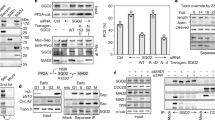
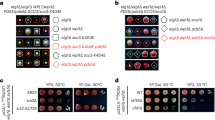
Abstract
Sister chromatids are held together by cohesins1. At anaphase, separase is activated by degradation of its inhibitory partner, securin2,3. Separase then cleaves cohesins4,5,6, thus allowing sister chromatid separation. Fission yeast securin (Cut2) has destruction boxes and a separase (Cut1) interaction site in the amino and carboxyl terminus, respectively7,8. Here we show that securin is essential for separase stability and also for proper repair of DNA damaged by ultraviolet, X-ray and γ-ray irradiation. The cut2EA2 mutant is defective in the repair of ultraviolet damage lesions, although the DNA damage checkpoint is activated normally. In double mutant analysis of ultraviolet sensitivity, checkpoint kinase chk1 (ref. 9) and excision repair rad13 (ref. 10) mutants were additive with cut2EA2, whereas recombination repair rhp51 (ref. 11) and cohesin subunit rad21 (ref. 12) mutants were not. Cohesin was hyper-modified on ultraviolet irradiation in a Rad3 kinase-dependent way13. Experiments using either mutant cohesin that cannot be cleaved by separase or a protease-dead separase provide evidence that this DNA repair function of securin–separase acts through the cleavage of cohesin. We propose that the securin–separase complex might aid DNA repair by removing local cohesin in interphase cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Nasmyth, K. Segregating sister genomes: the molecular biology of chromosome separation. Science 297, 559–565 (2002)
Funabiki, H. et al. Cut2 proteolysis required for sister-chromatid seperation in fission yeast. Nature 381, 438–441 (1996)
Cohen-Fix, O., Peters, J. M., Kirschner, M. W. & Koshland, D. Anaphase initiation in Saccharomyces cerevisiae is controlled by the APC-dependent degradation of the anaphase inhibitor Pds1p. Genes Dev. 10, 3081–3093 (1996)
Uhlmann, F., Lottspeich, F. & Nasmyth, K. Sister-chromatid separation at anaphase onset is promoted by cleavage of the cohesin subunit Scc1. Nature 400, 37–42 (1999)
Uhlmann, F., Wernic, D., Poupart, M. A., Koonin, E. V. & Nasmyth, K. Cleavage of cohesin by the CD clan protease separin triggers anaphase in yeast. Cell 103, 375–386 (2000)
Hauf, S., Waizenegger, I. C. & Peters, J. M. Cohesin cleavage by separase required for anaphase and cytokinesis in human cells. Science 293, 1320–1323 (2001)
Kumada, K. et al. Cut1 is loaded onto the spindle by binding to Cut2 and promotes anaphase spindle movement upon Cut2 proteolysis. Curr. Biol. 8, 633–641 (1998)
Jager, H., Herzig, A., Lehner, C. F. & Heidmann, S. Drosophila separase is required for sister chromatid separation and binds to PIM and THR. Genes Dev. 15, 2572–2584 (2001)
Walworth, N., Davey, S. & Beach, D. Fission yeast chk1 protein kinase links the rad checkpoint pathway to cdc2. Nature 363, 368–371 (1993)
Carr, A. M. et al. Evolutionary conservation of excision repair in Schizosaccharomyces pombe: evidence for a family of sequences related to the Saccharomyces cerevisiae RAD2 gene. Nucleic Acids Res. 21, 1345–1349 (1993)
Muris, D. F. et al. Cloning the RAD51 homologue of Schizosaccharomyces pombe. Nucleic Acids Res. 21, 4586–4591 (1993)
Birkenbihl, R. P. & Subramani, S. Cloning and characterization of rad21 an essential gene of Schizosaccharomyces pombe involved in DNA double-strand-break repair. Nucleic Acids Res. 20, 6605–6611 (1992)
Bentley, N. J. et al. The Schizosaccharomyces pombe rad3 checkpoint gene. EMBO J. 15, 6641–6651 (1996)
Rao, H., Uhlmann, F., Nasmyth, K. & Varshavsky, A. Degradation of a cohesin subunit by the N-end rule pathway is essential for chromosome stability. Nature 410, 955–959 (2001)
Jallepalli, P. V. et al. Securin is required for chromosomal stability in human cells. Cell 105, 445–457 (2001)
Saka, Y., Esashi, F., Matsusaka, T., Mochida, S. & Yanagida, M. Damage and replication checkpoint control in fission yeast is ensured by interactions of Crb2, a protein with BRCT motif, with Cut5 and Chk1. Genes Dev. 11, 3387–3400 (1997)
