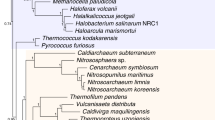
Abstract
Genomes hold within them the record of the evolution of life on Earth. But genome fusions and horizontal gene transfer seem to have obscured sufficiently the gene sequence record such that it is difficult to reconstruct the phylogenetic tree of life. Here we determine the general outline of the tree using complete genome data from representative prokaryotes and eukaryotes and a new genome analysis method that makes it possible to reconstruct ancient genome fusions and phylogenetic trees. Our analyses indicate that the eukaryotic genome resulted from a fusion of two diverse prokaryotic genomes, and therefore at the deepest levels linking prokaryotes and eukaryotes, the tree of life is actually a ring of life. One fusion partner branches from deep within an ancient photosynthetic clade, and the other is related to the archaeal prokaryotes. The eubacterial organism is either a proteobacterium, or a member of a larger photosynthetic clade that includes the Cyanobacteria and the Proteobacteria.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Lake, J. A., Sabatini, D. D. & Nonomura, Y. in Ribosomes (eds Nomura, M., Tissieres, A. & Lengyel, P.) 543–557 (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, 1974)
Woese, C. R. Archaebacteria. Sci. Am. 244, 98–105 (1981)
Dayhoff, M. O. Atlas of Protein Sequence and Structure (National Biomedical Research Foundation, Silver Spring, Maryland, 1972)
Pace, N. R., Olsen, G. J. & Woese, C. R. Ribosomal RNA phylogeny and the primary lines of evolutionary descent. Cell 45, 325–326 (1986)
Lake, J. A. Origin of the eukaryotic nucleus determined by rate-invariant analysis of ribosomal RNA sequences. Nature 331, 184–186 (1988)
Galtier, N., Tourasse, N. & Gouy, M. A nonhyperthermophilic common ancestor to extant life forms. Science 283, 220–221 (1999)
Gogarten, J. P. et al. Evolution of the vacuolar H + -Atpase—implications for the origin of eukaryotes. Proc. Natl Acad. Sci. USA 86, 6661–6665 (1989)
Iwabe, N., Kuma, K., Hasegawa, M., Osawa, S. & Miyata, T. Evolutionary relationship of archaebacteria, eubacteria, and eukaryotes inferred from phylogenetic trees of duplicated genes. Proc. Natl Acad. Sci. USA 86, 9355–9359 (1989)
Martin, W., Mustafa, A. Z., Henze, K. & Schnarrenberger, C. Higher-plant chloroplast and cytosolic fructose-1,6-bisphosphatase isoenzymes: Origins via duplication rather than prokaryote-eukaryote divergence. Plant Mol. Biol. 32, 485–491 (1996)
Brown, J. R. & Doolittle, W. F. Archaea and the prokaryote-to-eukaryote transition. Microbiol. Mol. Biol. Rev. 61, 456–502 (1997)
Feng, D. F., Cho, G. & Doolittle, R. F. Determining divergence times with a protein clock: Update and reevaluation. Proc. Natl Acad. Sci. USA 94, 13028–13033 (1997)
Gupta, R. S. Protein phylogenies and signature sequences: A reappraisal of evolutionary relationships among archaebacteria, eubacteria, and eukaryotes. Microbiol. Mol. Biol. Rev. 62, 1435–1491 (1998)
Rivera, M. C., Jain, R., Moore, J. E. & Lake, J. A. Genomic evidence for two functionally distinct gene classes. Proc. Natl Acad. Sci. USA 95, 6239–6244 (1998)
Esser, C. et al. A genome phylogeny for mitochondria among α-Proteobacteria and a predominantly eubacterial ancestry of yeast nuclear genes. Mol. Biol. Evol. doi:10.1093/molbev/msh160 (2004)
