Abstract
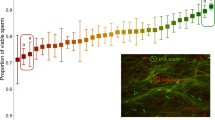
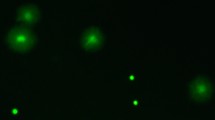
Mating with more than one male is the norm for females of many species. In addition to generating competition between the ejaculates of different males1,2, multiple mating may allow females to bias sperm use3,4. In Drosophila melanogaster, the last male to inseminate a female sires approximately 80% of subsequent progeny5. Both sperm displacement, where resident sperm are removed from storage by the incoming ejaculate of the copulating male6, and sperm incapacitation, where incoming seminal fluids supposedly interfere with resident sperm7, have been implicated in this pattern of sperm use5,6,7,8,9,10,11,12. But the idea of incapacitation is problematic because there are no known mechanisms by which an individual could damage rival sperm and not their own. Females also influence the process of sperm use13,14, but exactly how is unclear. Here we show that seminal fluids do not kill rival sperm and that any ‘incapacitation’ is probably due to sperm ageing during sperm storage. We also show that females release stored sperm from the reproductive tract (sperm dumping) after copulation with a second male and that this requires neither incoming sperm nor seminal fluids. Instead, males may cause stored sperm to be dumped or females may differentially eject sperm from the previous mating.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Simmons, L. W. Sperm Competition and its Evolutionary Consequences in the Insects (Princeton Univ. Press, Princeton, 2001)
Parker, G. A. Sperm competition and its evolutionary consequences in the insects. Biol. Rev. 45, 525–567 (1970)
Eberhard, W. G. Female Control: Sexual Selection by Cryptic Female Choice (Princeton Univ. Press, Princeton, 1996)
Pitnick, S. & Brown, W. D. Criteria for demonstrating female sperm choice. Evolution 54, 1052–1056 (2000)
Gromko, M. H., Gilbert, D. G. & Richmond, R. C. in Sperm Competition and the Evolution of Animal Mating Systems (ed. Smith, R. L.) 371–426 (Academic, London, 1984)
Lefevre, G. J. & Jonsson, U. B. Sperm transfer, storage, displacement, and utilization in Drosophila melanogaster. Genetics 47, 1719–1736 (1962)
Harshman, L. G. & Prout, T. Sperm displacement without sperm transfer in Drosophila melanogaster. Evolution 48, 758–766 (1994)
Price, C. S. P., Dyer, K. A. & Coyne, J. A. Sperm competition between Drosophila males involves both displacement and incapacitation. Nature 400, 449–452 (1999)
Civetta, A. Direct visualization of sperm competition and sperm storage in Drosophila. Curr. Biol. 9, 841–844 (1999)
Gilchrist, A. S. & Partridge, L. Male identity and sperm displacement in Drosophila melanogaster. J. Insect Physiol. 41, 1087–1092 (1995)
Scott, D. & Richmond, R. C. Sperm loss by remating Drosophila melanogaster females. J. Insect Physiol. 36, 451–456 (1990)
Gilchrist, A. S. & Partridge, L. Why is it difficult to model sperm displacement in Drosophila melanogaster: the relation between sperm transfer and copulation duration. Evolution 54, 534–542 (2000)
Miller, G. T. & Pitnick, S. Sperm–female coevolution in Drosophila. Science 298, 1230–1233 (2002)
Clark, A. G., Begun, D. J. & Prout, T. Female × male interactions in Drosophila sperm competition. Science 283, 217–220 (1999)
Birkhead, T. R. & Møller, A. P. Sperm Competition and Sexual Selection (Academic, London, 1998)
Simmons, L. W. & Siva-Jothy, M. S. in Sperm Competition and Sexual Selection (eds Birkhead, T. R. & Møller, A. P.) 341–434 (Academic Press, London, 1998)
Moore, H. D. M., Martin, M. & Birkhead, T. R. No evidence for killer sperm or other selective interactions between human spermatozoa in ejaculates of different males in vitro. Proc. R. Soc. Lond. B 266, 2343–2350 (1999)
