Abstract
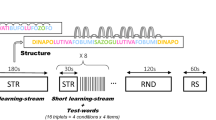
Memory consolidation resulting from sleep has been seen broadly: in verbal list learning1, spatial learning2,3, and skill acquisition in visual4,5,6,7,8 and motor9,10,11 tasks. These tasks do not generalize across spatial locations or motor sequences, or to different stimuli in the same location5,11,12. Although episodic rote learning constitutes a large part of any organism's learning, generalization is a hallmark of adaptive behaviour13. In speech, the same phoneme often has different acoustic patterns depending on context. Training on a small set of words improves performance on novel words using the same phonemes but with different acoustic patterns, demonstrating perceptual generalization14. Here we show a role of sleep in the consolidation of a naturalistic spoken-language learning task that produces generalization of phonological categories across different acoustic patterns. Recognition performance immediately after training showed a significant improvement that subsequently degraded over the span of a day's retention interval, but completely recovered following sleep. Thus, sleep facilitates the recovery and subsequent retention of material learned opportunistically at any time throughout the day. Performance recovery indicates that representations and mappings associated with generalization are refined and stabilized during sleep.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Ficca, G., Lombardo, P., Rossi, L. & Salzarulo, P. Morning recall of verbal material depends on prior sleep organization. Behav. Brain Res. 112, 159–163 (2000)
Smith, C. & Rose, G. Posttraining paradoxical sleep in rats is increased after spatial learning in the Morris water maze. Behav. Neurosci. 111, 1197–1204 (1997)
Plihal, W. & Born, J. Effects of early and late nocturnal sleep on priming and spatial memory. Psychophysiol. 36, 571–582 (1999)
Karni, A., Tanne, D., Rubenstein, B. S., Askenasy, J. J. M. & Sagi, D. Dependence on REM sleep of overnight perceptual skill. Science 265, 679–682 (1994)
Karni, A. & Sagi, D. The time course of learning a visual skill. Nature 365, 250–252 (1993)
Mednick, S. C. et al. The restorative effect of naps on perceptual deterioration. Nature Neurosci. 5, 677–681 (2002)
Stickgold, R., James, L. & Hobson, J. A. Visual discrimination learning requires sleep after training. Nature Neurosci. 3, 1237–1238 (2000)
Stickgold, R., Whidbee, D., Schirmer, B., Patel, V. & Hobson, J. A. Visual discrimination task improvement: A multi-step process occurring during sleep. J. Cogn. Neurosci. 12, 246–254 (2000)
Brashers-Krug, T., Shadmehr, R. & Bizzi, E. Consolidation in human motor memory. Nature 382, 252–255 (1996)
Walker, M. P., Brakefield, T., Morgan, A., Hobson, J. A. & Stickgold, R. Practice with sleep makes perfect: sleep-dependent motor skill learning. Neuron 35, 205–211 (2002)
Karni, A. et al. The acquisition of skilled motor performance: Fast and slow experience-driven changes in primary motor cortex. Proc. Natl Acad. Sci. USA 95, 861–868 (1998)
Karni, A. et al. Functional MRI evidence for adult motor cortex plasticity during motor skill learning. Nature 377, 155–158 (1995)
Karni, A. & Bertini, G. Learning perceptual skills: behavioral probes into adult cortical plasticity. Curr. Opin. Neurobiol. 7, 530–535 (1997)
Schwab, E. C., Nusbaum, H. C. & Pisoni, D. B. Some effects of training on the perception of synthetic speech. Hum. Factors 27, 395–408 (1985)
Greenspan, S. L., Nusbaum, H. C. & Pisoni, D. B. Perceptual learning of synthetic speech produced by rule. J. Exp. Psychol. Learn. Mem. Cogn. 14, 421–433 (1988)
Johnson, M. P. et al. Short-term memory, alertness and performance: a reappraisal of their relationship to body temperature. J. Sleep Res. 1, 24–29 (1992)
Gabrieli, J. D. E. Cognitive neuroscience of human memory. Annu. Rev. Psychol. 49, 87–115 (1998)
Liberman, A. M., Cooper, F. S., Shankweiler, D. P. & Studdert-Kennedy, M. Perception of the speech code. Psychol. Rev. 74, 431–461 (1967)
Ohman, S. E. Coarticulation in VCV utterances: Spectrographic measurements. J. Acoust. Soc. Am. 39, 151–168 (1966)
Rudnicky, A. I. & Cole, R. A. Effect of subsequent context on syllable perception. J. Exp. Psychol. Hum. Percept. Perform. 4, 638–647 (1978)
Nusbaum, H. C. & Pisoni, D. B. Automatic measurement of speech recognition performance: A comparison of six speaker-dependent recognition devices. Comp. Speech Lang. 2, 87–108 (1987)
Pisoni, D. B. in Handbook of Learning and Cognitive Processes Vol. 6 Linguistic Functions in Cognitive Theory (ed. Estes, W. K.) 167–233 (Lawrence Erlbaum Assoc., Hillsdale, New Jersey, 1978)
Kolers, P. A. & Perkins, D. N. Orientation of letters and errors in their recognition. Percept. Psychophys. 5, 265–269 (1969)
Egan, J. P. Articulation testing methods. Laryngoscope 58, 955–991 (1948)
Acknowledgements
We thank E. Van Cauter, A. S. Henly and N. P. Issa for critiques of the manuscript. This work was supported by grants from the McCormick Tribune Foundation to H.C.N. and from the National Institutes of Health to D.M.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Rights and permissions
About this article
Cite this article
Fenn, K., Nusbaum, H. & Margoliash, D. Consolidation during sleep of perceptual learning of spoken language. Nature 425, 614–616 (2003). https://doi.org/10.1038/nature01951
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature01951
This article is cited by
-
Optimizing the methodology of human sleep and memory research
Nature Reviews Psychology (2023)
-
Sleep’s role in updating aversive autobiographical memories
Translational Psychiatry (2022)
-
Sleep disorders in elementary school children with childhood apraxia of speech
Somnologie (2022)
-
The effect of training intensity on implicit learning rates in schizophrenia
Scientific Reports (2021)
-
Schematic memories develop quickly, but are not expressed unless necessary
Scientific Reports (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.