Karlin, S., Mrazek, J. & Campbell, A. M. Compositional biases of bacterial genomes and evolutionary implications. J. Bacteriol. 179, 3899–3913 (1997)
Gogarten, J. P., Hilario, E. & Olendzenski, L. The tree of life. ASM News 63, 404–405 (1997)
Doolittle, W. F. Phylogenetic classification and the universal tree. Science 284, 2124–2128 (1999)
Campbell, A. M. Lateral gene transfer in prokaryotes. Theor. Popul. Biol. 57, 71–77 (2000)
Ochman, H. & Jones, I. B. Evolutionary dynamics of full genome content in Escherichia coli. EMBO J. 19, 6637–6643 (2000)
Lake, J. A. & Rivera, M. C. Deriving the genomic tree of life in the presence of horizontal gene transfer: Conditioned Reconstruction. Mol. Biol. Evol. 21, 681–690 (2004)
Dickerson, R. E. in Diffraction and Related Studies (ed. Srinivasan, R.) 227–249 (Pergamon, Oxford/New York, 1980)
Snel, B., Bork, P. & Huynen, M. A. Genome phylogeny based on gene content. Nature Genet. 21, 108–110 (1999)
Fitz-Gibbon, S. T. & House, C. H. Whole genome-based phylogenetic analysis of free-living microorganisms. Nucleic Acids Res. 27, 4218–4222 (1999)
Tekaia, F., Lazcano, A. & Dujon, B. The genomic tree as revealed from whole proteome comparisons. Genome Res. 9, 550–557 (1999)
Montague, M. G. & Hutchison, C. A. Gene content phylogeny of herpesviruses. Proc. Natl Acad. Sci. USA 97, 5334–5339 (2000)
Lake, J. A., Henderson, E., Clark, M. W. & Matheson, A. T. Mapping evolution with ribosome structure: Intralineage constancy and interlineage variation. Proc. Natl Acad. Sci. USA 79, 5948–5952 (1982)
Gray, M. W., Burger, G. & Lang, B. F. Mitochondrial evolution. Science 283, 1476–1481 (1999)
Timmis, J. N., Ayliffe, M. A., Huang, C. Y. & Martin, W. Endosymbiotic gene transfer: Organelle genomes forge eukaryotic chromosomes. Nature Rev. Genet. 5, 123–135 (2004)
Gabaldon, T. & Huynen, M. A. Reconstruction of the proto mitochondrial metabolism. Science 301, 609 (2003)
Adams, K. L., Daley, D. O., Qiu, Y. L., Whelan, J. & Palmer, J. D. Repeated, recent and diverse transfers of a mitochondrial gene to the nucleus in flowering plants. Nature 408, 354–357 (2000)
Gray, M. W. Evolution of organellar genomes. Curr. Opin. Genet. Dev. 9, 678–687 (1999)
Collura, R. V. & Stewart, C. B. Insertions and duplications of mtDNA in the nuclear genomes of old-world monkeys and hominoids. Nature 378, 485–489 (1995)
Zischler, H., Geisert, H., vonHaseler, A. & Paabo, A. A nuclear fossil of the mitochondrial D-loop and the origin of modern humans. Nature 378, 489–492 (1995)
Margulis, L. Origin of the Eukaryotic Cells (Yale Univ. Press, New Haven, 1970)
Gupta, R. S., Aitken, K., Falah, M. & Singh, B. Cloning of Giardia lamblia heat-shock protein Hsp70 homologs—Implications regarding origin of eukaryotic cells and of endoplasmic-reticulum. Proc. Natl Acad. Sci. USA 91, 2895–2899 (1994)
Martin, W. & Muller, M. The hydrogen hypothesis for the first eukaryote. Nature 392, 37–41 (1998)
Lake, J. A. & Rivera, M. C. Was the nucleus the 1st endosymbiont. Proc. Natl Acad. Sci. USA 91, 2880–2881 (1994)
Moreira, D. & Lopez-Garcia, P. Symbiosis between methanogenic archaea and δ-proteobacteria as the origin of eukaryotes: The syntrophic hypothesis. J. Mol. Evol. 47, 517–530 (1998)
Horiike, T., Hamada, K., Kanaya, S. & Shinozawa, T. Origin of eukaryotic cell nuclei by symbiosis of Archaea in Bacteria is revealed by homology-hit analysis. Nature Cell Biol. 3, 210–214 (2001)
