Abstract
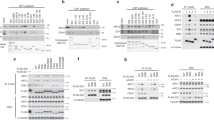
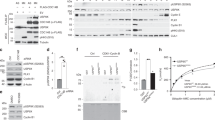
Cks proteins are small evolutionarily conserved proteins that interact genetically and physically with cyclin-dependent kinases. However, in spite of a large body of genetic, biochemical and structural research, no compelling unifying model of their functions has emerged1,2. Here we show, by investigating the essential role of Cks1 in Saccharomyces cerevisiae, that the protein is primarily involved in promoting mitosis by modulating the transcriptional activation of the APC/C protein–ubiquitin ligase activator Cdc20. Cks1 is required for both the periodic dissociation of Cdc28 kinase from the CDC20 promoter and the periodic association of the proteasome with the promoter. We propose that the essential role of Cks1 is to recruit the proteasome to, and/or dissociate the Cdc28 kinase from, the CDC20 promoter, thus facilitating transcription by remodelling transcriptional complexes or chromatin associated with the CDC20 gene.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Endicott, J. A. & Nurse, P. The cell cycle and suc1: From structure to function? Structure 3, 321–325 (1995)
Pines, J. Cell cycle: Reaching for a role for the Cks proteins. Curr. Biol. 6, 1399–1402 (1996)
Tang, Y. & Reed, S. I. The cdk-associated protein Cks1 functions both in G1 and G2 in Saccharomyces cerevisiae. Genes Dev. 7, 822–832 (1993)
Kaiser, P. et al. Cyclin-dependent kinase and Cks/Suc1 interact with the proteasome in yeast to control proteolysis of M-phase targets. Genes Dev. 13, 1190–1202 (1999)
Ghiara, J. B. et al. A cyclin B homolog in S. cerevisiae: Chronic activation of the Cdc28 protein kinase by cyclin prevents exit from mitosis. Cell 65, 163–174 (1991)
Amon, A., Tyers, M., Futcher, B. & Nasmyth, K. Mechanisms that help the yeast cell cycle clock tick: G2 cyclins transcriptionally activate G2 cyclins and repress G1 cyclins. Cell 74, 993–1007 (1993)
Nasmyth, K. Control of the yeast cell cycle by the cdc28 protein kinase. Curr. Opin. Cell Biol. 5, 166–179 (1993)
Visintin, R., Prinz, S. & Amon, A. Cdc20 and cdh1: A family of substrate-specific activators of APC-dependent proteolysis. Science 278, 460–463 (1997)
Lim, H. H., Goh, P.-Y. & Surana, U. Cdc20 is essential for the cyclosome-mediated proteolysis of both Pds1 and clb2 during M phase in budding yeast. Curr. Biol. 8, 231–234 (1998)
Jensen, S., Segal, M., Clarke, D. J. & Reed, S. I. A novel role of the budding yeast separin Esp1 in anaphase spindle elongation: evidence that proper spindle association of Esp1 is regulated by Pds1. J. Cell Biol. 152, 27–40 (2001)
Spellman, P. T. et al. Comprehensive identification of cell cycle-regulated genes of the yeast Saccharomyces cerevisiae by microarray hybridization. Mol. Biol. Cell 9, 3273–3297 (1998)
Maher, M., Cong, F., Kindelberger, D., Nasmyth, K. & Dalton, S. Cell cycle-regulated transcription of the CLB2 gene is dependent on Mcm1 and a ternary complex factor. Mol. Cell. Biol. 15, 3129–3137 (1995)
Pic, A. et al. The forkhead protein Fkh2 is a component of the yeast cell cycle transcription factor SFF. EMBO J. 19 14, 3750–3761 (2000)
Koranda, M., Schleiffer, A., Endler, L. & Ammerer, G. Forkhead-like transcription factors recruit Ndd1 to the chromatin of G2/M-specific promoters. Nature 406, 94–98 (2000)
Zhu, G. et al. Two yeast forkhead genes regulate the cell cycle and pseudohyphal growth. Nature 406, 90–94 (2000)
Bourne, Y. et al. Crystal structure and mutational analysis of the human cdk2 kinase complex with cell cycle-regulatory protein CksHs1. Cell 84, 863–874 (1996)
Ferdous, A., Gonzalez, F., Sun, L., Kodadek, T. & Johnston, S. A. The 19S regulatory particle of the proteasome is required for efficient transcription elongation by RNA polymerase. Mol. Cell 7, 981–991 (2001)
Gonzalez, F., Delahodde, A., Kodadek, T. & Johnston, S. A. Recruitment of a 19S proteasome subcomplex to an activated promoter. Science 296, 548–550 (2002)
Hilt, W., Enenkel, C., Gruhler, A., Singer, T. & Wolf, D. H. The PRE4 gene codes for a subunit of the yeast proteasome necessary for peptidylglutamyl-peptide-hydrolyzing activity. Mutations link proteasome and stress- and ubiquitin-dependent proteolysis. J. Biol. Chem. 268, 3479–3486 (1993)
Guthrie, C., Fink, G. R. Guide to Yeast Genetics and Molecular Biology 194 (Academic, San Diego, California, 1991)
Reed, S. I., Hadwiger, J. A. & Lorincz, A. Protein kinase activity associated with the product of the yeast cell division cycle gene CDC28. Proc. Natl Acad. Sci. USA 82, 4055–4059 (1985)
Collart, M. A. & Oliviero, S. in Current Protocols in Molecular Biology Vol. 2, 13.12 (Current Protocols, Wiley and Sons, New York, 1993)
Kaiser, P., Flick, K., Wittenberg, C. & Reed, S. I. Regulation of transcription by ubiquitination without proteolysis: Cdc34/SCFMet30-mediated inactivation of the transcription factor Met4. Cell 102, 303–314 (2000)
Acknowledgements
We thank S. A. Johnston for the sug1-25 strain, M. Koranda and G. Ammerer for the tagged Mcm1 and Ndd1 strains, K. Nasymth for the myc18-CDC20 allele, D. Wolff for the pre1 and pre4 strains used to construct the double mutant in our strain background, and K. Flick and S. Haase for discussions and technical advice about northern blotting experiments and ChIP assays. This work was supported by a grant from the NIH to S.I.R. M.C.M. acknowledges fellowship support from the International Agency for Research on Cancer and the Swiss National Science Foundation, and P.K. acknowledges fellowship support from the Austrian Science Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Morris, M., Kaiser, P., Rudyak, S. et al. Cks1-dependent proteasome recruitment and activation of CDC20 transcription in budding yeast. Nature 423, 1009–1013 (2003). https://doi.org/10.1038/nature01720
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature01720
This article is cited by
-
Increased expression of Cks1 protein is associated with lymph node metastasis and poor prognosis in nasopharyngeal carcinoma
Diagnostic Pathology (2017)
-
Cks confers specificity to phosphorylation-dependent CDK signaling pathways
Nature Structural & Molecular Biology (2013)
-
Cdk1 promotes kinetochore bi-orientation and regulates Cdc20 expression during recovery from spindle checkpoint arrest
The EMBO Journal (2012)
-
The E2F functional analogue SBF recruits the Rpd3(L) HDAC, via Whi5 and Stb1, and the FACT chromatin reorganizer, to yeast G1 cyclin promoters
The EMBO Journal (2009)
-
Coupling and coordination in gene expression processes: a systems biology view
Nature Reviews Genetics (2008)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.