Jenzer, B. & Steinmann-Zwicky, M. Cell-autonomous and somatic signals control sex-specific gene expression in XY germ cells of Drosophila. Mech. Dev. 100, 3–13 (2001)
Wheeler, M. R. The insemination reaction in intraspecific matings. Univ. Texas Publ. Genet. 4720, 78–115 (1947)
Heed, W. B. in Genetics, Speciation, and the Founder Principle (eds Giddings, L. V., Kaneshiro, K. Y. & Anderson, W. W.) 253–278 (Oxford Univ. Press, Oxford, 1990)
Alonzo-Pimentel, H., Tolbert, L. P. & Heed, W. B. Ultrastructural examination of the insemination reaction in Drosophila. Cell Tissue Res. 275, 467–479 (1994)
Arthur, B. I., Hauschteck-Jungen, E., Nothiger, R. & Ward, P. I. A female nervous system is necessary for normal sperm storage in Drosophila melanogaster: a masculinized nervous system is as good as none. Proc. R. Soc. Lond. B 265, 1749–1753 (1998)
Xue, L. & Noll, M. Drosophila female sexual behavior induced by sterile males showing copulation complementation. Proc. Natl Acad. Sci. USA 97, 3272–3275 (2000)
Gromko, M. H., Newport, M. A. E. & Kortier, M. G. Sperm dependence of female receptivity to remating in Drosophila melanogaster. Evolution 38, 1273–1282 (1984)
Pizzari, T. & Birkhead, T. R. Female feral fowl eject sperm of subdominant males. Nature 405, 787–789 (2000)
Cordoba-Aguilar, A. Male copulatory sensory stimulation induces female ejection of rival sperm in a damselfly. Proc. R. Soc. Lond. B 266, 779–784 (1999)
Otronen, M. & Siva-Jothy, M. T. The effects of postcopulatory male behaviour on ejaculate distribution within the female sperm storage organs of the fly Dryomyza anilis (Diptera: Dryomyzidae). Behav. Ecol. Sociobiol. 29, 33–37 (1991)
Civetta, A. & Clarke, A. G. Chromosomal effects on male and female components of sperm precedence in Drosophila. Genet. Res. 75, 143–151 (2000)
DeVries, J. K. Insemination and sperm storage in Drosophila melanogaster. Evolution 18, 271–282 (1964)
Hunter, F. M. & Birkhead, T. R. Sperm viability and sperm competition in insects. Curr. Biol. 12, 121–123 (2002)
Acknowledgements
We thank E. Kubli, J. Peng, Y. Choffat, M. Steinmann-Zwicky, J. Henner, M. Noll and E. Frei for supplying flies. Without the generosity of these people, this project would not have been possible. Further thanks go to Carl Zeiss AG for providing a microscope while we tested the sperm staining technique, to M. Oswald for his technical help, and to the Zoology Museum for support and for financing accommodation for R.R.S. during one visit to Zürich. We also thank T. Birkhead, B. Holland, J. Kotiaho, L. Simmons, M. Siva-Jothy, J. Slate, P. Stockley and T. Tregenza for comments on the work, and W. Blanckenhorn for statistical advice. This work was supported by the SNF (D.J.H.) and the US National Science Foundation (R.R.S.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Rights and permissions
About this article
Cite this article
Snook, R., Hosken, D. Sperm death and dumping in Drosophila. Nature 428, 939–941 (2004). https://doi.org/10.1038/nature02455
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature02455
This article is cited by
-
Replenishment of Drosophila Male Pheromone After Mating
Journal of Chemical Ecology (2024)
-
Polyandrous mating increases offspring production and lifespan in female Drosophila arizonae
Behavioral Ecology and Sociobiology (2018)
-
Drosophila melanogaster females restore their attractiveness after mating by removing male anti-aphrodisiac pheromones
Nature Communications (2016)
-
Multiple mechanisms of cryptic female choice act on intraspecific male variation in Drosophila simulans
Behavioral Ecology and Sociobiology (2016)
-
Age-Related Sperm Production, Transfer, and Storage in the Sweet Potato Weevil, Cylas formicarius (Fabricius) (Coleoptera: Curculionidae)
Journal of Insect Behavior (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.