Abstract
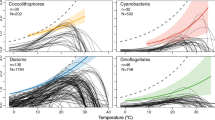
Phenology, the study of annually recurring life cycle events such as the timing of migrations and flowering, can provide particularly sensitive indicators of climate change1. Changes in phenology may be important to ecosystem function because the level of response to climate change may vary across functional groups and multiple trophic levels. The decoupling of phenological relationships will have important ramifications for trophic interactions, altering food-web structures and leading to eventual ecosystem-level changes. Temperate marine environments may be particularly vulnerable to these changes because the recruitment success of higher trophic levels is highly dependent on synchronization with pulsed planktonic production2,3. Using long-term data of 66 plankton taxa during the period from 1958 to 2002, we investigated whether climate warming signals4 are emergent across all trophic levels and functional groups within an ecological community. Here we show that not only is the marine pelagic community responding to climate changes, but also that the level of response differs throughout the community and the seasonal cycle, leading to a mismatch between trophic levels and functional groups.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Hughes, L. Biological consequences of global warming: is the signal already apparent? Trends Ecol. Evol. 15, 56–61 (2000)
Hjort, J. Fluctuations in the great fisheries of northern Europe. Rapp. Conserv. Explor. Mer 20, 1–228 (1914)
Cushing, D. H. Plankton production and year-class strength in fish population: an update of the match/mismatch hypothesis. Adv. Mar. Biol. 26, 250–293 (1990)
Root, T. L. et al. Fingerprints of global warming on wild animals and plants. Nature 421, 57–60 (2003)
Walther, G. et al. Ecological responses to recent climate change. Nature 416, 389–395 (2002)
Parmesan, C. & Yohe, G. A globally coherent fingerprint of climate change impacts across natural systems. Nature 421, 37–42 (2003)
Harrington, R., Woiwod, I. & Sparks, T. Climate change and trophic interactions. Trends Ecol. Evol. 14, 146–150 (1999)
Warner, A. J. & Hays, G. C. Sampling by the Continuous Plankton Recorder survey. Prog. In Oceanogr. 34, 237–256 (1994)
Sverdrup, H. U. On conditions for the vernal blooming of phytoplankton. J. Cons. Perm. Int. Explor. Mer. 18, 287–295 (1953)
Siegismund, F. & Schrum, C. Decadal changes in wind forcing over the North Sea. Clim. Res. 18, 39–45 (2001)
Eilertsen, H. C., Sandberg, S. & Tollefsen, H. Photoperiodic control of diatom spore growth; a theory to explain the onset of phytoplankton blooms. Mar. Ecol. Prog. Ser. 116, 303–307 (1995)
McQuoid, M. R. & Hobson, L. A. Diatom resting stages. J. Phycol. 32, 889–902 (1996)
Townsend, D. W., Keller, M. D., Sieracki, M. E. & Ackleson, S. G. Spring phytoplankton blooms in the absence of vertical water column stratification. Nature 360, 59–62 (1992)
van Haren, H., Mills, D. K. & Wetsteyn, L. P. M. J. Detailed observations of the phytoplankton spring bloom in the stratifying central North Sea. J. Mar. Res. 56, 655–680 (1998)
Eilertsen, H. C. & Wyatt, T. Phytoplankton models and life history strategies. S. Afr. J. Mar. Sci 22, 323–338 (2000)
Greve, W. in Phenology: An Integrative Environmental Science (ed. Schwartz, M. D.) 385–403 (Kluwer, Netherlands, 2004)
Wear, R. G. Incubation in British decapod Crustacea and the effects of temperature on the rate and success of embryonic development. J. Mar. Biol. Assoc. UK 54, 745–762 (1974)
Lindley, J. A. Diversity, biomass and production of decapod crustacean larvae in a changing environment. Invertebr. Reprod. Dev. 33, 209–219 (1998)
Hirst, A. G. & Bunker, A. J. Growth of marine planktonic copepods: global rates and patterns in relation to chlorophyll a, temperature, and body weight. Limnol. Oceanogr. 48, 1988–2010 (2003)
Greve, W., Reiners, F. & Nast, J. Biocoenotic changes of the zooplankton in German Bight: the possible effects of eutrophication and climate. ICES J. Mar. Sci. 53, 951–956 (1996)
Van Beusekom, J. E. E. & Diel-Christensen, S. North Sea Quality Status Report 64–71 (Danish Environmental Protection Agency, Copenhagen, 1994)
Beaugrand, G., Brander, K. M., Lindley, J. A., Souissi, S. & Reid, P. C. Plankton effect on cod recruitment in the North Sea. Nature 426, 661–664 (2003)
Beaugrand, G., Reid, P. C., Ibanez, F., Lindley, J. A. & Edwards, M. Reorganisation of North Atlantic marine copepod biodiversity and climate. Science 296, 1692–1694 (2002)
Schiermeier, Q. Climate findings let fishermen off the hook. Nature 428, 4 (2004)
McCarty, J. P. Ecological consequences of recent climate change. Conserv. Biol. 15, 320–331 (2001)
Colebrook, J. M. & Robinson, G. A. Continuous Plankton Records: seasonal cycles of phytoplankton and copepods in the north-eastern Atlantic and the North Sea. Bull. Mar. Ecol. 6, 123–139 (1965)
Colebrook, J. M. Continuous Plankton Records: seasonal cycles of phytoplankton and copepods in the north Atlantic Ocean and the North Sea. Mar. Biol. 51, 23–32 (1979)
Beaugrand, G., Ibanez, F. & Lindley, J. A. An overview of statistical method applied to the CPR data. Prog. In Oceanogr. 58, 235–262 (2003)
Pyper, B. J. & Peterman, R. M. Comparison of methods to account for autocorrelation in correlation analyses of fish data. Can. J. Fish. Aquat. Sci. 55, 2127–2140 (1998)
Acknowledgements
A funding consortium made up of governmental agencies from Canada, France, Iceland, Ireland, the Netherlands, Portugal, the UK and the USA financially supports the CPR survey. Main support for this work was provided by UK DEFRA and UK NERC. We would also like to thank J. Bishop, K. Brander, B. Clarke, R. Harris, R. Myers, D. Schoeman and A. Walne for comments on the manuscript and the owners and crews of the ships that tow the CPRs on a voluntary basis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Supplementary Figure 1
Supplementary Figure 1 contains additional information on a species-by-species basis. Individual taxa correlations between the time series of taxa phenology and Sea Surface Temperature are shown in the figure. (PDF 215 kb)
Rights and permissions
About this article
Cite this article
Edwards, M., Richardson, A. Impact of climate change on marine pelagic phenology and trophic mismatch. Nature 430, 881–884 (2004). https://doi.org/10.1038/nature02808
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature02808
This article is cited by
-
Adaptive strategies of high and low nucleic acid prokaryotes in response to declining resource availability and selective grazing by protozoa
Science China Earth Sciences (2024)
-
Good eutrophication status is a challenging goal for coastal waters
Ambio (2024)
-
The pace of shifting seasons in lakes
Nature Communications (2023)
-
Climate-driven zooplankton shifts cause large-scale declines in food quality for fish
Nature Climate Change (2023)
-
The response of zooplankton network indicators to winter water warming using shallow artificial reservoirs as model case study
Scientific Reports (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.