature 414, 799–806 (2001)
Fingar, D. C., Salama, S., Tsou, C., Harlow, E. & Blenis, J. Mammalian cell size is controlled by mTOR and its downstream targets S6K1 and 4EBP1/eIF4E. Genes Dev. 16, 1472–1487 (2002)
Pende, M. et al. Hypoinsulinaemia, glucose intolerance and diminished β-cell size in S6K1-deficient mice. Nature 408, 994–997 (2000)
Zick, Y. Insulin resistance: a phosphorylation-based uncoupling of insulin signaling. Trends Cell Biol. 11, 437–441 (2001)
Bouzakri, K. et al. Reduced activation of phosphatidylinositol-3 kinase and increased serine 636 phosphorylation of insulin receptor substrate-1 in primary culture of skeletal muscle cells from patients with type 2 diabetes. Diabetes 52, 1319–1325 (2003)
Tanaka, T. et al. Activation of peroxisome proliferator-activated receptor delta induces fatty acid β-oxidation in skeletal muscle and attenuates metabolic syndrome. Proc. Natl Acad. Sci. USA 100, 15924–15929 (2003)
Luquet, S. et al. Peroxisome proliferator-activated receptor delta controls muscle development and oxidative capability. FASEB J. 17, 2299–2301 (2003)
Dressel, U. et al. The peroxisome proliferator-activated receptor beta/delta agonist, GW501516, regulates the expression of genes involved in lipid catabolism and energy uncoupling in skeletal muscle cells. Mol. Endocrinol. 17, 2477–2493 (2003)
Patti, M. E. et al. Coordinated reduction of genes of oxidative metabolism in humans with insulin resistance and diabetes: Potential role of PGC1 and NRF1. Proc. Natl Acad. Sci. USA 100, 8466–8471 (2003)
Mootha, V. K. et al. PGC-1α-responsive genes involved in oxidative phosphorylation are coordinately downregulated in human diabetes. Nature Genet. 34, 267–273 (2003)
Boden, G. Role of fatty acids in the pathogenesis of insulin resistance and NIDDM. Diabetes 46, 3–10 (1997)
Kahn, B. B. Type 2 diabetes: when insulin secretion fails to compensate for insulin resistance. Cell 92, 593–596 (1998)
Le Marchand-Brustel, Y. et al. Fatty acid-induced insulin resistance: role of insulin receptor substrate 1 serine phosphorylation in the retroregulation of insulin signalling. Biochem. Soc. Trans. 31, 1152–1156 (2003)
Jiang, G. & Zhang, B. B. Pi 3-kinase and its up- and down-stream modulators as potential targets for the treatment of type II diabetes. Front. Biosci. 7, d903–d907 (2002)
Radimerski, T., Montagne, J., Hemmings-Mieszczak, M. & Thomas, G. Lethality of Drosophila lacking TSC tumor suppressor function rescued by reducing dS6K signaling. Genes Dev. 16, 2627–2632 (2002)
Kahn, B. B. & Flier, J. S. Obesity and insulin resistance. J. Clin. Invest. 106, 473–481 (2000)
Proietto, J., Filippis, A., Nakhla, C. & Clark, S. Nutrient-induced insulin resistance. Mol. Cell. Endocrinol. 151, 143–149 (1999)
Greiwe, J. S., Kwon, G., McDaniel, M. L. & Semenkovich, C. F. Leucine and insulin activate p70 S6 kinase through different pathways in human skeletal muscle. Am. J. Physiol. Endocrinol. Metab. 281, E466–E471 (2001)
Patti, M. E. Nutrient modulation of cellular insulin action. Ann. NY Acad. Sci. 892, 187–203 (1999)
Hara, K. et al. Amino acid sufficiency and mTOR regulate p70 S6 kinase and eIF-4E BP1 through a common effector mechanism. J. Biol. Chem. 273, 14484–14494 (1998)
Patti, M. E., Brambilla, E., Luzi, L., Landaker, E. J. & Kahn, C. R. Bidirectional modulation of insulin action by amino acids. J. Clin. Invest. 101, 1519–1529 (1998)
Dennis, P. B. et al. Mammalian TOR: a homeostatic ATP sensor. Science 294, 1102–1105 (2001)
Tremblay, F. & Marette, A. Amino acid and insulin signaling via the mTOR/p70 S6 kinase pathway. A negative feedback mechanism leading to insulin resistance in skeletal muscle cells. J. Biol. Chem. 276, 38052–38060 (2001)
Iwatsuka, H., Shino, A. & Suzuoki, Z. General survey of diabetic features of yellow KK mice. Endocrinol. Jpn 17, 23–35 (1970)
Zhang, Y. et al. Positional cloning of the mouse obese gene and its human homologue. Nature 372, 425–432 (1994)
Manning, B. D. & Cantley, L. C. Rheb fills a GAP between TSC and TOR. Trends Biochem. Sci. 28, 573–576 (2003)
Picard, F. et al. SRC-1 and TIF2 control energy balance between white and brown adipose tissues. Cell 111, 931–941 (2002)
Doty, F. D., Entzminger, G. Jr, Hauck, C. D. & Staab, J. P. Practical aspects of birdcage coils. J. Magn. Reson. 138, 144–154 (1999)
Marette, A., Tulp, O. L. & Bukowiecki, L. J. Mechanism linking insulin resistance to defective thermogenesis in brown adipose tissue of obese diabetic SHR/N-cp rats. Int. J. Obes. 15, 823–831 (1991)
Hirosumi, J. et al. A central role for JNK in obesity and insulin resistance. Nature 420, 333–336 (2002)
Acknowledgements
We thank T. Opgenorth and C. Rondinone for sharing their results before publication; G. S. Hotamisligil, S. Y. Kim and D. J. Withers for their critical reading of the manuscript; and S. Cinti, P. B. Dennis, A. Dulloo, L. Fajas, A. Greenberg, B. M. Spiegelman, G. Solinas, J. Tanti and M. Wymann for discussions. We are also grateful to M.-F. Champy, W. Theilkaes, N. Messaddeq, I. Obergfoell and J. F. Spetz for the blood analysis, studies with MRI, technical assistance with electron microscopy, for photography and for assistance in the animal experiments, respectively. Work in the laboratory of J.A. is supported by grants from CNRS, INSERM, ULP, Hôpital Universitaire de Strasbourg, NIH, EMBO and the European community, and the laboratory of S.C.K. and G.T. is supported by the Novartis Institutes for Biomedical Research and a grant from the Swiss Cancer League.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Supplementary Figure 1
Reduced adiposity in S6K1-/- mice. (PDF 720 kb)
Supplementary Figure 2
Increased mitochondria and resistance to diet-induced obesity. (PDF 277 kb)
Rights and permissions
About this article
Cite this article
Um, S., Frigerio, F., Watanabe, M. et al. Absence of S6K1 protects against age- and diet-induced obesity while enhancing insulin sensitivity. Nature 431, 200–205 (2004). https://doi.org/10.1038/nature02866
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature02866
This article is cited by
-
mTORC1 in energy expenditure: consequences for obesity
Nature Reviews Endocrinology (2024)
-
Identification and characterization of TOR in Macrobrachium rosenbergii and its role in muscle protein and lipid production
Scientific Reports (2024)
-
Geroprotector drugs and exercise: friends or foes on healthy longevity?
BMC Biology (2023)
-
The stress-responsive protein REDD1 and its pathophysiological functions
Experimental & Molecular Medicine (2023)
-
Enhanced Sestrin expression through Tanshinone 2A treatment improves PI3K-dependent inhibition of glioma growth
Cell Death Discovery (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.