Abstract
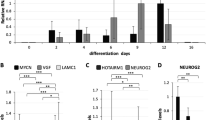
MicroRNAs (miRNAs) are phylogenetically widespread small RNAs of 18–25 nucleotides in length, and are found in animals and plants. These small RNAs can regulate gene expression at a translational level through interactions with their target messenger RNAs, and they have a role in the development of Caenorhabditis elegans and plants. Although more than two hundred miRNAs have been found in mammals, their mRNA targets remain to be identified. Here, we demonstrate that the expression of Hes1, basic helix–loop–helix transcriptional repressor, is regulated by miRNA-23 (miR-23) in NT2 cells. miR-23 is almost complementary to part of the coding region, just upstream of the termination codon, of Hes1 mRNA. Reduction in the level of miR-23 by small interfering RNAs resulted in the accumulation of Hes1, and hindered the retinoic-acid-induced neuronal differentiation of NT2 cells. Thus, our results indicate that miR-23 regulates the expression of Hes1 at the post-transcriptional level, and participates in retinoic-acid-induced neuronal differentiation of NT2 cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Eddy, S. R. Non-coding RNA genes and the modern RNA world. Nature Rev. Genet. 2, 919–929 (2001)
Lee, R. C., Feinbaum, R. L. & Ambros, V. The C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14. Cell 75, 843–854 (1993)
Reinhart, B. J. et al. The 21-nucleotide let-7 RNA regulates developmental timing in C. elegans. Nature 403, 901–906 (2000)
Pasquinelli, A. E. et al. Conservation of the sequence and temporal expression of let-7 heterochronic regulatory RNA. Nature 408, 86–89 (2000)
Slack, F. J. et al. The lin-41 RBCC gene acts in the C. elegans heterochronic pathway between the let-7 regulatory RNA and the LIN-29 transcription factor. Mol. Cell 5, 659–669 (2000)
Lau, N. C., Lim, L. P., Weinstein, E. G. & Bartel, D. P. An abundant class of tiny RNAs with probable regulatory roles in C. elegans. Science 294, 858–862 (2001)
Lee, R. C. & Ambros, V. An extensive class of small RNAs in Caenorhabditis elegans. Science 294, 862–864 (2001)
Lagos-Quintana, M. et al. Identification of novel genes coding for small expressed RNAs. Science 294, 853–858 (2001)
Mourelatos, Z. et al. miRNPs: a novel class of ribonucleoproteins containing numerous microRNAs. Genes Dev. 16, 720–728 (2002)
Reinhart, B. J., Weinstein, E. G., Rhoades, M. W., Bartel, B. & Bartel, D. P. MicroRNAs in plants. Genes Dev. 16, 1616–1626 (2002)
Lagos-Quintana, M. et al. Identification of tissue-specific microRNAs from mouse. Curr. Biol. 12, 735–739 (2002)
Rhoades, M. W. et al. Prediction of plant microRNA targets. Cell 110, 513–520 (2002)
Lagos-Quintana, M., Rauhut, R., Meyer, J., Borkhardt, A. & Tuschl, T. New microRNAs from mouse and human. RNA 9, 175–179 (2003)
Lim, L. P., Glasner, M. E., Yekta, S., Burge, C. B. & Bartel, D. P. Vertebrate microRNA genes. Science 299, 1540 (2003)
Lim, L. P. et al. The microRNAs of Caenorhabditis elegans. Genes Dev. 17, 991–1008 (2003)
Dostie, J., Mourelatos, Z., Yang, M., Sharma, A. & Dreyfuss, G. Numerous microRNPs in neuronal cells containing novel microRNAs. RNA 9, 180–186 (2003)
Llave, C., Xie, Z., Kasschau, K. D. & Carrington, J. C. Cleavage of Scarecrow-like mRNA targets directed by a class of Arabidopsis miRNA. Science 297, 2053–2056 (2002)
Tang, G., Reinhart, B. J., Bartel, D. P. & Zamore, P. D. A biochemical framework for RNA silencing in plants. Genes Dev. 17, 49–63 (2003)
Olsen, P. H. & Ambros, V. The lin-4 regulatory RNA controls developmental timing in Caenorhabditis elegans by blocking LIN-14 protein synthesis after the initiation of translation. Dev. Biol. 216, 671–680 (1999)
Seggerson, K., Tang, L. & Moss, E. G. Two genetic circuits repress the Caenorhabditis elegans heterochronic gene lin-28 after translation initiation. Dev. Biol. 243, 215–225 (2002)
Lee, Y., Jeon, K., Lee, J. T., Kim, S. & Kim, V. N. MicroRNA maturation: stepwise processing and subcellular localization. EMBO J. 21, 4663–4670 (2002)
Grishok, A. et al. Genes and mechanisms related to RNA interference regulate expression of the small temporal RNAs that control C. elegans developmental timing. Cell 106, 23–34 (2001)
Hutvagner, G. et al. A cellular function for the RNA-interference enzyme Dicer in the maturation of the let-7 small temporal RNA. Science 293, 834–838 (2001)
Ketting, R. F. et al. Dicer functions in RNA interference and in synthesis of small RNA involved in developmental timing in C. elegans. Genes Dev. 15, 2654–2659 (2001)
Martinez, J., Patkaniowska, A., Urlaub, H., Luhrmann, R. & Tuschl, T. Single-stranded antisense siRNAs guide target RNA cleavage in RNAi. Cell 110, 563–574 (2002)
Zeng, Y., Wagner, E. J. & Cullen, B. R. Both natural and designed micro RNAs can inhibit the expression of cognate mRNAs when expressed in human cells. Mol. Cell 9, 1327–1333 (2002)
