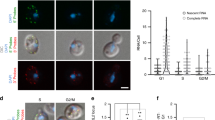
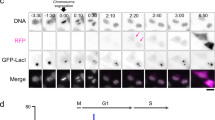
Abstract
In animal cells, the process of DNA replication takes place in a programmed manner, with each gene region designated to replicate at a fixed time slot in S phase. Housekeeping genes undergo replication in the first half of S phase in all cell types, whereas the replication of many tissue specific genes is developmentally controlled, being late in most tissues but early in the tissue of expression1. Here we employ nuclear DNA injection as an experimental system to test whether this phenomenon is due to differences in the ability to set up transcriptional competence during S phase2,3. Our results show that, regardless of sequence, exogenous genes are a better template for transcription when injected into nuclei of cells in early as opposed to late S phase, and this expression state, once initiated, is preserved after cell division. DNA injected in late S phase is apparently repressed because it is packaged into chromatin containing deacetylated histones, and the same is true for late replicating chromosomal DNA. These findings suggest a mechanistic connection between replication timing and gene expression that might help to explain how epigenetic states can be maintained in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Simon, I. & Cedar, H. in DNA Replication in Eukaryotic Cells (ed. DePamphilis, M. L.) 387–408 (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, 1996)
Gottesfeld, J. & Bloomer, L. S. Assembly of transcriptionally active 5S RNA gene chromatin in vitro. Cell 28, 781–791 (1982)
Brown, D. D. The role of stable complexes that repress and activate eucaryotic genes. Cell 37, 359–365 (1984)
Roth, S. Y. & Allis, C. D. Histone acetylation and chromatin assembly: a single escort, multiple dances? Cell 87, 5–8 (1996)
Grunstein, M. Histone acetylation in chromatin structure and transcription. Nature 389, 349–352 (1997)
Ohe, Y., Zhao, D., Saijo, N. & Podack, E. R. Construction of a novel bovine papillomavirus vector without detectable transforming activity suitable for gene transfer. Hum. Gene Ther. 6, 325–333 (1995)
Jeppesen, P. & Turner, B. M. The inactive X chromosome in female mammals is distinguished by a lack of histone H4 acetylation, a cytogenetic marker for gene expression. Cell 74, 281–289 (1993)
Workman, J. L. & Kingston, R. E. Alteration of nucleosome structure as a mechanism of transcriptional regulation. Annu. Rev. Biochem. 67, 545–579 (1998)
Lee, D. Y., Hayes, J. J., Pruss, D. & Wolffe, A. P. A positive role for histone acetylation in transcription factor access to nucleosomal DNA. Cell 72, 73–84 (1993)
Sobel, R. E., Cook, R. G., Perry, C. A., Annunziato, A. T. & Allis, C. D. Conservation of deposition-related acetylation sites in newly synthesized histones H3 and H4. Proc. Natl Acad. Sci. USA 92, 1237–1247 (1995)
Wolffe, A. P. in DNA Replication in Eukaryotic Cells (ed. DePamphilis, M. L.) 271–293 (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, 1996)
Rountree, M. R., Bachman, K. E. & Baylin, S. B. DNMT1 binds HDAC2 and a new co-repressor, DMAP1, to form a complex at replication foci. Nature Genet. 25, 269–277 (2000)
Allshire, R. & Bickmore, W. Pausing for thought on the boundaries of imprinting. Cell 102, 705–708 (2000)
Weintraub, H. Assembly of an active chromatin structure during replication. Nucleic Acids Res. 7, 781–792 (1979)
Solomon, M. J. & Varshavsky, A. A nuclease-hypersensitive region forms de novo after chromosome replication. Mol. Cell. Biol. 7, 3822–3825 (1987)
Simon, I. et al. Developmental regulation of DNA replication timing at the human β globin locus. EMBO J. 20, 6150–6157 (2001)
Cimbora, D. M. et al. Long-distance control of origin choice and replication timing in the human beta-globin locus are independent of the locus control region. Mol. Cell. Biol. 20, 5581–5591 (2000)
Donaldson, A. D. et al. CLB5-dependent activation of late replication origins in S. cerevisiae. Mol. Cell 2, 173–182 (1998)
Santocanale, C. & Diffley, J. F. A Mec1- and Rad53-dependent checkpoint controls late-firing origins of DNA replication. Nature 395, 615–618 (1998)
Shirahige, K. et al. Regulation of DNA-replication origins during cell-cycle progression. Nature 395, 618–621 (1998)
Siegfried, Z. et al. DNA methylation represses transcription in vivo. Nature Genet. 22, 203–206 (1999)
Yang, L., Li, R., Mohr, I. J., Clark, R. & Botchan, M. R. Activation of BPV-1 replication in vitro by the transcription factor E2. Nature 353, 628–632 (1991)
Graessmann, M. & Graessmann, A. Microinjection of tissue culture cells. Methods Enzymol. 101, 482–492 (1983)
Kutsukake, M., Komatsu, A., Yamamoto, D. & Ishiwa-Chigusa, S. A tyramine receptor gene mutation causes a defective olfactory behavior in Drosophila melanogaster. Gene 245, 31–42 (2000)
Simon, I. et al. Asynchronous replication of imprinted genes is established in the gametes and maintained during development. Nature 401, 929–932 (1999)
Brandeis, M. & Hunt, T. The proteolysis of mitotic cyclins in mammalian cells persists from the end of mitosis until the onset of S phase. EMBO J. 15, 5280–5289 (1996)
Hebbes, T. R., Clayton, A. L., Thorne, A. W. & Crane-Robinson, C. Core histone hyperacetylation co-maps with generalized DNase I sensitivity in the chicken beta-globin chromosomal domain. EMBO J. 13, 1823–1830 (1994)
Eden, S., Hashimshony, T., Keshet, I., Cedar, H. & Thorne, A. W. DNA methylation models histone acetylation. Nature 394, 842–843 (1998)
Acknowledgements
We thank B. Joshua, S. Beyit and B. Giloh for their help in developing the injection technology, and O. Meyuhas, M. Brandeis, E. Bachrach and D. Yaffe for providing vectors used in this study. This research was supported by grants from the N.I.H., the Israel Academy of Sciences and the Israel Cancer Research Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Rights and permissions
About this article
Cite this article
Zhang, J., Xu, F., Hashimshony, T. et al. Establishment of transcriptional competence in early and late S phase. Nature 420, 198–202 (2002). https://doi.org/10.1038/nature01150
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature01150
This article is cited by
-
Chromosomal coordination and differential structure of asynchronous replicating regions
Nature Communications (2021)
-
Replication timing and epigenome remodelling are associated with the nature of chromosomal rearrangements in cancer
Nature Communications (2019)
-
Control of DNA replication timing in the 3D genome
Nature Reviews Molecular Cell Biology (2019)
-
Delaying histone deacetylase response to injury accelerates conversion into repair Schwann cells and nerve regeneration
Nature Communications (2017)
-
Linking replication stress with heterochromatin formation
Chromosoma (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.