Rivera, M. C. & Lake, J. A. Evidence that eukaryotes and eocyte prokaryotes are immediate relatives. Science 257, 74–76 (1992)
Daubin, V., Gouy, M. & Perriere, B. A phylogenomic approach to bacterial phylogeny: Evidence of a core of genes sharing a common history. Genome Res. 12, 1080–1090 (2002)
Brochier, C., Forterre, P. & Gribaldo, S. Archaeal phylogeny based on proteins of the transcription and translation machineries: tackling the Methanopyrus kandleri paradox. Genome Biol. 5, R17 (2004)
Wolf, Y. I., Rogozin, I. B., Grishin, N. V., Tatusov, R. L. & Koonin, E. V. Genome trees constructed using five different approaches suggest new major bacterial clades. BMC Evol. Biol. 1, 8 (2001)
Altschul, S. F. et al. Gapped BLAST and PSI_BLAST: a new generation of protein database search programs. Nucleic Acids Res. 25, 3389–3402 (1997)
Lake, J. A. Reconstructing evolutionary trees from DNA and protein sequences—Paralinear distances. Proc. Natl Acad. Sci. USA 91, 1455–1459 (1994)
Lockhart, P. J., Steel, M. A., Hendy, M. D. & Penny, D. Recovering evolutionary trees under a more realistic model of sequence evolution. Mol. Biol. Evol. 11, 605–612 (1994)
Lake, J. A. Calculating the probability of multitaxon evolutionary trees—Bootstrappers Gambit. Proc. Natl Acad. Sci. USA 92, 9662–9666 (1995)
Lake, J. A. Optimally recovering rate variation information from genomes and sequences: Pattern filtering. Mol. Biol. Evol. 15, 1224–1231 (1998)
Woese, C. R., Kandler, O. & Wheelis, M. L. Towards a natural system of organisms: Proposal for the domains Archaea, Bacteria and Eucarya. Proc. Natl Acad. Sci. USA 87, 4576–4579 (1990)
Acknowledgements
We thank A. B. Simonson for helpful discussions and editing, M. Kowalczyk for illustrations, J. Garey for suggesting the original title ‘One ring to rule them all’, and J. A. Servin and R. G. Skophammer for suggestions. This work was supported by grants from the National Science Foundation, NASA Astrobiology Program, The Department of Energy, and the National Institutes of Health to J.A.L.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Supplementary Data
This file contains three figures, 13 references, an Introduction and two analysis sections (“Reconstructing the Prokaryotic Tree of Life” and “Assessing whether the choice of conditioning genomes has affected the ring of life?”). (DOC 413 kb)
Supplementary Discussion
This file contains 1 figure and 3 references. It presents a rationale for classifying the tree of life when genome fusions have created new phylogenetic groups. (DOC 47 kb)
Rights and permissions
About this article
Cite this article
Rivera, M., Lake, J. The ring of life provides evidence for a genome fusion origin of eukaryotes. Nature 431, 152–155 (2004). https://doi.org/10.1038/nature02848
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature02848
This article is cited by
-
Eukaryogenesis and oxygen in Earth history
Nature Ecology & Evolution (2022)
-
On biological individuation
Theory in Biosciences (2022)
-
Division of labour in a matrix, rather than phagocytosis or endosymbiosis, as a route for the origin of eukaryotic cells
Biology Direct (2020)
-
Genome and pan-genome analysis to classify emerging bacteria
Biology Direct (2019)
-
PCR-based Sepsis@Quick test is superior in comparison with blood culture for identification of sepsis-causative pathogens
Scientific Reports (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.