Zeng, Y. & Cullen, B. R. Sequence requirements for microRNA processing and function in human cells. RNA 9, 112–123 (2003)
Ohtsuka, T. et al. Hes1 and Hes5 as notch effectors in mammalian neuronal differentiation. EMBO J. 18, 2196–2207 (1999)
Kageyama, R., Ohtsuka, T. & Tomita, K. The bHLH gene Hes1 regulates differentiation of multiple cell types. Molecules Cells 29, 1–7 (2000)
Ohtsuka, T., Sakamoto, M., Guillemot, F. & Kageyama, R. Roles of the basic helix-loop-helix genes Hes1 and Hes5 in expansion of neural stem cells of the developing brain. J. Biol. Chem. 276, 30467–30474 (2001)
Cheung, W. M., Chu, P. W., Lung, C. H. & Ip, N. Y. Expression of retinoid receptors during the retinoic acid-induced neuronal differentiation of human embryonal carcinoma cells. J. Neurochem. 75, 34–40 (2000)
Elbashir, S. M. et al. Duplexes of 21-nucleotide RNAs mediate RNA interference in mammalian cell culture. Nature 411, 494–498 (2001)
McManus, M. T. & Sharp, P. A. Gene silencing in mammals by small interfering RNAs. Nature Rev. Genet. 3, 737–747 (2002)
Fire, A. et al. Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature 391, 806–811 (1998)
Hammond, S. M., Caudy, A. A. & Hannon, G. J. Post-transcriptional gene silencing by double-stranded RNA. Nature Rev. Genet. 2, 110–119 (2001)
Pleasure, S. J. & Lee, V. M. NTera 2 cells: a human cell line which displays characteristics expected of a human committed neuronal progenitor cell. J. Neurosci. Res. 35, 585–602 (1993)
Kurie, J. M. et al. Cooperation between retinoic acid and phorbol esters enhances human teratocarcinoma differentiation. Differentiation 54, 115–122 (1993)
Hutvagner, G. & Zamore, P. D. A microRNA in a multiple-turnover RNAi enzyme complex. Science 297, 2056–2060 (2002)
Brennecke, J., Hipfner, D. R., Stark, A., Russell, R. B. & Cohen, S. M. Bantam encodes a developmentally regulated microRNA that controls cell proliferation and regulates the proapoptotic gene hid in Drosophila. Cell 113, 25–36 (2003)
Kawasaki, H. et al. Distinct roles of the co-activators p300 and CBP in retinoic-acid-induced F9-cell differentiation. Nature 393, 284–289 (1998)
Acknowledgements
The authors thank L. Nelson for critical reading of the manuscript. This research was supported by grants from the Ministry of Economy, Trade and Industry (METI) of Japan, and by a Grant-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Culture (MEXT) of Japan.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Additional information
In this article, the messenger RNA that is identified to be a target of microRNA-23 (miR-23) is from the gene termed human ‘homolog of ES1’ (HES1), accession number Y07572, and not from the gene encoding the transcriptional repressor ‘Hairy enhancer of split’ HES1 (accession number NM_00524) as stated in our paper. We incorrectly identified the gene because of the confusing nomenclature. The function of HES1 Y07572 is unknown but the encoded protein shares homology with a protein involved in isoprenoid biosynthesis. Our experiments in NT2 cells had revealed that the protein levels of the repressor Hes1 were diminished by miR-23. Although we have unpublished data that suggest the possibility that miR-23 might also interact with Hes1 repressor mRNA, the explanation for the finding that the level of repressor Hes1 protein decreases in response to miR-23 remains undefined with respect to mechanism and specificity. Given the interpretational difficulties resulting from our error, we respectfully retract the present paper. Further studies aimed at clarifying the physiological role of miR-23 will be submitted to a peer-reviewed journal subject to the outcome of our ongoing research.
Supplementary information
About this article
Cite this article
Kawasaki, H., Taira, K. RETRACTED ARTICLE: Hes1 is a target of microRNA-23 during retinoic-acid-induced neuronal differentiation of NT2 cells. Nature 423, 838–842 (2003). https://doi.org/10.1038/nature01730
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature01730
This article is cited by
-
RETRACTED ARTICLE: LncRNA XIST inhibits ovarian cancer cell growth and metastasis via regulating miR-150-5p/PDCD4 signaling pathway
Naunyn-Schmiedeberg's Archives of Pharmacology (2021)
-
MiR-34 and MiR-200: Regulator of Cell Fate Plasticity and Neural Development
NeuroMolecular Medicine (2019)
-
MicroRNA-23a regulates epithelial-to-mesenchymal transition in endometrial endometrioid adenocarcinoma by targeting SMAD3
Cancer Cell International (2016)
-
miR-23a/b regulates the balance between osteoblast and adipocyte differentiation in bone marrow mesenchymal stem cells
Bone Research (2016)
-
Diverse roles of miR-335 in development and progression of cancers
Tumor Biology (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.