Abstract
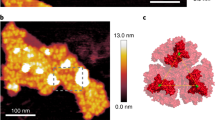
Prochlorococcus, the most abundant genus of photosynthetic organisms1, owes its remarkably large depth distribution in the oceans to the occurrence of distinct genotypes adapted to either low- or high-light niches2,3. The pcb genes, encoding the major chlorophyll-binding, light-harvesting antenna proteins in this genus4, are present in multiple copies in low-light strains but as a single copy in high-light strains5. The basis of this differentiation, however, has remained obscure. Here we show that the moderate low-light-adapted strain Prochlorococcus sp. MIT 9313 has one iron-stress-induced pcb gene encoding an antenna protein serving photosystem I (PSI)—comparable to isiA genes from cyanobacteria6,7—and a constitutively expressed pcb gene encoding a photosystem II (PSII) antenna protein. By comparison, the very low-light-adapted strain SS120 has seven pcb genes encoding constitutive PSI and PSII antennae, plus one PSI iron-regulated pcb gene, whereas the high-light-adapted strain MED4 has only a constitutive PSII antenna. Thus, it seems that the adaptation of Prochlorococcus to low light environments has triggered a multiplication and specialization of Pcb proteins comparable to that found for Cab proteins in plants and green algae8.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Partensky, F., Hess, H. R. & Vaulot, D. Prochlorococcus, a marine photosynthetic prokaryote of global significance. Microbiol. Mol. Biol. Rev. 63, 106–127 (1999)
Moore, L. R., Rocap, G. & Chisholm, S. W. Physiology and molecular phylogeny of coexisting Prochlorococcus ecotypes. Nature 393, 464–467 (1998)
Moore, L. R. & Chisholm, S. W. Photophysiology of the marine cyanobacterium Prochlorococcus: Ecotypic differences among cultured isolates. Limnol. Oceanogr. 44, 628–638 (1999)
La Roche, J. et al. Independent evolution of the prochlorophyte and green plant chlorophyll a/b light-harvesting proteins. Proc. Natl Acad. Sci. USA 93, 15244–15248 (1996)
Garczarek, L. et al. Expression and phylogeny of the multiple antenna genes of the low-light-adapted strain Prochlorococcus marinus SS120. Plant Mol. Biol. 46, 683–693 (2001)
Bibby, T. S., Nield, J. & Barber, J. Iron deficiency induces the formation of an antenna ring around trimeric photosystem I in cyanobacteria. Nature 412, 743–745 (2001)
Boekema, E. J. et al. A giant chlorophyll-protein complex induced by iron deficiency in cyanobacteria. Nature 412, 745–748 (2001)
Green, B. R. & Durnfold, D. G. The chlorophyll-carotenoid proteins of oxygenic photosynthesis. Annu. Rev. Plant Physiol. Plant Mol. Biol. 47, 685–714 (1996)
Bibby, T. S., Nield, J., Partensky, F. & Barber, J. Oxyphotobacteria: Antenna ring around PSI. Nature 413, 590 (2001)
Jordan, P. et al. Three-dimensional structure of cyanobacterial photosystem I at 2.5 Å resolution. Nature 411, 909–917 (2001)
Kamiya, N. & Shen, J.-R. Crystal structure of oxygen-evolving photosystem II from Thermosynechococcus vulcanus at 3.7 Å resolution. Proc. Natl Acad. Sci. USA 100, 98–102 (2003)
Strauss, N. A. in The Molecular Biology of Cyanobacteria (ed. Bryant, D. A.) 731–750 (Kluwer, Dordrecht, The Netherlands, 1994)
Partensky, F. & Garczarek, L. in Photosynthesis in Algae. Advances in Photosynthesis Series (ed. Larkum, A. W. D.) (Kluwer, Dordrecht, The Netherlands, in the press)
Sidler, W. in The Molecular Biology of Cyanobacteria Vol. 14 (ed. Bryant, D. A.) 139–216 (Kluwer, Dordrecht, The Netherlands, 1994)
Partensky, F. et al. The divinyl-chlorophyll a/b-protein complexes of two strains of the oxyphototrophic marine prokaryote Prochlorococcus—Characterization and response to changes in growth irradiance. Photosynth. Res. 51, 209–222 (1997)
Hankamer, B. et al. Isolation and biochemical characterization of monomeric and dimeric photosystem II complexes from spinach and their relevance to the organisation of photosystem II in vivo. Eur. J. Biochem. 243, 422–429 (1997)
Bhaya, D. et al. Isolation of regulated genes of the cyanobacterium Synechocystis sp. strain PCC 6803 by differential display. J. Bacteriol. 182, 5692–5699 (2000)
Mary, I. & Vaulot, D. Two-component systems in Prochlorococcus marinus: Genomic analysis and differential expression under stress. FEMS Microbiol. (in the press)
van Heel, M., Harauz, G. & Orlova, E. V. A new generation of the IMAGIC image processing system. J. Struct. Biol. 116, 17–24 (1996)
van Heel, M. & Frank, J. Use of multivariate statistics in analysing the images of biological macromolecules. Ultramicroscopy 6, 187–194 (1981)
Acknowledgements
J.B. thanks the Biotechnology and Biological Sciences Research Council for financial support. J.N. holds a University Research Fellowship of The Royal Society. F.P. and I.M. were supported by the EU FPS program Margenes, and I.M. was awarded a Ph.D. grant from Région Bretagne. We thank F. Le Gall for preparing large volumes of Prochlorococcus cultures.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Rights and permissions
About this article
Cite this article
Bibby, T., Mary, I., Nield, J. et al. Low-light-adapted Prochlorococcus species possess specific antennae for each photosystem. Nature 424, 1051–1054 (2003). https://doi.org/10.1038/nature01933
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature01933
This article is cited by
-
Phylogenetic and spectroscopic insights on the evolution of core antenna proteins in cyanobacteria
Photosynthesis Research (2023)
-
Structure of a tetrameric photosystem I from a glaucophyte alga Cyanophora paradoxa
Nature Communications (2022)
-
Phycobilisome light-harvesting efficiency in natural populations of the marine cyanobacteria Synechococcus increases with depth
Communications Biology (2022)
-
Remembering James Barber (1940–2020)
Photosynthesis Research (2022)
-
Independent iron and light limitation in a low-light-adapted Prochlorococcus from the deep chlorophyll maximum
The ISME Journal (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.