Lindsay, H. D. et al. S-phase-specific activation of Cds1 kinase defines a subpathway of the checkpoint response in Schizosaccharomyces pombe. Genes Dev. 12, 382–395 (1998)
Stewart, E., Chapman, C. R., Al-Khodairy, F., Carr, A. M. & Enoch, T. rqh1+, a fission yeast gene related to the Bloom's and Werner's syndrome genes, is required for reversible S phase arrest. EMBO J. 16, 2682–2692 (1997)
Murakami, H. & Okayama, H. A kinase from fission yeast responsible for blocking mitosis in S phase. Nature 374, 817–819 (1995)
Yonemasu, R. et al. Characterization of the alternative excision repair pathway of UV-damaged DNA in Schizosaccharomyces pombe. Nucleic Acids Res. 25, 1553–1558 (1997)
Nakamura, T., Nagao, K., Nakaseko, Y. & Yanagida, M. Cut1/separase C-terminus affects spindle pole body positioning in interphase of fission yeast: pointed nuclear formation. Genes Cells 7, 1113–1124 (2002)
Tomonaga, T. et al. Characterization of fission yeast cohesin: essential anaphase proteolysis of Rad21 phosphorylated in the S phase. Genes Dev. 14, 2757–2770 (2000)
Yamamoto, A., Guacci, V. & Koshland, D. Pds1p, an inhibitor of anaphase in budding yeast, plays a critical role in the APC and checkpoint pathway(s). J. Cell Biol. 133, 99–110 (1996)
Sanchez, Y. et al. Control of the DNA damage checkpoint by chk1 and rad53 protein kinases through distinct mechanisms. Science 286, 1166–1171 (1999)
Bernal, J. A. et al. Human securin interacts with p53 and modulates p53-mediated transcriptional activity and apoptosis. Nature Genet. 32, 306–311 (2002)
Aono, N., Sutani, T., Tomonaga, T., Mochida, S. & Yanagida, M. Cnd2 has dual roles in mitotic condensation and interphase. Nature 417, 197–202 (2002)
Hirano, T., Funahashi, S., Uemura, T. & Yanagida, M. Isolation and characterization of Schizosaccharomyces pombe cut mutants that block nuclear division but not cytokinesis. EMBO J. 5, 2973–2979 (1986)
Uzawa, S., Samejima, I., Hirano, T., Tanaka, K. & Yanagida, M. The fission yeast cut1+ gene regulates spindle pole body duplication and has homology to the budding yeast ESP1 gene. Cell 62, 913–925 (1990)
Esashi, F. & Yanagida, M. Cdc2 phosphorylation of Crb2 is required for reestablishing cell cycle progression after the damage checkpoint. Mol. Cell 4, 167–174 (1999)
Acknowledgements
We would like to thank C. Shimoda and T. Nakamura for the S. pombe genomic library, T. Carr, H. Okayama and N. Walworth for strains, and T. Matsumoto for introducing X-ray and γ-ray irradiation. This study was supported by the COE Grant of the Ministry of Education, Culture, Sports, Science and Technology of Japan. K.N. was a recipient of a predoctoral fellowship from the Japan Society for Promotion of Science.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Nagao, K., Adachi, Y. & Yanagida, M. Separase-mediated cleavage of cohesin at interphase is required for DNA repair. Nature 430, 1044–1048 (2004). https://doi.org/10.1038/nature02803
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature02803
This article is cited by
-
Separase activity distribution can be a marker of major molecular response and proliferation of CD34+ cells in TKI-treated chronic myeloid leukemia patients
Annals of Hematology (2020)
-
Separase is a marker for prognosis and mitotic activity in breast cancer
British Journal of Cancer (2017)
-
Cryo-EM structure of a metazoan separase–securin complex at near-atomic resolution
Nature Structural & Molecular Biology (2017)
-
PLK1 regulation of PCNT cleavage ensures fidelity of centriole separation during mitotic exit
Nature Communications (2015)
-
Overexpression and constitutive nuclear localization of cohesin protease Separase protein correlates with high incidence of relapse and reduced overall survival in glioblastoma multiforme
Journal of Neuro-Oncology (2014)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.